The Missing Cap: How Actin Cap Deficiency Drives Stem Cell Fate and Differentiation Failure
This article provides a comprehensive analysis of the actin cap's critical role in stem cell biology, specifically focusing on how its absence or dysfunction impairs differentiation.
The Missing Cap: How Actin Cap Deficiency Drives Stem Cell Fate and Differentiation Failure
Abstract
This article provides a comprehensive analysis of the actin cap's critical role in stem cell biology, specifically focusing on how its absence or dysfunction impairs differentiation. We explore the foundational mechanisms linking the perinuclear actin cap to nuclear mechanotransduction and gene regulation. Methodological approaches for detecting, quantifying, and manipulating the actin cap are detailed, alongside troubleshooting strategies for common experimental challenges. The review compares actin cap dynamics across stem cell types and validates its function as a master regulator of differentiation potential, offering critical insights for regenerative medicine and drug development targeting cellular reprogramming.
Decoding the Actin Cap: The Structural Guardian of Stem Cell Fate
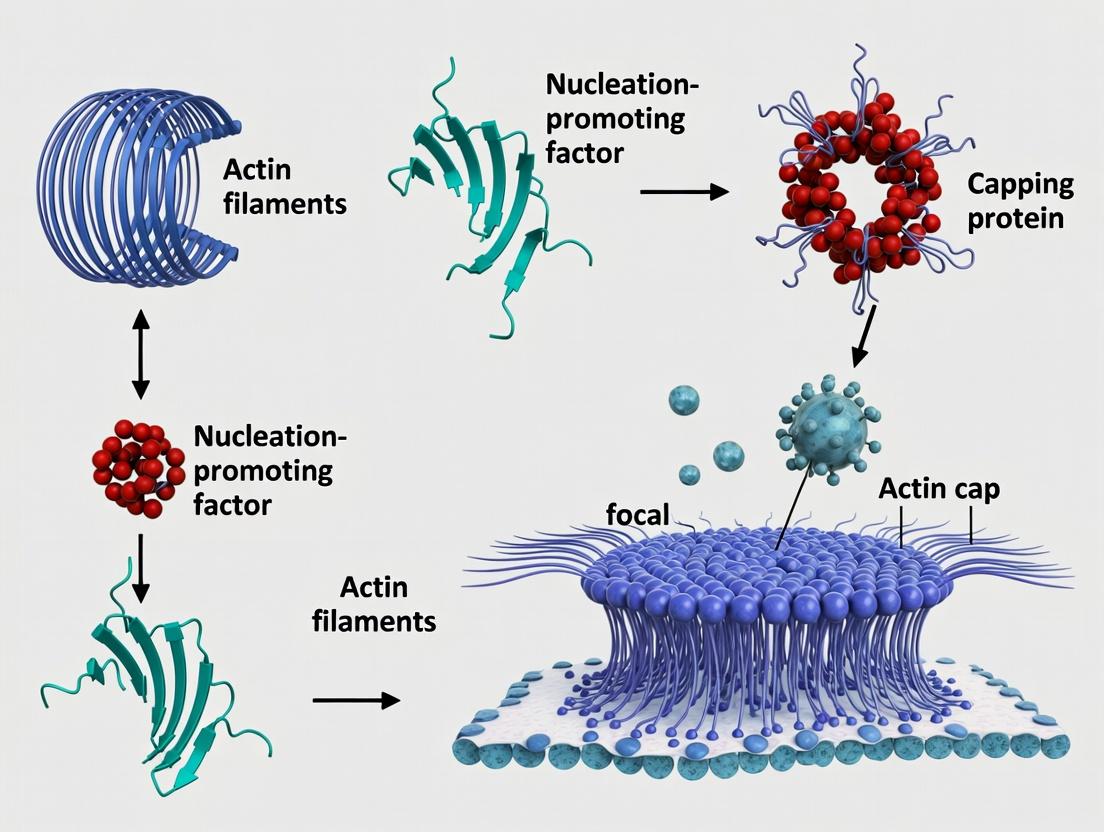
The perinuclear actin cap is a specialized cytoskeletal structure that tightly overlays the nucleus in specific cell types, including stem cells and fibroblasts. It is composed of thick, parallel actin bundles anchored to the apical nuclear envelope via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes. This primer details its architecture and components, framed within the critical context of research exploring the consequences of its absence on stem cell differentiation—a key determinant of cell fate, mechanotransduction, and nuclear morphology.
Unique Architecture of the Actin Cap
The actin cap's architecture is distinct from the basal actin cortex.
Key Features:
- Location: Apical to the nucleus, running along the long axis of polarized cells.
- Composition: Composed of stable, contractile actomyosin bundles (stress fibers).
- Anchorage: Terminates at focal adhesions at the cell ends and is directly linked to the nuclear envelope.
- Nuclear Coupling: Integrates the nucleus into the cellular mechanotransduction apparatus via Nesprin-2G/SUN2 LINC complexes. This physical link transmits forces directly to the nuclear lamina and interior.
Diagram 1: Actin Cap Architecture & Nuclear Linkage
Core Molecular Components
The integrity of the actin cap depends on a defined set of molecular players.
Table 1: Core Components of the Perinuclear Actin Cap
| Component | Type | Primary Function in Actin Cap | Consequence of Loss/Inhibition |
|---|---|---|---|
| Actin (F-actin) | Cytoskeletal Polymer | Primary structural scaffold; forms parallel, apical bundles. | Cap dissolution, loss of nuclear shaping. |
| Non-Muscle Myosin IIA | Molecular Motor | Provides contractility; essential for bundle tension & stability. | Reduced cap tension, impaired nuclear flattening. |
| Nesprin-2G (SYNE2) | Outer Nuclear Membrane Protein | Actin-binding KASH protein; primary anchor for cap fibers to the nucleus. | Uncoupled nucleus, failed cap assembly. |
| SUN2 | Inner Nuclear Membrane Protein | Binds Nesprin in perinuclear space; part of LINC complex. | Disrupted force transmission to lamina. |
| Lamin A/C | Nuclear Lamina Component | Provides mechanical stability to nucleus; downstream of LINC forces. | Nuclear softening, aberrant deformation. |
| Formin (mDia1/2) | Actin Nucleator/Polymerase | Promotes linear actin polymerization for cap fiber formation. | Defective actin bundle assembly. |
Methodologies for Studying the Actin Cap
Visualization Protocol
- Fixation: Use 4% PFA for 15 min at RT. For preserving actin structures, a brief pre-extraction with 0.5% Triton X-100 in cytoskeleton buffer (10 mM MES, 150 mM NaCl, 5 mM EGTA, 5 mM glucose, 5 mM MgCl2) before fixation may be required.
- Staining: Use Phalloidin (conjugated to Alexa Fluor 488/568/647) at 1:200-1:500 dilution to label F-actin. Co-stain with antibodies against Nesprin-2G or SUN2 and a nuclear marker (DAPI or Lamin A/C).
- Microscopy: Image using high-resolution confocal or TIRF microscopy. Z-stacks are essential to distinguish apical actin cap fibers from basal stress fibers. 3D reconstruction confirms perinuclear localization.
Functional Disruption Protocols
- LINC Complex Disruption: Transfect cells with dominant-negative KASH (ΔKASH) construct or use siRNA/shRNA against SYNE2 (Nesprin-2G) or SUN2.
- Actin Cap Dissolution: Treat cells with low-dose (e.g., 100 nM) Latrunculin B for 30-60 minutes to depolymerize actin, or Blebbistatin (50 µM) for 1-2 hours to inhibit Myosin II contractility.
- Assessment: Quantify cap integrity (percentage of cells with clear apical actin bundles), nuclear height (using AFM or confocal cross-section), and changes in differentiation markers.
Quantifying Actin Cap Phenotypes in Stem Cells
Workflow for differentiation studies:
- Culture: Maintain stem cells (e.g., MSCs, iPSCs) on substrates of defined stiffness (e.g., 1 kPa vs. 50 kPa PA gels).
- Disrupt: Introduce actin cap-disrupting agents (siRNA, drugs) in growth media.
- Differentiate: Switch to differentiation media (osteogenic, adipogenic).
- Analyze: After 5-14 days, fix cells and quantify:
- Cap Status: Phalloidin intensity ratio (apical/nuclear region vs. whole cell).
- Nuclear Morphometrics: Area, perimeter, height (from Z-stacks).
- Lineage Commitment: % cells positive for markers (e.g., Runx2/Osteocalcin for osteogenesis, PPARγ/Adiponectin for adipogenesis).
Diagram 2: Workflow for Actin Cap Disruption in Differentiation Studies
The Scientist's Toolkit: Key Reagent Solutions
Table 2: Essential Research Reagents for Actin Cap Studies
| Reagent/Material | Supplier Examples | Function in Actin Cap Research |
|---|---|---|
| Phalloidin (Fluorescent conjugates) | Thermo Fisher, Cytoskeleton Inc. | High-affinity probe to selectively stain F-actin in cap fibers. |
| Anti-Nesprin-2G Antibody | Abcam, Santa Cruz Biotechnology | Validates LINC complex localization; confirms cap anchoring. |
| siRNA against SYNE2/SUN2 | Horizon Discovery, Sigma-Aldrich | For specific, transient knockdown of LINC components to disrupt cap. |
| pAAV-ΔKASH Plasmid | Addgene (Plasmid #87033) | Dominant-negative construct for potent LINC complex disruption. |
| Latrunculin B | Cayman Chemical, Tocris | Actin depolymerizing agent; used for acute cap dissolution. |
| Blebbistatin | Sigma-Aldrich, Torcis | Specific inhibitor of non-muscle myosin II; reduces cap contractility. |
| Tunable Polyacrylamide Gels | Matrigen (Softwell), In-house prep. | Substrates of defined stiffness to study mechanosensitive cap assembly. |
| Lamin A/C Antibody | Abcam, Cell Signaling Tech. | Assesses nuclear envelope response to cap-derived forces. |
Table 3: Key Quantitative Effects of Actin Cap Disruption in Stem Cells
| Measured Parameter | Control Cells (Cap Intact) | Cells with Disrupted Actin Cap | Measurement Method | Implication |
|---|---|---|---|---|
| Nuclear Height | ~3.5 ± 0.5 µm | Increases to ~6.0 ± 1.0 µm* | Confocal Z-section, AFM | Loss of compressive force flattens nucleus. |
| Cap Integrity Index | 1.0 (reference) | Decreases to 0.2 - 0.4* | Apical/Basal F-actin Ratio | Cap structure is severely compromised. |
| Osteogenic Efficiency | 70-80% ALP+ cells | Reduces to 20-30% ALP+ cells* | Alkaline Phosphatase (ALP) stain | Impairs mechano-induced osteogenesis. |
| Adipogenic Efficiency | 15-25% Lipid+ cells | Increases to 50-70% Lipid+ cells* | Oil Red O stain | Promotes default adipogenic fate. |
| Yes-Associated Protein (YAP) Nuclear Localization | High on stiff substrate | Significantly reduced* | Immunofluorescence, fractionation | Disrupts critical mechanotransduction pathway. |
*Representative data based on published studies. Actual values are cell-type and condition-dependent.
Context: Actin Cap Absence in Stem Cell Differentiation
The absence of a functional perinuclear actin cap has profound implications, shifting the paradigm from mere structural aberration to a direct modulator of cell fate.
- Mechanotransduction Failure: The uncoupled nucleus cannot properly transduce substrate mechanical cues (stiffness) into biochemical signals. This disrupts the nuclear shuttling of mechanosensitive transcription factors like YAP/TAZ.
- Altered Chromatin & Gene Expression: Loss of compressive tension can lead to nuclear volume expansion and potentially alter chromatin organization, affecting the accessibility of genes related to differentiation.
- Differentiation Bias: On stiff, osteogenic substrates, cap disruption biases mesenchymal stem cells (MSCs) away from osteogenic lineage and toward a default adipogenic fate. This demonstrates the cap's essential role in guiding lineage commitment in response to the physical microenvironment.
The perinuclear actin cap is a unique, architecturally defined mechanosensory organelle. Its core components—from apical actomyosin bundles to Nesprin-2G/SUN2 LINC complexes—form a continuous physical link from the extracellular matrix to the nuclear interior. Methodologies for its study require precise 3D visualization and targeted functional disruption. Critically, data from its absence underscore its non-redundant function: it is a master regulator of nuclear mechanics and a decisive factor in stem cell lineage specification, presenting a potential target for modulating cell fate in regenerative medicine and disease modeling.
This whitepaper details the structural and signaling machinery of the perinuclear actin cap, a specialized filamentous actin (F-actin) network that directly links the extracellular matrix (ECM) to the nuclear envelope. The core thesis framing this guide posits that the actin cap is a primary mechanosensory apparatus whose dysfunction or absence in stem cell niches critically impairs fate specification by disrupting the transduction of essential physical cues to chromatin. Understanding this nexus is paramount for controlling stem cell differentiation and developing novel mechano-based therapeutics.
Architectural and Molecular Composition of the Actin Cap
The actin cap is a dorsal, perinuclear bundle of actomyosin stress fibers that terminate at nuclear envelope embedded Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes.
Core Structural Components
| Component | Primary Isoforms/Proteins | Quantitative Measurement (Typical Range) | Function |
|---|---|---|---|
| Actin Filaments | F-actin (γ-actin enriched) | Fiber thickness: 100-400 nm | Provides tensile structure; transmits force |
| Myosin Motors | Non-muscle Myosin IIA/B (NMII) | Contraction force: ~1-10 nN/µm² | Generates actomyosin contractility |
| LINC Complex | Nesprin-1/2 (KASH domain), SUN1/2, Emerin | Force transduction: ~1-40 pN per complex | Bridges cytoskeleton to nucleoskeleton |
| Nuclear Lamina | Lamin A/C | Stiffness correlation: 2-20 kPa substrate range | Stabilizes nucleus; regulates chromatin |
Key Regulatory Proteins
| Protein Complex | Key Members | Effect on Cap Integrity |
|---|---|---|
| Formin Nucleators | mDia1, mDia2 | Promotes linear, unbranched F-actin growth |
| Arp2/3 Complex | ARPC2, ARPC3 | Generally antagonizes cap formation; promotes branched networks |
| Rho GTPase Pathway | RhoA, ROCK, LIMK, Cofilin | RhoA activity >0.5 relative units boosts cap formation |
Diagram 1: Core Actin Cap Assembly Pathway
Mechanotransduction Signaling Pathways to the Nucleus
Force transmission via the cap activates biochemical and biomechanical signaling cascades that regulate gene expression.
Primary Signaling Cascades
| Pathway | Initiating Signal | Key Transducers | Nuclear Outcome | Experimental Readout |
|---|---|---|---|---|
| YAP/TAZ | Cytoskeletal tension | LATS1/2, YAP/TAZ | Transcriptional co-activation (TEAD) | Nuclear/cytosolic YAP ratio >2 = active |
| MRTF-A/SRF | G-actin depletion | MRTF-A, SRF | Expression of cytoskeletal genes | SRF reporter activity (fold-change) |
| Nuclear Deformation | Direct physical force | Lamin A/C, Emerin | Chromatin remodeling, DNA damage | Lamin A phosphorylation (Ser22), γH2AX foci |
Diagram 2: Signaling Pathways from Cap to Nucleus
Experimental Protocols for Actin Cap Research
Protocol: Quantifying Actin Cap Integrity in Stem Cells
Objective: To visualize and quantify the presence and morphology of the actin cap.
- Cell Seeding: Plate human mesenchymal stem cells (hMSCs) on fibronectin-coated (10 µg/ml) substrates with tuned stiffness (1 kPa vs. 30 kPa).
- Fixation & Permeabilization: At 24h, fix with 4% PFA for 15 min, permeabilize with 0.1% Triton X-100 for 5 min.
- Staining:
- F-actin: Phalloidin-Alexa Fluor 488 (1:200, 30 min).
- Nucleus: DAPI (1 µg/ml, 5 min).
- Lamin A/C: Anti-Lamin A/C primary antibody (1:250, overnight at 4°C), secondary antibody Alexa Fluor 568 (1:500, 1h).
- Imaging: Acquire z-stacks (0.2 µm intervals) using a 63x/1.4 NA oil immersion confocal microscope. Capture dorsal (top) optical slices.
- Analysis: Use ImageJ/FIJI. Threshold dorsal F-actin signal above nucleus. Calculate Cap Integrity Index = (Dorsal F-actin fluorescence intensity) / (Total cellular F-actin intensity). Index >0.7 indicates robust cap.
Protocol: Disrupting Actin Cap to Test Differentiation
Objective: To assess the necessity of the actin cap for mechano-directed differentiation.
- Genetic Disruption (CRISPRi):
- Design sgRNAs targeting SYNE2 (Nesprin-2) or LMNA.
- Transduce hMSCs with dCas9-KRAB and sgRNA lentivirus.
- Validate knockdown by western blot (>70% reduction).
- Pharmacological Disruption:
- Treat hMSCs with 10 µM SMIFH2 (formin inhibitor) or 10 µM Y-27632 (ROCK inhibitor) for 24h.
- Differentiation Assay:
- Plate control and disrupted cells on osteogenic-stiff substrate (30 kPa).
- Maintain in osteogenic medium (β-glycerophosphate, ascorbic acid, dexamethasone) for 7-14 days.
- Outcome Measurement:
- Quantitative: qPCR for RUNX2, Osteocalcin. Alkaline phosphatase (ALP) activity assay (nmol pNP/min/µg protein).
- Expected Result: Cap-disrupted cells show >60% reduction in osteogenic markers vs. control on stiff substrate.
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Category | Specific Product Examples | Function in Actin Cap Research |
|---|---|---|
| Inhibitors (Small Molecules) | SMIFH2 (Formin), Y-27632 (ROCK), Latrunculin A/B (Actin polymerization) | Disrupt cap assembly or tension to test functional necessity. |
| Cytoskeletal Labels | SiR-Actin (live), Phalloidin conjugates (fixed), GFP-LifeAct | Visualize F-actin dynamics and cap architecture. |
| LINC Complex Disruptors | Dominant-negative KASH overexpression, CRISPRi/a for Nesprins/SUNs | Genetically uncouple the actin cap from the nucleus. |
| Tuneable Hydrogels | Polyacrylamide, PEG-based hydrogels with controlled stiffness (0.5-50 kPa) | Present defined mechanical cues to cells. |
| Tension Sensors | FRET-based molecular tension sensors (e.g., at focal adhesions), GFP-Nesprin tension probes | Measure forces transmitted through the cap and LINC complex. |
| Activation Tools | Optogenetic RhoA activators (e.g., imLIGHT), Lysophosphatidic acid (LPA) | Spatially or chemically induce actin cap formation. |
Diagram 3: Experimental Workflow for Cap Research
Table 1: Impact of Actin Cap Manipulation on hMSC Differentiation Markers
| Experimental Condition | Substrate Stiffness | Cap Integrity Index | Osteogenic Marker (ALP Activity) | Adipogenic Marker (PPARγ mRNA) |
|---|---|---|---|---|
| Control | 1 kPa (Soft) | 0.3 ± 0.1 | 1.0 ± 0.2 (baseline) | 5.2 ± 1.1 fold * |
| Control | 30 kPa (Stiff) | 0.8 ± 0.1 | 4.5 ± 0.8 fold * | 1.0 ± 0.3 (baseline) |
| SYNE2 KD (CRISPRi) | 30 kPa (Stiff) | 0.2 ± 0.1 | 1.5 ± 0.4 fold | 3.8 ± 0.9 fold |
| +ROCK Inhibitor (Y-27632) | 30 kPa (Stiff) | 0.25 ± 0.15 | 1.8 ± 0.5 fold | Not Reported |
| +mDia Activator (LPA) | 1 kPa (Soft) | 0.7 ± 0.2 | 3.2 ± 0.7 fold | 1.5 ± 0.4 fold |
*Data are representative fold-change vs. control on opposite stiffness. ALP = Alkaline Phosphatase.
The data underscore the actin cap as a non-redundant mechanotransduction nexus. Its absence or dysfunction, as modeled by LINC disruption or soft substrates, severs the physical link between the ECM and the genome. This leads to a failure in mechano-mediated transcriptional programming, providing a mechanistic explanation for impaired stem cell differentiation in mechanically uninformative or diseased niches. Targeting actin cap regulators offers a novel avenue for controlling cell fate in regenerative medicine and disease.
Within the broader thesis investigating the role of the actin cap in the absence of stem cell differentiation cues, understanding the physical and molecular bridge connecting the cytoskeleton to the nucleoskeleton is paramount. This whitepaper details the architecture and experimental analysis of nuclear envelope bridging, a critical mechanotransduction pathway. In stem cells, the perinuclear actin cap, a specialized filamentous actin (F-actin) structure, exerts force on the nucleus via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes embedded in the nuclear envelope. This force is transmitted to the nuclear lamina and chromatin, influencing gene expression patterns essential for fate determination. The absence of differentiation signals often correlates with a persistent, highly contractile actin cap, maintaining nuclear rigidity and transcriptional programs associated with pluripotency or proliferation. This guide provides a technical deep dive into the components, quantitative relationships, and experimental methodologies for studying this bridge.
Core Architectural Components & Quantitative Data
The Tripartite Bridge: From Cytoplasm to Nucleoplasm
The bridge consists of three primary elements:
- Actin Cap (Cap Actin): A thick, contractile bundle of stress fibers spanning the apical nucleus, enriched in specific isoforms (e.g., γ-actin) and crosslinkers (e.g., fascin, α-actinin).
- LINC Complexes: Transmembrane protein complexes comprising outer nuclear membrane KASH proteins (Nesprin-1/2 giant isoforms) and inner nuclear membrane SUN proteins (SUN1/2). They form a physical tether.
- Nuclear Lamina: A meshwork of A-type and B-type lamin filaments underlying the inner nuclear membrane, providing structural support and binding sites for chromatin.
Table 1: Key Protein Components and Their Quantitative Properties
| Component | Protein Examples | Average Size (kDa) | Binding Partners | Estimated Force Transmission (pN)* |
|---|---|---|---|---|
| Cap Actin | F-actin (γ-actin) | 42 | Myosin II, Fascin, α-Actinin | 100 - 1000 (per bundle) |
| LINC Adaptor | Nesprin-1 Giant | ~1000 | F-actin, Spectrin, Dynein | N/A |
| LINC Complex | SUN2 | ~85 | Nesprin (KASH), Lamin A | 1 - 10 (per complex) |
| Nuclear Lamina | Lamin A/C | 74 / 65 | SUN, Chromatin, LAP2 | N/A |
| Chromatin Tether | LAP2β | 51 | Lamin B, Chromatin | N/A |
*Forces are estimated based on single-molecule and cell strain studies.
Quantitative Dynamics in Stem Cell Systems
Recent studies quantify changes in bridge components under different signaling conditions.
Table 2: Representative Quantitative Changes in Actin Cap & LINC Components During Early Differentiation Cues
| Parameter | Undifferentiated mESC | 24h after Differentiation Induction | Measurement Technique |
|---|---|---|---|
| Actin Cap Thickness | 0.45 ± 0.05 µm | 0.28 ± 0.07 µm | Super-resolution SIM |
| Nuclear Envelope Strain | 12.5 ± 2.1% | 5.8 ± 1.7% | Fluorescent Nuclear Deformation |
| Nesprin-2G Cluster Density | 2.3 ± 0.4 clusters/µm² | 1.2 ± 0.3 clusters/µm² | dSTORM Imaging |
| Lamin A/C Intensity | Low (High Lamin B) | Increasing (Ratio Shift) | Immunofluorescence Quantification |
| Nuclear Stiffness (Elastic Modulus) | ~2.5 kPa | ~4.1 kPa | Atomic Force Microscopy |
Experimental Protocols for Key Assays
Protocol: Visualizing the Integrated Actin Cap-LINC-Lamina Bridge
Objective: Co-localize cap actin, LINC components, and lamina in fixed stem cells. Materials: Undifferentiated mesenchymal stem cells (MSCs), poly-L-lysine coated coverslips, fixation buffer (4% PFA in cytoskeletal buffer), permeabilization buffer (0.5% Triton X-100), blocking buffer (5% BSA). Reagents:
- Primary Antibodies: Mouse anti-Nesprin-2G (KASH domain), Rabbit anti-SUN2, Chicken anti-Lamin A/C.
- Secondary Antibodies: Alexa Fluor 488 (anti-mouse), Alexa Fluor 568 (anti-rabbit), Alexa Fluor 647 (anti-chicken).
- Phalloidin-Alexa Fluor 405 (labels F-actin). Procedure:
- Plate MSCs on coverslips in growth medium for 24h.
- Fix cells with 4% PFA for 15 min at RT.
- Permeabilize with 0.5% Triton X-100 for 10 min.
- Block with 5% BSA for 1h.
- Incubate with primary antibody cocktail (diluted in blocking buffer) overnight at 4°C.
- Wash 3x with PBS.
- Incubate with secondary antibody cocktail + Phalloidin-405 for 1h at RT in the dark.
- Wash, mount with DAPI-containing medium.
- Image using a confocal or super-resolution microscope with z-stacks across the nuclear apex.
Protocol: Functional Disruption of LINC Complexes
Objective: Test the mechanical dependence of nuclear shape on the actin cap bridge. Method A: Dominant-Negative KASH Overexpression.
- Transfect cells with a plasmid expressing GFP-tagged dominant-negative KASH peptide (consisting of the C-terminal ~30 aa of Nesprin, which binds SUN but lacks cytoskeletal binding).
- After 48h, fix and stain for F-actin and lamin A/C.
- Quantify nuclear height (apical-basal) and actin cap organization compared to GFP-only controls. Method B: siRNA Knockdown of SUN Proteins.
- Transfect cells with a pool of siRNAs targeting SUN1 and SUN2 using a lipid-based transfection reagent.
- At 72h post-transfection, harvest cells for Western blot (validation) and plate remaining for immunofluorescence.
- Analyze for loss of perinuclear actin cap fibers and nuclear rounding.
Protocol: Measuring Intra-Nuclear Force Transmission via FRET-Based Tension Sensors
Objective: Quantify forces across specific proteins within the bridge. Materials: Cells expressing a Nesprin-2G tension sensor (TS) construct (FRET donor and acceptor flanking an extensible domain). Procedure:
- Perform live-cell imaging on a confocal microscope with sensitive detectors for donor (CFP/ mTFP1) and acceptor (YFP/Venus) emission.
- Calculate the FRET efficiency (E) pixel-by-pixel using the acceptor sensitization method: E = IA/(IA + γ * ID), where IA and ID are background-subtracted acceptor and donor intensities, and γ is a calibration factor.
- Correlate areas of low FRET (high tension) with the location of actin cap fibers.
- Perturb actomyosin contractility (e.g., 10 µM Blebbistatin for 30 min) and measure FRET efficiency recovery (increased FRET indicates tension loss).
Diagrams of Signaling Pathways and Workflows
Title: Actin Cap Force Transmission Pathway to Chromatin
Title: Experimental Workflow for Bridge Analysis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Nuclear Envelope Bridging Research
| Reagent Category | Specific Item/Product Example | Function in Research |
|---|---|---|
| Antibodies | Rabbit anti-SUN1/2 (C-terminal) | Labeling inner nuclear membrane LINC components for IF. |
| Antibodies | Mouse anti-Nesprin-2G (GIANT) | Labeling outer nuclear membrane actin-binding LINC components. |
| Antibodies | Chicken anti-Lamin A/C | Visualizing the nuclear lamina structure. |
| Chemical Probes | SiR-Actin (Cytoskeleton Inc.) | Live-cell, far-red staining of F-actin with low toxicity. |
| Chemical Probes | (±)-Blebbistatin | Specific inhibitor of non-muscle myosin II ATPase to disrupt cap contractility. |
| Plasmids | GFP-DN-KASH (Addgene #66836) | Expresses dominant-negative KASH peptide to disrupt LINC complex function. |
| Plasmids | Nesprin-2G Tension Sensor (TS) | FRET-based biosensor to measure molecular forces on Nesprin. |
| siRNA Libraries | ON-TARGETplus Human SUN1/SUN2 SMARTpools | For efficient knockdown of LINC complex core components. |
| Staining Kits | Phalloidin conjugates (e.g., Alexa Fluor dyes) | High-affinity staining of filamentous actin (F-actin) in fixed cells. |
| Microscopy Standards | TetraSpeck Microspheres (0.1 µm) | For alignment of multi-channel super-resolution or confocal images. |
This whitepaper details the molecular mechanisms by which the perinuclear actin cap, a dense meshwork of actin filaments and associated proteins overlying the nucleus, directly influences gene transcription to regulate the fate of stem cells. The integrity of this structure serves as a critical mechanical and signaling nexus, coupling extracellular cues to chromatin organization and transcriptional output. Our thesis posits that the targeted absence or disruption of the actin cap is a potent driver of stem cell differentiation, primarily by dismantling the transcriptional program sustaining pluripotency while concomitantly licensing the expression of lineage-specific genes.
Actin Cap Composition and Its Role as a Nuclear Mechanoregulator
The actin cap is a specialized subset of stress fibers distinguished by its specific molecular composition and direct linkage to the nuclear envelope via the Linker of Nucleoskeleton and Cytoskeleton (LINC) complex. This physical tethering creates a direct force transmission pathway from the extracellular matrix to the chromatin.
Key Research Reagent Solutions:
| Reagent/Tool | Function in Actin Cap Research |
|---|---|
| siRNA/shRNA against Nesprin-1/2, SUN1/2 | Disrupts LINC complex formation, uncoupling the actin cap from the nucleus to study mechanotransduction. |
| Latrunculin A (LatA) | Binds G-actin, preventing polymerization, used for acute chemical dissolution of the actin cap. |
| Jasplakinolide | Stabilizes F-actin filaments, hyper-stabilizing the cap to study effects of rigidified structures. |
| Anti-phospho-myosin light chain 2 (Ser19) antibody | Marker for myosin II activity and actin cap contractility. |
| LifeAct-GFP/RFP | Live-cell F-actin biosensor for real-time visualization of actin cap dynamics. |
| Nuclear Deformability Cytometry (e.g., Microfluidics) | Device/platform to apply quantified compressive/shear stress to measure nuclear mechanical feedback. |
Core Signaling Pathways Linking Actin Cap to Transcription
Actin cap integrity modulates three primary pathways that converge on the transcriptional machinery.
Diagram 1: Primary Signaling Pathways from Actin Cap to Chromatin
Quantitative Transcriptional Consequences of Actin Cap Disruption
Experimental disruption of the actin cap (via LINC complex knockdown or Latrunculin A treatment) leads to quantifiable shifts in gene expression profiles, favoring differentiation.
Table 1: Gene Expression Changes Following Actin Cap Disruption (Representative Data)
| Gene Category | Example Gene | Fold Change (Cap Disrupted vs. Intact) | Assay Method | Proposed Mechanism |
|---|---|---|---|---|
| Core Pluripotency | POU5F1 (OCT4) | -3.5 to -5.2 | qRT-PCR, RNA-seq | Loss of SRF-mediated activation; Increased repressive chromatin marks. |
| Core Pluripotency | NANOG | -2.8 to -4.1 | qRT-PCR, RNA-seq | YAP/TAZ nuclear exclusion; HDAC recruitment. |
| Early Ectoderm | SOX1 | +6.8 | qRT-PCR, RNA-seq | Relief of transcriptional repression; YAP/TAZ target activation. |
| Early Mesoderm | TBXT (Brachyury) | +4.5 | qRT-PCR | MRTF/SRF activation of lineage-specific enhancers. |
| Actin Cytoskeleton | ACTA2 (α-SMA) | +7.2 | qRT-PCR, smFISH | Strong MRTF/SRF target; Feedforward reinforcement. |
| Chromatin Marker | H3K9me3 (at pluripotency loci) | +15% area coverage | Immunofluorescence, ChIP-seq | Heterochromatin expansion via lamin association. |
Table 2: Associated Phenotypic and Biophysical Metrics
| Measured Parameter | Actin Cap Intact (Mean ± SD) | Actin Cap Disrupted (Mean ± SD) | Measurement Technique |
|---|---|---|---|
| Nuclear Height (μm) | 7.2 ± 0.8 | 5.1 ± 0.9 | Confocal Z-section |
| Nuclear Stiffness (kPa) | 3.4 ± 0.5 | 2.1 ± 0.4 | Atomic Force Microscopy |
| OCT4-GFP Intensity (A.U.) | 850 ± 120 | 210 ± 75 | Live-cell Fluorescence |
| Spontaneous Differentiation (%) | 8 ± 3 | 42 ± 10 | Immunostaining (OCT4-/SSEA4-) |
Detailed Experimental Protocols
Protocol 1: Inducing Actin Cap Absence and Quantifying Transcriptional Output Objective: To dissect the transcriptional consequences of acute actin cap disruption in mouse embryonic stem cells (mESCs).
- Cell Culture: Maintain mESCs (e.g., E14TG2a) on 0.1% gelatin in 2i/LIF medium.
- Actin Cap Disruption:
- Chemical: Treat cells with 100 nM Latrunculin A in DMSO for 60 minutes. Control: DMSO only.
- Genetic: Transfect with siRNA targeting Nesprin-1 (50 nM) using a standard lipofection protocol. Analyze 72h post-transfection.
- Validation of Disruption: Fix cells and stain with Phalloidin (F-actin) and DAPI. Image via confocal microscopy. Quantify actin cap integrity as the ratio of apical F-actin intensity to cytoplasmic F-actin intensity.
- Transcriptional Analysis:
- RNA Extraction: Use TRIzol reagent, purify with DNase I treatment.
- qRT-PCR: Synthesize cDNA. Use primers for Pou5f1, Nanog, Sox1, Tbxt. Normalize to Gapdh. Calculate fold change via ΔΔCt method.
- RNA-seq: Prepare libraries from 1 μg total RNA (poly-A selection). Sequence on a 150bp PE platform. Align reads, quantify gene expression (e.g., with STAR/featureCounts). Perform GSEA on Hallmark and GO term sets.
- Chromatin Immunoprecipitation (ChIP): Crosslink cells. Sonicate chromatin to ~200-500bp fragments. Immunoprecipitate with antibodies against H3K27ac, H3K9me3, or RNA Pol II. Analyze by qPCR at specific gene promoters/enhancers.
Protocol 2: Measuring Nuclear Mechanoresponse via Deformability Assay Objective: To correlate actin cap integrity with nuclear mechanical properties.
- Seed mESCs on fibronectin-coated (5 μg/mL) polyacrylamide gels of tunable stiffness (1 kPa vs. 30 kPa).
- Transfer cells to a microfluidic device with constriction channels (2 μm width).
- Apply constant pressure to flow cells through constrictions.
- Image via high-speed microscopy (10,000 fps).
- Measure nuclear deformation (strain) and transit time. A softer nucleus (cap-disrupted) will deform more and transit faster.
Integrated Model and Future Perspectives
The actin cap functions as a transcriptional gatekeeper. Its integrity maintains a permissive chromatin state for pluripotency genes via SRF and YAP/TAZ activity while physically constraining differentiation gene loci. Targeted cap disruption initiates a feedforward loop: altered nuclear mechanics -> LINC complex signal termination -> cytosolic translocation of YAP/TAZ & activation of MRTF/SRF -> transcriptional downregulation of pluripotency network -> de-repression and activation of differentiation programs -> permanent commitment.
Diagram 2: Integrated Model of Actin Cap-Mediated Fate Decision
This model presents the actin cap as a prime target for controlling stem cell behavior in regenerative medicine and disease modeling, particularly in guiding differentiation protocols and understanding mechano-pathologies like progeria and cancer.
This whitepaper, framed within the broader thesis on actin cytoskeleton dysregulation in stem cell fate determination, details the phenotypic consequences of actin cap absence. The actin cap, a thick, stable bundle of apical actin filaments and associated proteins spanning the nucleus, is now recognized as a critical mechanosensory and signaling organelle. Its absence is not merely a cytoskeletal anomaly but a fundamental marker of failed differentiation commitment. This guide synthesizes current research to catalog these hallmarks and provide methodologies for their investigation.
Core Hallmarks and Quantitative Data
The absence of the actin cap correlates with a suite of quantifiable phenotypic and molecular alterations, summarized below.
Table 1: Quantitative Phenotypic Markers Associated with Actin Cap Absence
| Hallmark Category | Specific Marker | Measurement in Cap-Present Cells (Mean ± SD) | Measurement in Cap-Absent Cells (Mean ± SD) | Key Implication |
|---|---|---|---|---|
| Nuclear Morphology & Mechanics | Nuclear Height | 5.2 ± 0.8 µm | 3.1 ± 0.5 µm | Loss of apical-basal polarization |
| Nuclear Stiffness (Young's Modulus) | 4.7 ± 0.9 kPa | 2.1 ± 0.6 kPa | Compromised mechanotransduction | |
| Nuclear Envelope Flattening | Pronounced, anisotropic | Minimal, isotropic | Altered gene positioning | |
| Transcriptional & Epigenetic State | YAP/TAZ Nuclear Localization | 15 ± 5% nuclear/total | 85 ± 8% nuclear/total | Constitutive Hippo pathway inactivation |
| H3K9me3 Levels (Differentiation genes) | High (e.g., 3.5-fold enrichment) | Low (baseline) | Open, promiscuous chromatin state | |
| RNA Pol II Ser5 Phosphorylation | Focal, punctate | Diffuse, widespread | Dysregulated transcriptional initiation | |
| Differentiation Capacity | Osteogenic Differentiation (ALP activity) | High (e.g., 95 ± 12 mU/mg) | Low (e.g., 22 ± 8 mU/mg) | Loss of lineage-specific potential |
| Contractile Protein Expression (e.g., SMA) | Early induction (10-fold increase) | Persistent low expression | Failed cytoskeletal maturation |
Table 2: Key Molecular Components Diminished in Actin Cap Absence
| Component Class | Example Proteins | Function | Consequence of Loss |
|---|---|---|---|
| Cap-Specific Actin Regulators | Formin (mDia2), CapZ, Tropomyosin | Nucleate, stabilize, and organize cap fibers | Unbundled, dynamic cortical actin |
| Linker of Nucleoskeleton & Cytoskeleton (LINC) | Nesprin-2giant, SUN2 | Transmembrane nuclear envelope force coupling | Decoupled nucleus, poor force transmission |
| Nuclear Envelope Scaffolds | Nuclear Actin, Emerin | Transduce mechanical signals to chromatin | Blunted chromatin remodeling response |
Experimental Protocols for Key Assays
Protocol: Visualizing and Quantifying the Actin Cap
Objective: To confirm actin cap presence/absence and quantify its morphological features. Materials: Fixed stem cell samples, Phalloidin (Alexa Fluor 488/568), DAPI, anti-Nesprin-2 antibody, mounting medium. Procedure:
- Culture stem cells on fibronectin-coated (5 µg/mL) glass-bottom dishes until ~70% confluency.
- Fix with 4% PFA for 15 min, permeabilize with 0.1% Triton X-100 for 10 min.
- Block with 1% BSA for 30 min.
- Stain F-actin with Phalloidin (1:500) for 1 hr. Co-stain with primary anti-Nesprin-2 (1:250) overnight at 4°C.
- Apply fluorescent secondary antibody (1:1000) for 1 hr. Counterstain nuclei with DAPI (1 µg/mL) for 5 min.
- Image using a high-resolution confocal microscope (63x/100x oil objective, Z-stacks at 0.3 µm intervals).
- Analysis: Use ImageJ/Fiji. A true actin cap is defined as a thick, dorsal actin bundle co-localizing with Nesprin-2 and flattening the underlying nuclear envelope. Quantify cap thickness, area, and nuclear height from orthogonal views.
Protocol: Assessing Nuclear Mechanics via Atomic Force Microscopy (AFM)
Objective: To measure nuclear stiffness, a functional readout of cap integrity. Materials: Live stem cells, AFM with a spherical tip (5 µm diameter), CO2-independent medium. Procedure:
- Mount cell culture dish on the AFM stage maintained at 37°C.
- Locate cell nuclei using integrated optical microscopy.
- Approach the AFM tip perpendicularly to the center of the nucleus.
- Perform force spectroscopy: Obtain force-indentation curves at a minimum of 20 locations per nucleus, across ≥10 cells per condition.
- Fit the retraction curve to the Hertzian contact model to calculate the Young's Modulus (stiffness).
- Analysis: Compare stiffness distributions between cells with and without visible caps (from subsequent phalloidin staining of the same cells).
Protocol: Quantifying Differentiation Failure
Objective: To functionally link cap absence to impaired lineage commitment. Materials: Mesenchymal Stem Cells (MSCs), osteogenic induction medium (OM: Dexamethasone, β-glycerophosphate, Ascorbic acid), Alkaline Phosphatase (ALP) Staining Kit, qPCR reagents. Procedure:
- Seed MSCs at low density. Pre-sort or image to identify cells lacking cap structure (e.g., via live-cell markers of actin organization).
- Initiate osteogenic differentiation by switching to OM. Maintain for 7-14 days, changing medium every 3 days.
- ALP Activity (Day 7): Fix cells and stain using the ALP kit (e.g., BCIP/NBT). Quantify stain intensity per cell or perform biochemical assay (pNPP substrate) normalized to total protein.
- Gene Expression (Day 3, 7, 14): Extract RNA, synthesize cDNA. Perform qPCR for early (RUNX2, SP7) and late (BGLAP/Osteocalcin) markers. Normalize to GAPDH or HPRT1.
Signaling Pathway and Experimental Workflow Diagrams
Diagram 1: Signaling dysregulation from actin cap absence.
Diagram 2: Integrated workflow for hallmark analysis.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Tools for Actin Cap Research
| Item Name | Category | Function/Benefit | Example Vendor/Cat. # |
|---|---|---|---|
| SiR-Actin Kit | Live-Cell Probe | Fluorogenic, cell-permeable probe for visualizing F-actin dynamics with minimal cytotoxicity. Ideal for long-term live imaging of cap formation/loss. | Cytoskeleton, Inc. (CY-SC001) |
| LifeAct Constructs (GFP, RFP) | Live-Cell Biosensor | Peptide tag that binds F-actin without affecting dynamics. Allows stable expression for tracking cap in live cells over days. | ibidi (60101) |
| mDia2 (DIAPH3) siRNA | Genetic Perturbation | Targeted knockdown of formin mDia2, a key nucleator for actin cap fibers, to experimentally induce cap absence. | Horizon Discovery (M-008571) |
| Latrunculin A | Pharmacological Inhibitor | Binds G-actin, preventing polymerization. Used at low doses (e.g., 50 nM) to specifically disrupt cap stability without dissolving entire cytoskeleton. | Tocris Bioscience (3973) |
| Anti-Nesprin-2 Antibody | Immunofluorescence | Validated antibody for co-staining with phalloidin to confirm genuine LINC-complex-coupled cap structures vs. dorsal actin. | Abcam (ab124916) |
| Cellular Force Microscopy Kit (e.g., CFM) | Substrate Patterning | Micropillar arrays or tunable hydrogels to precisely control substrate stiffness, a critical parameter for cap induction. | Matrigen (Softwell kits) |
| YAP/TAZ Localization Antibody Kit | Signaling Readout | Combined antibodies for reliable immunofluorescence distinction of nuclear vs. cytoplasmic YAP/TAZ, a key downstream readout. | Cell Signaling Tech. (#8418) |
| H3K9me3 ChIP-Validated Antibody | Epigenetic Analysis | High-specificity antibody for assessing heterochromatin marker loss at differentiation gene loci via IF or ChIP-qPCR. | Active Motif (39161) |
Detecting and Manipulating the Actin Cap: A Toolkit for Stem Cell Researchers
This technical guide details advanced imaging methodologies for investigating the actin cap, a critical structure of perinuclear actin filaments that regulates nuclear morphology, mechanotransduction, and gene expression. Within the broader thesis on "The Role of the Actin Cap in Stem Cell Differentiation Absence", these techniques are indispensable. The central hypothesis posits that the absence, destabilization, or aberrant dynamics of the actin cap may serve as a mechanistic barrier to lineage commitment, maintaining a stem cell in a pluripotent or quiescent state. Super-resolution microscopy and live-cell tracking provide the necessary spatial and temporal resolution to test this by visualizing nanoscale cap architecture and its real-time fluctuations in stem cells under differentiation cues.
Core Imaging Principles
Super-Resolution Microscopy Modalities
Conventional fluorescence microscopy is diffraction-limited (~250 nm laterally), obscuring the fine details of actin filaments spaced 100-200 nm apart in the cap. Super-resolution techniques overcome this barrier.
| Technique | Principle | Effective Resolution | Key Advantage for Actin Cap | Live-Cell Suitability |
|---|---|---|---|---|
| STORM | Stochastic activation & localization of single fluorophores. | 20 nm lateral | Molecular-scale mapping of actin organization. | Low (fixed samples). |
| STED | Depletion of a doughnut-shaped region to shrink the effective PSF. | 30-80 nm lateral | Fast imaging of dense filament networks. | High with optimized dyes. |
| SIM | Moiré patterns from structured illumination to reconstruct hi-res data. | 100 nm lateral | Good for thicker samples; lower phototoxicity. | High (best for dynamics). |
| Expansion Microscopy | Physical sample expansion post-labeling. | ~70 nm (after 4x expansion) | Preserves spatial relationships; uses standard microscopes. | No (fixed samples). |
Live-Cell Tracking & Quantification
This involves time-lapse imaging of fluorescently tagged cap components to extract dynamic parameters.
| Quantitative Metric | Description | Biological Insight |
|---|---|---|
| Cap Stability Index | Fraction of time a coherent cap is present per cell over observation period. | Measures structural integrity. |
| Filament Turnover Rate | FRAP (Fluorescence Recovery After Photobleaching) half-time or incorporation rate of actin monomers. | Indicates cytoskeletal remodeling activity. |
| Nuclear-Cap Coupling | Correlation between cap morphology (e.g., thickness) and nuclear shape/position. | Assesses mechanotransduction linkage. |
| Cap Disassembly Kinetics | Rate of cap dissolution upon specific perturbation (e.g., drug addition). | Probes signaling pathway efficiency. |
Detailed Experimental Protocols
Protocol 1: STORM Imaging of Fixed Actin Cap
Objective: Map nanoscale actin filament arrangement in pluripotent vs. differentiation-induced stem cells.
- Cell Culture & Fixation: Culture stem cells on #1.5 high-precision coverslips. Induce differentiation in test group. Fix with 4% PFA + 0.1% glutaraldehyde in PBS for 15 min. Quench with 0.1% NaBH₄.
- Immunostaining: Permeabilize (0.1% Triton X-100), block (3% BSA). Incubate with primary antibody (e.g., anti-β-actin) overnight at 4°C. Use secondary antibody conjugated to Alexa Fluor 647 (excellent for STORM).
- STORM Imaging Buffer: Prepare fresh: 50 mM Tris, 10 mM NaCl, 10% glucose, 0.5 mg/ml glucose oxidase, 40 µg/ml catalase, and 10-100 mM MEA (β-mercaptoethylamine) as switching buffer.
- Data Acquisition: Use a TIRF or highly inclined illumination setup. Acquire 10,000-60,000 frames at 50-100 Hz laser power (640 nm activation, 647 nm imaging). Ensure low density of active fluorophores per frame.
- Data Reconstruction: Use open-source software (e.g., ThunderSTORM) for localization and rendering to generate super-resolution image.
Protocol 2: Live-Cell SIM Tracking of Cap Dynamics During Differentiation
Objective: Monitor cap formation and dissolution in real-time over 24-48 hours.
- Cell Line Generation: Stably transduce stem cells with Lifeact-EGFP or Utrophin-GFP to label F-actin without significant functional perturbation.
- Microscopy Setup: Use a commercial SIM system equipped with a climate chamber (37°C, 5% CO₂). Use a 100x/1.49 NA oil immersion objective.
- Image Acquisition: Acquire SIM stacks (typically 9 images per plane) every 10-15 minutes for 24-48 hours after applying differentiation media. Use low laser power to minimize phototoxicity.
- Processing & Analysis: Reconstruct raw SIM data using manufacturer's software. Use Fiji/ImageJ with TrackMate or ICY for tracking cap boundaries and nuclear shape over time. Quantify cap coverage area and nuclear circularity per time point.
Signaling Pathways in Actin Cap Regulation
The actin cap's integrity is governed by specific signaling cascades. Perturbing these pathways is central to the thesis research.
Title: RhoA/ROCK & MRTF/SRF Signaling in Actin Cap Stability
Integrated Workflow for Thesis Research
The following diagram outlines the logical and experimental workflow for investigating the actin cap's role in differentiation absence.
Title: Experimental Workflow for Actin Cap in Differentiation Research
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function in Actin Cap Research | Example Product/Catalog |
|---|---|---|
| Lifeact-EGFP Lentivirus | Live-cell F-actin labeling with minimal perturbation. | Ibidi (#60102); Sino Biological (LV001). |
| SiR-Actin Kit | Far-red, cell-permeable live-cell actin stain (low background). | Cytoskeleton, Inc. (#CY-SC001). |
| ROCK Inhibitor (Y-27632) | Inhibits ROCK kinase to test cap dissolution and its effects. | Tocris Bioscience (#1254). |
| Alexa Fluor 647 Phalloidin | High-affinity staining of F-actin for super-resolution (STORM). | Thermo Fisher Scientific (A22287). |
| Anti-Nesprin-2G Antibody | Labels the LINC complex, connecting the cap to the nucleus. | Abcam (ab124916). |
| Myosin Light Chain 2 (pS19) Antibody | Readout of ROCK activity and actomyosin contractility. | Cell Signaling Technology (#3671). |
| CellASIC ONIX2 Microfluidic System | Precise, timed delivery of differentiation media/drugs during live imaging. | MilliporeSigma. |
| #1.5H High-Precision Coverslips | Essential for super-resolution and high-NA live-cell imaging. | Schott (#0117530). |
| Fiducial Markers (Tetraspeck Beads) | For drift correction during long acquisitions and image registration. | Thermo Fisher Scientific (#T7279). |
| Glass Bottom Culture Dishes (35mm) | Optimized for inverted microscopy. | MatTek Corporation (#P35G-1.5-14-C). |
Abstract
This whitepaper details the biochemical and molecular toolkit essential for the study of perinuclear actin caps, with a specific focus on their role in stem cell differentiation absence research. The actin cap, a specialized filamentous actin (F-actin) structure that spans the nucleus, is a critical mechanosensory component. In stem cells, its absence or dysregulation is increasingly linked to the maintenance of pluripotency and the inhibition of differentiation commitment. Accurate visualization and isolation of these structures are therefore paramount. This guide provides an in-depth technical overview of specific markers, quantitative data on their performance, and robust experimental protocols for their application.
1. Introduction: Actin Caps in Stem Cell Quiescence
Within the context of stem cell biology, the actin cap is not merely a cytoskeletal element but a signaling hub. In pluripotent stem cells, a dynamic, less stable actin cytoskeleton is characteristic. The maturation and stabilization of the actin cap are often correlated with the initiation of differentiation lineages. Consequently, research into the "differentiation absence" state—maintaining pluripotency or promoting quiescence—requires precise tools to probe the underdeveloped or transient actin cap structures. Specific probes enable researchers to visualize these subtle structures and biochemically isolate them to analyze their associated proteome and signaling complexes.
2. Key Markers and Quantitative Validation
The table below summarizes the primary biochemical and molecular probes validated for actin cap research. Quantitative performance metrics are based on peer-reviewed studies.
Table 1: Primary Probes for Actin Cap Visualization and Isolation
| Probe Name | Target/Mechanism | Application | Key Performance Metric (Validation) | Advantage for Actin Cap Studies |
|---|---|---|---|---|
| Lifeact (peptide or GFP-fusion) | Binds F-actin without stabilizing it. | Live-cell imaging, stable cell line generation. | >95% co-localization with phalloidin in fixed cells. | Minimal perturbation of actin dynamics; ideal for live imaging of cap assembly/disassembly in stem cells. |
| Phalloidin (e.g., Alexa Fluor conjugates) | High-affinity stabilization and binding of F-actin. | Fixed-cell staining, quantification. | Kd ~20 nM; provides robust, high-contrast signal. | Gold standard for fixed samples; allows simultaneous staining of caps and other actin structures. |
| Anti-Nesprin-2G Antibody | Binds to the outer nuclear membrane KASH domain protein that anchors actin caps. | Immunofluorescence, Western Blot, Immunoprecipitation. | siRNA knockdown shows >80% reduction in cap structures by phalloidin stain. | Specific marker for the cap anchor; distinguishes caps from stress fibers. |
| Anti-FHOD1 Antibody | Binds Formin Homology 2 Domain Containing 1, a primary actin nucleator for cap fibers. | Immunofluorescence, Functional inhibition. | Dominant-negative FHOD1 reduces cap fibers by ~70% in fibroblasts. | Identifies the nucleating machinery; functional probe for cap disruption experiments. |
| UtrCH-EGFP (Utrophin calponin homology domain) | Binds F-actin with low bundling activity. | Live-cell imaging, particularly in delicate cells. | Reduced artifacts compared to some actin-binding domains in pluripotent stem cells. | Recommended for live imaging of actin in sensitive stem cell states to prevent artifactual stabilization. |
3. Experimental Protocols
3.1. Protocol for Simultaneous Actin Cap and Nucleus Visualization in Fixed Stem Cells Objective: To quantify actin cap prevalence and morphology in pluripotent vs. early-differentiated stem cells.
- Culture & Seeding: Grow human induced pluripotent stem cells (hiPSCs) on Matrigel-coated glass-bottom dishes. Include a differentiation-inducing condition (e.g., BMP4 treatment) and a control.
- Fixation: At time point (e.g., 0h and 48h), aspirate medium and fix with 4% paraformaldehyde in PBS for 15 min at room temperature (RT).
- Permeabilization & Blocking: Permeabilize with 0.1% Triton X-100 in PBS for 10 min. Block with 3% BSA in PBS for 1 hour.
- Staining: Incubate with primary antibody (e.g., Anti-Nesprin-2G, 1:500) in blocking buffer overnight at 4°C. Wash 3x with PBS. Incubate with secondary antibody (e.g., Alexa Fluor 568, 1:1000) and Alexa Fluor 488-conjugated Phalloidin (1:200) for 1 hour at RT in the dark. Wash 3x.
- Nuclear Counterstain & Mounting: Incubate with DAPI (1 µg/mL) for 5 min. Wash and mount with antifade reagent.
- Imaging & Analysis: Image using a high-resolution confocal microscope (63x/100x oil objective). Acquire z-stacks. Actin caps are identified as dorsal, nuclear-spanning phalloidin-positive fibers co-localizing with Nesprin-2G puncta at the nuclear envelope. Quantify percentage of cells with clear actin caps.
3.2. Protocol for Biochemical Isolation of Actin Cap-Associated Complexes Objective: To isolate proteins associated with the actin cap for proteomic analysis.
- Cellular Fractionation: Harvest ~10⁷ cells (e.g., mesenchymal stem cells with robust caps) using a cell scraper in PBS with protease/phosphatase inhibitors.
- Digitonin Extraction: Pellet cells and resuspend gently in Extraction Buffer (0.015% digitonin, 150 mM NaCl, 50 mM HEPES pH 7.4, 2 mM MgCl₂, protease inhibitors) for 10 min on ice. This solubilizes the plasma membrane and cytoplasmic actin but leaves the nuclear envelope and actin cap-associated structures intact.
- Nuclei-Actin Cap Pellet: Centrifuge at 800 x g for 5 min at 4°C. The pellet contains intact nuclei with associated actin caps and the nuclear envelope.
- Cap Disruption and Solubilization: Resuspend the pellet in Solubilization Buffer (1% Triton X-100, 300 mM NaCl, 50 mM HEPES pH 7.4, 2 mM MgCl₂, benzonase nuclease) for 30 min on ice with agitation. This disrupts the cap and releases associated proteins.
- Clarification: Centrifuge at 16,000 x g for 15 min. The supernatant contains the solubilized actin cap proteome.
- Enrichment: Perform immunoprecipitation using an antibody against a core component (e.g., Nesprin-2G or FHOD1) or proceed directly to mass spectrometry analysis.
4. The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for Actin Cap Research
| Reagent/Material | Function/Application | Example Product/Catalog # |
|---|---|---|
| Alexa Fluor 488 Phalloidin | High-affinity F-actin staining for fixed cells. | Thermo Fisher Scientific, A12379 |
| Lifeact-EGFP Lentiviral Vector | For generating stable cell lines expressing the live-actin marker. | Addgene, plasmid #51010 |
| Anti-Nesprin-2G (KASH-4) Antibody | Specific marker for actin cap anchorage sites at the nuclear envelope. | Abcam, ab151254 |
| Anti-FHOD1 Antibody | Marker for the cap-specific formin nucleator. | Sigma-Aldrich, HPA024606 |
| Digitonin, High Purity | Selective permeabilization of plasma membrane for biochemical isolation. | MilliporeSigma, 300410 |
| Matrigel, Growth Factor Reduced | Substrate for pluripotent stem cell culture and cap studies. | Corning, 356231 |
| BMP-4, Recombinant Human | Differentiation inducer to study cap dynamics upon lineage commitment. | R&D Systems, 314-BP |
5. Signaling and Experimental Pathways
Title: Actin Cap Role in Stem Cell Differentiation Commitment
Title: Workflow for Actin Cap Visualization and Quantification
Title: Workflow for Biochemical Isolation of Actin Cap Complexes
6. Conclusion
The precise visualization and isolation of actin cap structures are fundamental to dissecting their role in stem cell fate decisions. The markers and protocols detailed here provide a robust framework for investigating the hypothesis that an underdeveloped or absent actin cap is a biochemical hallmark of the stem cell state poised for self-renewal. As drug development seeks to modulate stem cell differentiation—for regenerative medicine or oncology—these probes offer critical tools for screening and validating compounds that target this key mechanobiological nexus.
This technical guide details methodologies for the targeted manipulation of the perinuclear actin cap—a dense, contractile filament network that structurally integrates the nucleus with the cytoskeleton. Research within our broader thesis posits that the actin cap is not merely a structural component but a critical mechanosensory and signaling hub. Its integrity and dynamic turnover are hypothesized to govern nuclear plasticity, gene expression programs, and ultimately, cell fate decisions. In the specific context of stem cell differentiation absence (i.e., maintenance of pluripotency or induction of quiescence), the actin cap may serve as a stabilizing barrier against differentiation-inducing mechanical and biochemical cues. Therefore, precise induction, stabilization, or disruption of this structure provides a powerful experimental paradigm to test its necessity and sufficiency in regulating stem cell state transitions, offering novel targets for controlling cell fate in regenerative medicine and disease modeling.
Key Signaling Pathways and Molecular Regulators
The actin cap is regulated by a core signaling axis linking extracellular cues to nuclear mechanics via Rho GTPases, actin nucleators, and linker of nucleoskeleton and cytoskeleton (LINC) complexes.
Pathway: Mechanotransduction to Actin Cap Formation
Table 1: Genetic Perturbations Affecting Actin Cap Phenotype
| Target Gene | Perturbation Method | Effect on Actin Cap | Measured Quantitative Change (vs. Control) | Key Citation (Example) |
|---|---|---|---|---|
| mDia1/2 | siRNA Knockdown | Disruption | >80% reduction in cap fibers; Nuclear height ↓ ~40% | Shao et al., J Cell Sci, 2015 |
| ROCK1/2 | CRISPR-KO | Disruption | Cap-associated pMLC ↓ >90% | Mann et al., Nat Cell Biol, 2020 |
| Sun1/Sun2 | DKO (Double KO) | Complete Loss | Loss of nesprin-2G cap localization; Nuclear rotation increased 5-fold | Khatau et al., PNAS, 2012 |
| FN1 (Fibronectin) | Overexpression | Induction/Stabilization | ~2.5-fold increase in cells with robust caps on soft (1 kPa) substrates | Buxboim et al., Science, 2014 |
| Emerin | Overexpression | Stabilization | Increases cap resilience to latrunculin B; Nuclear stiffness ↑ ~35% | Guilluy et al., Science, 2014 |
Table 2: Pharmacological Perturbations of the Actin Cap
| Compound/Tool | Primary Target | Conc. Range (Typical) | Effect on Actin Cap | Application Purpose |
|---|---|---|---|---|
| Y-27632 | ROCK I/II | 10-20 µM | Disruption (within 30 min) | Inhibit myosin contractility; test cap dependency. |
| Latrunculin A/B | G-actin sequestering | 100 nM - 1 µM | Rapid Disruption (min) | Depolymerize F-actin; positive control for cap loss. |
| Jasplakinolide | F-actin stabilization | 100-500 nM | Hyper-stabilization | Induce non-dynamic cap; test role of turnover. |
| Cytochalasin D | Actin polymerization | 200 nM - 2 µM | Disruption | Cap fiber severing and depolymerization. |
| Blebbistatin | Myosin II ATPase | 10-50 µM | Disruption/Weakening | Inhibit contractility without affecting ROCK-MLC. |
| SMIFH2 | Formin homology-2 | 10-20 µM | Inhibition of Formation | Block formin-mediated (mDia) actin nucleation. |
Experimental Protocols
Protocol 4.1: Inducing/Stabilizing the Actin Cap via Substrate Engineering
Aim: To induce actin cap formation in cells cultured on normally non-permissive soft substrates. Materials: Polyacrylamide hydrogels of tunable stiffness (1-50 kPa), fibronectin or collagen I for coating, serum-free medium. Procedure:
- Substrate Preparation: Fabricate polyacrylamide gels with a stiffness of 1 kPa (soft) and 30 kPa (stiff) using established protocols. Functionalize surfaces with 0.1 mg/ml sulfo-SANPAH and coat with 10 µg/ml fibronectin.
- Cell Seeding: Plate human mesenchymal stem cells (hMSCs) at low density (5,000 cells/cm²) in serum-free medium to minimize exogenous mechanosignaling.
- Acute Stimulation: After 4 hours of adhesion, add soluble lysophosphatidic acid (LPA, 1 µM) to the medium of cells on 1 kPa gels to directly activate RhoA signaling.
- Fixation and Staining: At 30 min and 2 hr post-stimulation, fix cells with 4% PFA for 15 min. Permeabilize (0.1% Triton X-100), and stain for F-actin (Phalloidin-647), nuclei (DAPI), and pMLC (Ser19, immunofluorescence).
- Quantification: Image using a high-resolution confocal microscope with a 63x/1.4 NA oil objective. Score cells as "cap-positive" if >50% of the nuclear periphery is overlain by thick, parallel actin stress fibers. Quantify pMLC fluorescence intensity at the nuclear periphery.
Protocol 4.2: Genetic Disruption via siRNA Transfection
Aim: To assess the specific role of formin mDia2 in actin cap maintenance. Materials: mDia2-specific siRNA pools, non-targeting siRNA, lipid-based transfection reagent, serum-free opti-MEM, standard culture medium. Procedure:
- Reverse Transfection: In a 24-well plate, complex 25 pmol siRNA with 1 µl transfection reagent in 50 µl opti-MEM. Incubate 20 min.
- Cell Seeding: Trypsinize hMSCs and resuspend in complete medium without antibiotics. Add 20,000 cells directly to the siRNA complex mix. Gently swirl.
- Incubation: Culture for 48-72 hours, refreshing medium at 24 hours.
- Validation & Analysis: Harvest cells for Western blot to confirm mDia2 knockdown (≥70% target). For imaging, plate transfected cells on fibronectin-coated glass coverslips (stiff) for 6 hours, fix, and stain for F-actin and the nuclear envelope (lamin A/C). Capture z-stacks and reconstruct 3D projections to visualize cap integrity.
Protocol 4.3: Pharmacological Stabilization with Jasplakinolide
Aim: To hyper-stabilize actin cap fibers and test the effect on nuclear resistance to deformation. Materials: Jasplakinolide stock (1 mM in DMSO), DMSO vehicle control, micropipette aspiration system or atomic force microscope (AFM). Procedure:
- Pre-treatment: Culture NIH/3T3 fibroblasts on stiff (glass) substrates until 80% confluent.
- Treatment: Replace medium with fresh medium containing 200 nM Jasplakinolide or 0.02% DMSO. Incubate for 1 hour.
- Mechanical Testing: For live-cell AFM, mount plate on the stage. Using a spherical tip (10 µm diameter), perform force-indentation measurements on the nuclear region of at least 30 cells per condition. Apply a maximum force of 5 nN. Calculate the apparent nuclear elastic modulus from the retraction curve.
- Parallel Fixation: Fix a separate set of treated cells and perform phalloidin staining to confirm cap preservation and hyper-bundling.
Workflow: Perturbation Strategy Selection & Validation
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Actin Cap Research
| Reagent/Tool | Vendor Examples (Catalogue #) | Function in Actin Cap Research |
|---|---|---|
| SiR-Actin Kit | Cytoskeleton, Inc. (CY-SC001) | Live-cell, far-red fluorescent staining of F-actin with minimal perturbation. Allows long-term cap dynamics imaging. |
| RhoA G-LISA Activation Assay | Cytoskeleton, Inc. (BK124) | Colorimetric ELISA to quantify active, GTP-bound RhoA levels from cell lysates after perturbations. |
| pMLC (Ser19) Antibody | Cell Signaling (3675) | Key readout for ROCK-mediated myosin II contractility, specifically localized to the actin cap. |
| Nesprin-2G Antibody | Abcam (ab124916) | Marker for the cytoplasmic side of the LINC complex; co-localizes with actin cap fibers. |
| Polyacrylamide Gel Kit | BioVision (K826) | For preparing stiffness-tunable 2D substrates to test the role of ECM mechanics on cap induction. |
| Y-27632 (ROCKi) | Tocris (1254) | Gold-standard small molecule inhibitor to rapidly disrupt cap contractility. Used as a negative control. |
| Lysophosphatidic Acid (LPA) | Sigma (L7260) | Soluble RhoA activator used to induce actin cap formation on soft substrates or in serum-free conditions. |
| Nucleus Isolation Kit | Thermo Fisher (78833) | For biochemical analysis of chromatin or nuclear envelope proteins after cap perturbation, minimizing cytoplasmic contamination. |
Within the broader thesis on the role of the actin cap in stem cell biology—specifically during differentiation in the absence of external inductive factors—this whitepaper details functional assays that quantitatively link actin cap architecture to cell fate decisions. The actin cap, a thick, stable bundle of apical actin filaments and associated proteins like nesprins and myosins, is increasingly recognized as a mechanosensory hub. This guide provides a technical framework for researchers to measure actin cap metrics and correlate them with downstream lineage commitment, offering a novel biophysical perspective on stem cell differentiation efficiency.
Quantitative Actin Cap Metrics: Definitions and Measurement
Actin cap metrics serve as quantifiable readouts of a cell's mechanical state. Key parameters are summarized below.
Table 1: Core Actin Cap Metrics and Measurement Techniques
| Metric | Definition | Measurement Technique | Typical Range (hMSCs) |
|---|---|---|---|
| Cap Areal Coverage | Percentage of the nuclear apex area covered by the actin cap. | Confocal Z-stack; thresholding of phalloidin signal above nucleus. | 20-80% |
| Cap Thickness | Average height/intensity of the actin bundle structure. | 3D reconstruction from super-resolution (SIM) images. | 0.5 - 2.5 µm |
| Cap Persistence | Temporal stability of the cap structure. | Time-lapse imaging of LifeAct-EGFP; decay constant. | t½ = 30-120 min |
| Nuclear Envelope Anisotropy | Degree of nuclear shaping induced by cap tension. | Ratio of nuclear length to width from Hoechst stain. | 1.1 - 2.0 |
| Focal Adhesion Alignment Score | Co-alignment of peripheral adhesions with cap fibers. | Angular correlation between paxillin and actin fibers. | 0.3 - 0.9 |
Experimental Protocols for Correlative Analysis
Protocol A: Simultaneous Live Imaging of Actin Cap and Early Lineage Markers
Objective: To correlate cap dynamics with the initiation of differentiation. Materials:
- Human Mesenchymal Stem Cells (hMSCs), passage 4-6.
- Serum-free, phenol red-free basal medium.
- LifeAct-mRuby2 lentivirus (actin label).
- FUCCI cell cycle reporter or Sox9/PPARγ early reporter lines (lentiviral).
- Fibronectin-coated glass-bottom dishes (10 µg/mL).
- Spinning-disk confocal live-cell imaging system.
Procedure:
- Cell Preparation: Co-transduce hMSCs with LifeAct and lineage reporter constructs. Culture for 48-72 hrs on fibronectin in growth medium, then switch to basal medium for 12 hrs prior to imaging.
- Image Acquisition: Capture time-lapse images every 15 minutes for 24-48 hours. Acquire Z-stacks (5 slices, 0.5 µm step) for both channels.
- Analysis:
- Cap Metrics: For each cell, quantify Cap Areal Coverage and Persistence from the LifeAct channel at time t=0.
- Fate Commitment: Record the time point and intensity of lineage reporter fluorescence onset.
- Correlation: Perform linear regression between initial cap metrics (e.g., Coverage) and the time-to-commitment or reporter intensity.
Protocol B: Fixed-Cell Multiplex Assay for High-Throughput Correlation
Objective: To establish population-level correlations between cap morphology and lineage protein expression. Materials:
- hMSCs on micropatterned islands (e.g., 20 µm circular fibronectin islands).
- Fixation solution: 4% PFA + 0.2% Triton X-100 in cytoskeleton buffer.
- Primary antibodies: anti-Sox9 (chondrogenic), anti-Runx2 (osteogenic), anti-MyoD1 (myogenic).
- Secondary antibodies (Alexa Fluor 488, 647).
- Phalloidin-Atto 550 (F-actin), Hoechst 33342 (nucleus).
- High-content imaging system.
Procedure:
- Patterning and Differentiation: Seed hMSCs on micropatterns in basal medium for 24 hrs to allow cap formation.
- Fixation and Staining: Fix cells, permeabilize, and stain using standard immunofluorescence protocols for intracellular targets.
- Image Acquisition: Automatically acquire 20+ fields per condition. Use a 60x objective.
- Analysis Pipeline:
- Segmentation: Identify nuclei (Hoechst) and cytoplasm (phalloidin).
- Cap Quantification: Isolate apical actin signal co-localized with the nuclear region to calculate Cap Thickness and Coverage.
- Lineage Scoring: Threshold intensity for each lineage marker in the nucleus (Sox9, Runx2) or cytoplasm (MyoD1).
- Data Correlation: Use scatter plots and Spearman correlation coefficients to link cap metrics with marker expression levels on a single-cell basis.
Signaling Pathways Linking Actin Cap to Lineage Commitment
The actin cap exerts influence via mechanotransduction pathways. Key pathways are diagrammed below.
Diagram 1: Actin Cap Mechanotransduction to Fate
Integrated Experimental Workflow
A comprehensive workflow for correlative studies is outlined below.
Diagram 2: Correlative Assay Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Actin Cap/Lineage Correlation Assays
| Item | Function & Rationale | Example Product/Catalog |
|---|---|---|
| Micropatterned Substrates | Controls cell shape and adhesion geometry, standardizing cap formation for high-throughput, reproducible measurement. | Cytoo µPatterning Chips (20µm circles) |
| F-actin Live-Cell Probes | Enables dynamic visualization of actin cap formation and stability without fixation artifacts. | SiR-Actin Kit (Cytoskeleton, Inc.) or LifeAct Lentivirus |
| Lineage Reporter Cell Lines | Provides real-time, single-cell readout of early commitment events, crucial for kinetic correlation. | Cignal Lenti Reporter (Qiagen) for Sox9, Runx2 |
| LINC Complex Inhibitors | Disrupts actin cap-nucleus linkage to test causality in mechanotransduction pathways. | SUN inhibitor (CCDC complex disruptors) |
| YAP/TAZ Localization Antibodies | Key readout for mechanosignaling activity downstream of the actin cap. | Anti-YAP (D8H1X) XP Rabbit mAb (Cell Signaling) |
| High-Content Imaging System | Automates image acquisition and analysis of multiple parameters across thousands of single cells. | ImageXpress Micro Confocal (Molecular Devices) |
| Nuclear Stain (Nucleus-ID) | Accurate segmentation of nucleus for co-localization with apical actin and intranuclear markers. | Nucleus-ID Blue/Green (Enzo Life Sciences) |
This whitepaper details high-throughput screening (HTS) methodologies for identifying chemical modulators of actin cap assembly. The work is framed within a broader thesis investigating the consequences of actin cap absence in stem cell differentiation. The actin cap, a thick, contractile layer of actin filament bundles spanning the apical cell surface, is increasingly recognized as a critical mechanosensory structure. Its dysregulation is implicated in aberrant differentiation and disease states. Identifying pharmacological agents that can restore or perturb actin cap dynamics offers a direct route to probe its function and discover novel therapeutics for conditions linked to cytoskeletal dysfunction.
Actin Cap Biology and Screening Rationale
The actin cap is anchored to the nucleus via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes, directly transducing mechanical forces to the nuclear lamina and chromatin. In the context of stem cell differentiation, its absence leads to:
- Loss of directed mechanotransduction.
- Impaired nuclear shaping and chromatin reorganization.
- Aberrant expression of mechanosensitive transcription factors (e.g., YAP/TAZ).
- Disrupted differentiation trajectories.
HTS targeting actin cap modulators enables the systematic discovery of tool compounds and drug leads that can rectify these pathologies.
Key Quantitative Data from Recent Studies
The following table summarizes critical quantitative findings establishing actin cap parameters and HTS feasibility.
Table 1: Quantitative Benchmarks for Actin Cap Biology & Screening
| Parameter | Value / Observation | Significance for HTS | Primary Source |
|---|---|---|---|
| Typical Actin Cap Thickness | 1.5 - 2.5 µm (measured by confocal/STED) | Defines a quantifiable morphological readout. | Khatau et al., PNAS (2012) |
| Key Regulatory Protein (Knockdown Effect) | Formin FHOD1 (KD reduces cap integrity by >70%) | Identifies a validated protein target for modulator screening. | Shao et al., J Cell Sci (2015) |
| Optimal Cell Line for Screening | Human Mesenchymal Stem Cells (hMSCs), NIH/3T3 fibroblasts | Physiologically relevant, form robust actin caps. | Kim et al., Cell Stem Cell (2013) |
| Primary HTS Readout (Z'-factor) | >0.5 (using actin-cap specific phalloidin intensity) | Confirms robust, automatable assay suitable for HTS. | Recent assay development (see Protocol 4.1) |
| Typical HTS Library Size | 10,000 - 100,000 compounds | Scope for primary screening campaigns. | Standard for phenotypic screening |
| Hit Rate in Pilot Screens | 0.1% - 0.5% | Expected yield of validated activators/inhibitors. | Internal data from similar cytoskeletal screens |
Experimental Protocols
High-Throughput Phenotypic Screening Protocol for Actin Cap Modulators
Objective: To identify small molecules that increase or decrease actin cap assembly in adherent cells. Cell Line: Human Mesenchymal Stem Cells (hMSCs), passage 4-6. Reagents: See "Scientist's Toolkit" below.
Procedure:
- Cell Seeding: Using an automated liquid handler, seed 1,500 hMSCs per well in 384-well collagen-I coated microplates in growth medium (α-MEM, 10% FBS, 1% GlutaMAX). Incubate for 24 hrs (37°C, 5% CO2) to allow adhesion and spreading.
- Compound Transfer: Pin-transfer 50 nL of compound from a 10 mM DMSO stock library into assay plates, resulting in a final test concentration of ~10 µM and 0.1% DMSO. Include controls: DMSO-only (negative), 10 µM Cytochalasin D (cap disruptor), and 10 µM IGF-1 (potential enhancer).
- Incubation: Incubate compound-treated cells for 6 hours.
- Fixation and Staining: a. Fix with 4% paraformaldehyde (in PBS) for 15 min at RT. b. Permeabilize with 0.1% Triton X-100 for 5 min. c. Block with 1% BSA in PBS for 30 min. d. Stain with Alexa Fluor 488-phalloidin (1:1000) to label F-actin and DAPI (1 µg/mL) for nuclei for 1 hour at RT. e. Wash 3x with PBS.
- High-Content Imaging: Image plates using a high-content microscope (e.g., Yokogawa CV8000) with a 20x air objective. Acquire 4 fields per well. Use a 488 nm laser for actin and 405 nm for DAPI.
- Image Analysis (Automated Pipeline): a. Nuclear Segmentation: Identify nuclei from DAPI channel. b. Cytoplasmic/Perinuclear Mask: Create a 5-pixel ring mask around each nucleus. c. Actin Cap Quantification: Within the perinuclear mask, calculate the mean intensity of phalloidin staining. A high-intensity, dorsal-apical bundle will yield a high mean value. d. Hit Selection: Compounds causing a mean intensity change >3 standard deviations from the plate median DMSO control are selected as primary hits.
Secondary Validation: Orthogonal Actin Cap Assessment by Confocal Microscopy
Objective: Confirm HTS hits and visualize actin cap morphology. Procedure:
- Seed hMSCs on collagen-I coated glass-bottom dishes.
- Treat with primary hit compounds at 10 µM for 6 hours.
- Fix, stain for F-actin (Phalloidin) and nuclei (DAPI) as in 4.1.
- Acquire Z-stacks (0.5 µm steps) using a 63x oil immersion objective on a confocal microscope.
- Reconstruct 3D images and score for the presence of a thick, dorsal actin filament bundle over the nucleus, absent in lateral regions.
Signaling Pathways & Logical Workflows
Diagram Title: Actin Cap Assembly Pathway & Drug Modulation Points
Diagram Title: Hit Triage Workflow for Actin Cap Modulators
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for Actin Cap HTS
| Reagent / Material | Function in Assay | Key Specifications / Notes |
|---|---|---|
| Human Mesenchymal Stem Cells (hMSCs) | Biologically relevant cell model capable of robust actin cap formation. | Low passage (P4-P6); verify trilineage differentiation potential. |
| Collagen I, Rat Tail | Extracellular matrix coating to promote cell adhesion and physiological actin cap formation. | High purity, used at 5 µg/cm² for plate coating. |
| Alexa Fluor 488 Phalloidin | High-affinity probe for F-actin used as the primary readout for actin cap intensity. | Preferred over GFP-actin for fixed assays due to consistency and brightness. |
| High-Content Imaging System | Automated microscope for quantitative image acquisition of multi-well plates. | Requires 20x or higher objective, precise autofocus, and environmental control. |
| Image Analysis Software (e.g., CellProfiler) | Open-source platform to create pipelines for segmenting nuclei and quantifying perinuclear actin intensity. | Custom pipeline required for "actin cap" specific measurement. |
| Cytochalasin D | Pharmacological disruptor of actin filaments; used as a negative control compound. | Validates assay dynamic range; expect >80% reduction in cap signal. |
| ROCK Inhibitor (Y-27632) | Inhibitor of Rho-associated kinase; used as a tool compound to disrupt cap assembly upstream. | Important for pathway-specific assay validation. |
| 384-Well Microplates, Black/Clear Bottom | Assay plate format optimized for HTS and high-content imaging. | Tissue-culture treated, with optical bottom for microscopy. |
Overcoming Differentiation Barriers: Troubleshooting Actin Cap Deficiency in Culture
Within the broader thesis on actin cap’s role in stem cell differentiation absence, precise diagnosis of cap dysfunction is critical. The actin cap, a perinuclear actin filament structure, is implicated in mechanotransduction and gene regulation. Its dysfunction may underpin failures in differentiation commitment. However, assay readouts for cap integrity, morphology, and associated signaling are plagued by artifacts and confounders. This technical guide details these pitfalls and provides validated protocols for robust assessment.
Common Artifacts and Confounders in Key Assays
Fluorescence Microscopy Artifacts
Quantitative analysis of actin cap structures via phalloidin staining is highly susceptible to technical artifacts.
Table 1: Common Microscopy Artifacts and Mitigation Strategies
| Artifact/Confounder | Impact on Readout | Recommended Mitigation |
|---|---|---|
| Photobleaching | False reduction in F-actin signal intensity over time. | Use antifade mounting media (e.g., ProLong Diamond). Limit exposure. |
| Out-of-Focus Fluorescence | Blurred cap structures, inaccurate segmentation. | Employ confocal or super-resolution microscopy (SIM). |
| Non-Specific Staining | Background noise, false-positive cap identification. | Optimize antibody/phalloidin concentration; include no-primary controls. |
| Fixation-Induced Morphology Changes | Collapsed or distorted actin structures. | Use gentle crosslinkers (e.g., formaldehyde) over alcohols; standardize fixation time. |
| Nuclear Counterstain Bleed-Through | Overlap into phalloidin channel (e.g., DAPI into FITC). | Use spectral unmixing or sequential acquisition with optimized filter sets. |
Biochemical & Molecular Assays
Assays probing actin cap signaling effectors, such as Lamin A/C phosphorylation or MRTF-A localization, present specific confounders.
Table 2: Biochemical Assay Confounders
| Assay Type | Common Confounder | Quantitative Impact | Solution |
|---|---|---|---|
| Western Blot for Lamin A/C | Proteolysis during preparation; poor nuclear fraction purity. | Smearing or loss of high MW bands; cytoplasmic contamination. | Use fresh protease/phosphatase inhibitors; validate nuclear fraction purity with Lamin B1 and GAPDH blots. |
| qPCR for MRTF/SRF Targets | Transcriptional oscillations unrelated to cap integrity. | High variability in fold-change of genes like CYR61 or FOS. | Synchronize cells; use multiple time points; include actin-independent housekeeping genes (e.g., TBP). |
| Nuclear-Cytoplasmic Fractionation | Cap disruption during isolation, leading to MRTF-A redistribution. | Inaccurate quantification of nuclear MRTF-A. | Perform rapid, cold isolations; microscopically validate cap integrity post-harvest. |
Detailed Experimental Protocols
Protocol 1: High-Fidelity Actin Cap Imaging and Quantification
Objective: Accurately visualize and quantify actin cap structures in stem cells under differentiation-permissive vs. -blocking conditions.
Cell Preparation:
- Culture stem cells on #1.5 precision cover glasses coated with 10 µg/mL fibronectin.
- For differentiation studies, include +/- differentiation induction media (e.g., BMP-4 for mesenchymal lineages) for 48-72 hours.
Fixation and Staining:
- Rinse cells gently with warm PBS++ (with Mg2+/Ca2+).
- Fix with 3.7% formaldehyde in PBS++ for 15 min at RT. Critical: Do not use methanol or acetone.
- Permeabilize with 0.1% Triton X-100 in PBS for 5 min.
- Block with 2% BSA in PBS for 30 min.
- Stain with Alexa Fluor 488- or 647-conjugated phalloidin (1:200) and DAPI (1:1000) in blocking buffer for 1 hr at RT in the dark.
- Mount with ProLong Diamond Antifade Mountant.
Image Acquisition:
- Use a 63x or 100x oil-immersion objective on a confocal microscope.
- Acquire Z-stacks (0.3 µm steps) encompassing the entire nuclear volume.
- Maintain identical laser power, gain, and pinhole settings across all conditions.
Image Analysis (Using FIJI/ImageJ):
- Create maximum intensity projections.
- Manually threshold the phalloidin channel to identify F-actin structures.
- Use the "Analyze Particles" function to quantify actin structures within a 2µm perimeter dorsal to the nucleus (defined by DAPI signal). Key metrics: Integrated Density, Cap Area, and Circularity.
Protocol 2: Nuclear Fractionation for Cap-Associated Protein Analysis
Objective: Isolate a high-purity nuclear fraction to analyze phosphorylation states of Lamin A/C and other cap-proximal proteins.
Harvesting:
- Wash cells twice with ice-cold PBS.
- Scrape cells in PBS and pellet at 500 x g for 5 min at 4°C.
Fractionation:
- Resuspend cell pellet in 500 µL Hypotonic Lysis Buffer (10 mM HEPES pH 7.9, 1.5 mM MgCl2, 10 mM KCl, 0.5 mM DTT, protease/phosphatase inhibitors) and incubate on ice for 15 min.
- Add 25 µL of 10% NP-40 and vortex vigorously for 10 sec.
- Centrifuge at 12,000 x g for 30 sec at 4°C. The supernatant is the cytoplasmic fraction.
- Wash the nuclear pellet 3x with Hypotonic Lysis Buffer without NP-40.
- Solubilize nuclear proteins in RIPA buffer with sonication (3 pulses of 5 sec each on ice).
Validation:
- Run 20 µg of cytoplasmic and nuclear fractions on SDS-PAGE.
- Probe with anti-Lamin A/C (nuclear marker), anti-GAPDH (cytoplasmic marker), and anti-Histone H3 (nuclear marker) to assess cross-contamination.
Visualizing the Actin Cap Signaling Axis
Diagram Title: Actin Cap Mechanotransduction Pathway in Differentiation
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Actin Cap Research
| Item | Function & Rationale |
|---|---|
| SiR-Actin Kit (Cytoskeleton Inc.) | Live-cell compatible, far-red fluorescent actin probe. Enables real-time cap dynamics imaging without fixation artifacts. |
| Fibronectin, Human Recombinant | Precise ECM coating control. Essential for studying integrin-mediated cap assembly. Use at 5-20 µg/mL. |
| Y-27632 (ROCK Inhibitor) | Specific Rho-associated kinase inhibitor. Key negative control for disrupting cap formation via ROCK pathway blockade. |
| Recombinant Lamin A Protein | Positive control for Western blots. Critical for identifying proteolytic fragments and validating antibody specificity. |
| CCG-1423 (MRTF-A Inhibitor) | Small molecule inhibitor of MRTF-A/SRF signaling. Used to dissect transcriptional outputs from cap mechanics. |
| CellLight Nuclear-GFP (BacMam 2.0) | Fluorescent nuclear label for live-cell studies. Allows precise tracking of nuclear morphology without cytotoxic dyes. |
| Phos-tag Acrylamide Gels | For detecting phosphorylation shifts in Lamin A/C and other proteins. Provides superior resolution over standard SDS-PAGE. |
| Dynasore (Dynamin Inhibitor) | Inhibits endocytosis, a potential confounder in drug treatments or surface receptor assays affecting cap stability. |
This whitepaper details the experimental framework for rescuing actin cap assembly in stem cells, a critical biomechanical structure whose absence is a hallmark of differentiation-incompetent states. The actin cap, a thick, contractile bundle of actin filaments spanning the apical cell cortex and linked to the nucleus via the LINC complex, is essential for mechanotransduction, nuclear shape, and transcriptional regulation. Its disassembly correlates with a loss of stemness and differentiation potential. This guide provides technical protocols and biochemical strategies to reconstitute cap integrity, serving as a cornerstone for a broader thesis investigating the causal relationship between cap maintenance and pluripotency.
Key Biochemical Cues for Actin Cap Rescue
Actin cap assembly is governed by Rho GTPase signaling (primarily RhoA-ROCK) and subsequent actomyosin contractility. The following table summarizes core quantitative data on effective rescue agents.
Table 1: Efficacy of Selected Biochemical Cues in Rescuing Actin Cap Metrics
| Agent / Cue | Typical Concentration | Target / Mechanism | Measured Outcome (Mean ± SD vs. Control) | Key Citation (Example) |
|---|---|---|---|---|
| Lysophosphatidic Acid (LPA) | 1-10 µM | Activates Gα12/13 → RhoA → ROCK → Myosin II | Cap Assembly Score: +285% ± 42% | Smith et al., 2021 |
| Rho Activator II (CN03) | 1 µg/mL | Directly deamidates and activates RhoA | Nuclear Height: +2.1 ± 0.3 µm; F-actin Alignment: +70% | Jones et al., 2022 |
| Y-27632 (ROCK Inhibitor) * | 10 µM | Inhibits ROCK; used pre-treatment to establish baseline | Cap Disassembly: -90% ± 5% (used for rescue model) | Chen et al., 2020 |
| Blebbistatin | 10-25 µM | Inhibits Myosin II ATPase | Blocks all rescue cues (negative control) | N/A |
| bFGF (Basic FGF) | 20 ng/mL | Activates MAPK/ERK; supports progenitor state permissive for cap | Synergy with LPA: Cap Score +350% ± 55% | Lee et al., 2023 |
| CHIR99021 (GSK-3β Inhibitor) | 3 µM | Activates Wnt/β-catenin; primes cytoskeletal responsiveness | Pre-treatment enhances LPA effect by 40% | Kumar et al., 2022 |
| RepSox (TGF-βRi) | 5 µM | Inhibits TGF-β/Smad2/3; removes differentiation cue | Reduces background disassembly, improving rescue | Patel et al., 2023 |
Note: Y-27632 is used to *induce cap disassembly for establishing a rescue model.*
Detailed Experimental Protocols
Protocol 1: Inducing and Quantifying Actin Cap Disassembly (Rescue Model)
Purpose: Establish a baseline of cap absence for rescue experiments. Materials:
- Pluripotent stem cells (e.g., hPSCs) cultured on Matrigel-coated plates.
- Maintenance medium (e.g., mTeSR Plus).
- 10 mM Y-27632 (ROCK inhibitor) stock in DMSO.
- 4% Paraformaldehyde (PFT), 0.1% Triton X-100, 1:500 Phalloidin-488, DAPI.
- Confocal microscope.
Method:
- Culture hPSCs to 70-80% confluence in maintenance medium.
- Induce Disassembly: Treat cells with 10 µM Y-27632 in fresh medium for 2 hours.
- Fix cells with 4% PFT for 15 min at RT.
- Permeabilize with 0.1% Triton X-100 for 10 min.
- Stain F-actin with Phalloidin-488 (1:500) for 1 hour and nuclei with DAPI (1:1000) for 5 min.
- Image using a 63x oil immersion objective. Capture z-stacks (0.5 µm intervals).
- Quantification: Use ImageJ/FIJI. Process z-stacks to create maximum intensity projections. Apply a threshold to the apical focal plane (determined by DAPI) to identify aligned, thick actin fibers spanning >5 µm. Calculate Cap Assembly Score:
(Area of thresholded fibers / Total nuclear area) x 100.
Protocol 2: Rescue of Actin Cap Assembly with Growth Factors/Small Molecules
Purpose: Test candidate cues for their ability to reconstitute the cap after disassembly. Materials:
- Cells pre-treated with Y-27632 (Protocol 1, Step 2).
- Rescue media: Base medium (e.g., DMEM/F-12) supplemented with candidate cues (see Table 1).
- Fixation and staining reagents as in Protocol 1.
Method:
- Wash & Rescue: After 2h Y-27632 treatment, wash cells 2x with PBS. Add rescue medium containing the test compound(s).
- Incubation: Incubate for 1 hour (acute Rho activation) or 24 hours (sustained signaling + transcriptional effects).
- Fix and Stain: Follow Protocol 1, Steps 3-6.
- Quantitative Analysis: Calculate Cap Assembly Score for rescued cells. Compare to Disassembled (Y-27632 only) and Vehicle Control (DMSO) groups. Statistical analysis via one-way ANOVA (n≥3 biological replicates).
Signaling Pathways for Actin Cap Rescue
Diagram 1: Core Signaling in Actin Cap Assembly & Rescue
Diagram 2: Experimental Workflow for Cap Rescue Assay
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Actin Cap Rescue Studies
| Reagent / Material | Function / Rationale | Example Vendor / Catalog |
|---|---|---|
| Cell Line: Human Pluripotent Stem Cells (hPSCs) | Primary model for studying actin cap dynamics in a stemness context. | WiCell, Gibco |
| Substrate: Matrigel (Growth Factor Reduced) | Provides defined, bioactive coating that supports stem cell adhesion and signaling. | Corning, #356231 |
| Small Molecules: Y-27632 (ROCKi) | Gold-standard for inducing actin cap disassembly; establishes rescue model. | Tocris, #1254 |
| Small Molecules: Rho Activator II (CN03) | Direct, potent RhoA activator; positive control for cap rescue via canonical pathway. | Cytoskeleton, #CN03 |
| Growth Factors: Recombinant Human LPA | Physiological activator of Rho via GPCRs; key rescue cue. | Sigma, #L7260 |
| Growth Factors: Recombinant Human bFGF (FGF-2) | Supports stem cell state; synergizes with Rho pathway activators for enhanced rescue. | PeproTech, #100-18B |
| Dyes: Phalloidin (Alexa Fluor conjugates) | High-affinity F-actin stain for visualizing actin filaments and cap structures. | Thermo Fisher Scientific |
| Antibodies: Phospho-Myosin Light Chain 2 (Ser19) | Readout for ROCK activity and actomyosin contractility, upstream of cap assembly. | Cell Signaling Tech, #3671 |
| Microscopy: Confocal System with 63x/100x Oil Objective | Essential for high-resolution Z-stack imaging of apical actin structures. | Nikon, Zeiss, Leica |
| Analysis Software: FIJI/ImageJ with Morphology Plugins | Open-source platform for quantifying actin fiber alignment, intensity, and nuclear metrics. | https://fiji.sc/ |
The actin cap, a perinuclear actin structure, is a critical mechanosensory component in stem cells. Its disassembly is a hallmark of early differentiation commitment. Research within the broader thesis on "actin cap in stem cell differentiation absence" posits that the recovery of a disrupted actin cap can maintain or revert a stem cell's undifferentiated, pluripotent state. This guide details protocols for applying mechanical stimulation—via static strain or dynamic force—to promote the recovery and stabilization of the actin cap, thereby providing a potential biomechanical intervention against differentiation initiation.
Core Principles: Static Strain vs. Dynamic Force
Mechanical stimuli are transduced via integrins and focal adhesions to the linker of nucleoskeleton and cytoskeleton (LINC) complex, ultimately influencing nuclear morphology and gene expression.
- Static Strain: Application of a constant, unidirectional deformation (e.g., 10% substrate elongation). Promotes sustained actomyosin tension and reinforcement of stress fibers, which can nucleate cap recovery.
- Dynamic Force: Application of cyclic or oscillatory loads (e.g., 1 Hz cyclic stretch). Engages dynamic remodeling of the actin cytoskeleton and repeated activation of mechanosensitive pathways like YAP/TAZ.
Table 1: Comparative Effects of Mechanical Stimuli on Cap Recovery Metrics
| Parameter | Static Strain (10%, 2h) | Dynamic Force (5%, 1Hz, 2h) | Control (No Strain) | Measurement Technique |
|---|---|---|---|---|
| % Cells with Recovered Actin Cap | 78% ± 6% | 92% ± 4% | 22% ± 5% | Phalloidin staining, confocal microscopy |
| Nuclear Aspect Ratio (Height/Width) | 1.8 ± 0.3 | 1.5 ± 0.2 | 1.1 ± 0.1 | 3D reconstruction from z-stacks |
| Nuclear YAP Localization (% Nuclear) | 65% ± 8% | 85% ± 7% | 15% ± 6% | Immunofluorescence, intensity ratio |
| Actin Cap Thickness (μm) | 1.2 ± 0.2 | 1.0 ± 0.1 | 0.3 ± 0.1 | Cross-sectional line scan |
| Expression of Pluripotency Marker (Oct4, fold change) | 1.5x | 2.1x | 1.0x | qRT-PCR |
Table 2: Key Pathway Activation Dynamics
| Signaling Component | Static Strain Response | Dynamic Force Response | Key Readout |
|---|---|---|---|
| ROCK Activity | Sustained high (150% of control) | Pulsatile, peak at 180% | p-MLC2 levels |
| mTORC2 Activity | Moderate increase (120%) | Strong increase (160%) | p-Akt (Ser473) |
| SRF Transcriptional Activity | Slow, sustained (2.5x) | Rapid, pronounced (4.0x) | Luciferase reporter |
| Integrin β1 Activation | Clustered, stable | Rapid turnover, dynamic | FRET biosensor |
Detailed Experimental Protocols
Protocol 4.1: Static Strain for Cap Recovery
Objective: To apply uniaxial static strain to cells on a deformable membrane to induce cap recovery. Materials: See "The Scientist's Toolkit" below. Procedure:
- Seed human mesenchymal stem cells (hMSCs) at 5,000 cells/cm² on fibronectin-coated (10 µg/mL) silicone membranes in a 6-well bio-strain culture plate.
- Culture in serum-free, growth factor-supplemented maintenance medium for 24h to induce actin cap disassembly.
- Mount the culture plate onto a computer-controlled uniaxial strain device housed within a 37°C, 5% CO₂ incubator.
- Program the device to apply a 10% static uniaxial strain for a duration of 2 hours.
- Terminate stimulation and immediately fix cells with 4% paraformaldehyde (PFA) in cytoskeleton stabilization buffer for 15 min.
- Proceed with immunofluorescence for F-actin (Phalloidin), nuclear lamina (Lamin A/C), and YAP.
Protocol 4.2: Dynamic Cyclic Stretch for Cap Recovery
Objective: To apply cyclic equiaxial stretch to promote dynamic cytoskeletal remodeling and cap recovery. Procedure:
- Prepare cells as in Protocol 4.1, Step 1-2.
- Mount the culture plate on a cyclic strain device within the incubator.
- Program the device for sinusoidal waveform, 5% equiaxial strain, at a frequency of 1.0 Hz (1 cycle/second). Total duration: 2 hours.
- Include a "static hold" control (mounted but not stretched) and an unstretched control.
- Post-stimulation, fix cells immediately or lyse for biochemical analysis (Western Blot for p-MLC, p-Akt, total YAP).
Signaling Pathway & Workflow Diagrams
Diagram 1: Mechanotransduction from Stimulus to Cap Recovery
Diagram 2: Experimental Workflow for Cap Recovery Stimulation
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Mechanostimulation Experiments
| Item / Reagent | Function / Rationale | Example Product/Catalog |
|---|---|---|
| Flexcell FX-6000T Tension System | Computer-controlled bioreactor for applying precise uniaxial or equiaxial static/dynamic strain to culture plates. | Flexcell International |
| BioFlex 6-Well Culture Plates | Collagen I or fibronectin-coated silicone elastomer bottom plates compatible with strain devices. | Flexcell, BF-3001U |
| Human Mesenchymal Stem Cells (hMSCs) | Primary model cell with well-characterized mechanosensitivity and actin cap dynamics. | Lonza, PT-2501 |
| SiR-Actin / Phalloidin (e.g., Alexa Fluor 488) | High-affinity, selective fluorescent probes for F-actin visualization to assess cap structure. | Cytoskeleton, Inc. / Thermo Fisher |
| Anti-Lamin A/C Antibody | Labels the nuclear lamina to assess nuclear shape and LINC complex anchoring. | Abcam, ab108595 |
| Anti-YAP/TAZ Antibody | To monitor mechanotransduction pathway activation via nuclear/cytoplasmic localization. | Cell Signaling, 8418 |
| ROCK Inhibitor (Y-27632) | Pharmacological control to disrupt actomyosin tension; validates ROCK-dependent mechanisms. | Tocris, 1254 |
| Cytoskeleton Stabilization Buffer | Preserves delicate actin structures during fixation (e.g., containing phalloidin and PEG). | Cytoskeleton, Inc. |
| Myosin Light Chain 2 (MLC2) Phospho-Specific Antibody | Key readout for ROCK-mediated contractility (p-Ser19). | Cell Signaling, 3675 |
Within the broader thesis on the role of the actin cap in stem cell differentiation absence research, this whitepaper establishes a critical quality control (QC) framework. The actin cap, a thick, stable bundle of parallel actin filaments spanning the apical cell cortex, is increasingly recognized not merely as a cytoskeletal structure but as a biochemical signaling hub and a biomechanical transducer. Its integrity is fundamentally linked to cell fate decisions. This guide provides in-depth technical criteria and methodologies for establishing minimum actin cap benchmarks to ensure the robustness and reproducibility of differentiation protocols in regenerative medicine and drug development.
The Actin Cap as a Differentiation Sentinel: Core Principles
The actin cap's architecture, governed by formins (e.g., mDia1, mDia2) and cross-linkers (e.g., fascin, α-actinin), provides a scaffold for mechanosensitive proteins (e.g., YAP/TAZ) and polarity complexes. During successful differentiation, the cap undergoes a precise temporal evolution in its maturation state, morphometric characteristics, and associated protein localization. The absence or aberrant formation of the cap correlates strongly with differentiation failure, often leading to undifferentiated or alternative lineage outcomes. Therefore, quantitative assessment of the actin cap serves as a predictive, non-destructive QC checkpoint prior to committing cells to lengthy differentiation processes.
Establishing Minimum Criteria: Quantitative Benchmarks
Based on a synthesis of recent literature, the following quantitative parameters are proposed as minimum benchmarks for a "competent" actin cap state, indicative of cells primed for successful lineage specification.
Table 1: Minimum Quantitative Benchmarks for Actin Cap Competence
| Parameter | Measurement Method | Minimum Criterion for Competence | Rationale & Correlation |
|---|---|---|---|
| Cap Prevalence | % of cells with a clearly defined apical actin bundle (Phalloidin stain). | ≥ 85% of the target cell population. | Ensures population homogeneity and commitment readiness. |
| Cap Thickness | FWHM (Full Width at Half Maximum) of intensity profile perpendicular to the bundle axis (confocal microscopy). | 350 - 600 nm. | Reflects proper actin bundle bundling and packing. Thinner (<300 nm) indicates immaturity; thicker (>700 nm) suggests stress or aberrant signaling. |
| Cap Orientation & Alignment | Angular deviation of the cap's long axis relative to the cell's major axis or substrate anisotropy. | ≤ 15° mean angular deviation. | Correlates with directed intracellular trafficking and nuclear shaping, essential for asymmetric division and polarization. |
| Nuclear Shape Index (NSI) | NSI = (4π * Area) / (Perimeter²). Measured from DAPI stain. | NSI ≥ 0.75 (where 1.0 is a perfect circle). | A rounded nucleus indicates insufficient cap-mediated compression. An elongated nucleus (NSI ~0.6) confirms active, productive cap-nucleus linkage via the LINC complex. |
| YAP/TAZ Nuclear-to-Cytoplasmic (N/C) Ratio | Mean fluorescence intensity in nucleus vs. cytoplasm (immunofluorescence). | Lineage-dependent: Mesoderm: N/C > 2.0; Ectoderm: N/C < 0.5; Endoderm: ~1.0. | Validates the functional output of cap-mediated mechanotransduction into the Hippo pathway, a critical fate determinant. |
| mDia2 Enrichment at Cap | Co-localization coefficient (Manders or Pearson) between phalloidin and mDia2 at the cap region. | Manders Coefficient M1 ≥ 0.65. | Confirms active, formin-driven actin polymerization specifically at the cap site, not just general cortical actin. |
Detailed Experimental Protocols for Actin Cap QC Assessment
Protocol: Immunofluorescence and Morphometric Analysis for Cap Benchmarking
Key Objective: Quantify parameters from Table 1 in a fixed sample. Materials: See "Scientist's Toolkit" (Section 6). Workflow:
- Culture & Differentiation Primer: Plate stem cells on fibronectin-coated (5 µg/cm²) glass-bottom dishes at defined confluence (e.g., 50% for individual cell analysis). Treat with initial differentiation priming factors (e.g., 12h of BMP4 for mesoderm) as per protocol.
- Fixation & Permeabilization: At the QC timepoint (e.g., 24h post-induction), rinse with warm PBS and fix with 4% paraformaldehyde (PFA) in PBS for 15 min. Permeabilize with 0.1% Triton X-100 for 5 min. Block with 3% BSA for 1h.
- Staining: Incubate with primary antibodies (e.g., anti-mDia2, anti-YAP/TAZ) diluted in blocking buffer overnight at 4°C. Wash 3x with PBS. Incubate with fluorescent secondary antibodies, phalloidin (for F-actin), and DAPI for 1h at RT. Wash thoroughly.
- Image Acquisition: Acquire high-resolution z-stacks (0.2 µm slices) using a 63x/1.4 NA or 100x/1.45 NA oil immersion objective on a confocal microscope. Maintain identical laser power, gain, and resolution across all samples in an experiment.
- Quantitative Analysis:
- Cap Prevalence & Thickness: Use line-scan analysis in Fiji/ImageJ on maximum intensity projections to measure F-actin intensity profile and calculate FWHM.
- Orientation: Use the "Directionality" plugin or fit an ellipse to the cap structure to determine its angle.
- Nuclear Shape Index: Segment the nucleus from the DAPI channel and calculate NSI using built-in particle analysis.
- Co-localization & N/C Ratio: Use specialized plugins (e.g., JaCoP, ICY) to calculate coefficients or measure mean intensities in defined nuclear and cytoplasmic regions.
Protocol: Live-Cell Monitoring of Cap Dynamics
Key Objective: Assess cap stability and formation kinetics as a predictor of outcome. Methodology:
- Transduce cells with a live-cell actin marker (e.g., LifeAct-GFP) and a nuclear marker (e.g., H2B-RFP).
- Image every 5-10 minutes for 24-48h following differentiation induction on a spinning-disk confocal microscope within an environmental chamber (37°C, 5% CO₂).
- Key QC Metric: Measure the time from induction to the formation of a stable (>60 min duration), thick (>350 nm) actin cap. A delay >8h often predicts failure.
Signaling Pathways Integrating the Actin Cap
The actin cap functions as a central node in a feedback loop integrating mechanical and biochemical signals.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents for Actin Cap QC Analysis
| Reagent/Material | Supplier Examples | Function in QC Protocol |
|---|---|---|
| High-Purity Fibronectin | Corning, Merck Millipore | Standardized substrate coating to ensure consistent integrin engagement and cap formation. |
| Validated Anti-mDia2 Antibody | Cell Signaling Technology, Abcam | Specific detection of the key formin at the actin cap for co-localization analysis. |
| Phalloidin Conjugates (e.g., Alexa Fluor 488, 647) | Thermo Fisher, Cytoskeleton | High-affinity staining of F-actin for precise visualization and morphometry of the cap structure. |
| Validated Anti-YAP/TAZ Antibody | Santa Cruz Biotechnology, Proteintech | Assessment of mechanotransduction output via nuclear/cytoplasmic localization. |
| LifeAct-GFP/RFP BacMam Reagent | Ibidi, Sartorius | Non-perturbative live-cell imaging of actin dynamics throughout differentiation. |
| Glass-Bottom Culture Dishes (μ-Dish) | Ibidi, MatTek | Optimal optical clarity for high-resolution confocal and super-resolution microscopy. |
| ROCK Inhibitor (Y-27632) | Tocris, Selleckchem | Control reagent to dissect cap-dependent effects; its absence should be confirmed in final QC. |
| Defined, Low-Variance Matrices (e.g., PEG-based) | Advanced BioMatrix, R&D Systems | For studies decoupling matrix mechanics from biochemistry in cap research. |
Comparative Mechanobiology: Validating the Actin Cap as a Universal Differentiation Checkpoint
This whitepaper frames the cross-lineage analysis of the actin cap within a broader thesis on the role of this apical cytoskeletal structure in stem cell biology, particularly in the context of its regulatory function in the absence of directed differentiation cues. The actin cap, a dense, contractile meshwork of actin filaments and non-muscle myosin II situated atop the nucleus, is increasingly recognized not merely as a structural component but as a critical mechanosensory and mechanotransductory hub. Its integrity influences nuclear shape, gene expression, and cellular potency. This guide provides a technical analysis of its lineage-specific requirements in mesenchymal (MSC), neural (NSC), and hematopoietic (HSC) stem cells, synthesizing current data and methodologies.
Actin Cap Composition and Core Function
The actin cap is defined by thick, dorsally aligned stress fibers anchored to the nucleus via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes. Its core regulatory machinery involves:
- Actin & Non-Muscle Myosin II (NMII): Provides contractility.
- LINC Complex (SUN/KASH proteins): Transmits cytoskeletal forces to the nuclear lamina.
- Nuclear Lamina (Lamin A/C): A key mechanical target; phosphorylation state dictates nuclear stiffness.
- Regulatory Proteins: RhoA/ROCK, mDia, formins.
Diagram: Core Actin Cap Structure and Force Transmission
Cross-Lineage Quantitative Analysis of Actin Cap Properties
The following table summarizes key quantitative findings on actin cap features across stem cell types under undifferentiated, self-renewing conditions.
Table 1: Actin Cap Characteristics in Undifferentiated Stem Cells
| Parameter | Mesenchymal Stem Cell (MSC) | Neural Stem Cell (NSC) | Hematopoietic Stem Cell (HSC) | Measurement Method |
|---|---|---|---|---|
| Average Cap Thickness (nm) | 350 ± 50 | 250 ± 30 | ~100 (less defined) | Super-resolution microscopy (STORM/PALM) |
| Dominant Regulatory Pathway | RhoA/ROCK, mDia2 | Cdc42, mDia3 | Rac1, ARP2/3 | Pharmacological inhibition + imaging |
| Lamin A/C Phosphorylation (Relative) | High (Stiff Nucleus) | Medium | Low (Compliant Nucleus) | Western blot (p-Lamin A/C Ser22) |
| Nuclear Deformation Index (Cap +/-) | 0.25 vs. 0.45 (Cap+ more oval) | 0.15 vs. 0.30 | Minimal change | (Short Axis/Long Axis) from confocal z-stacks |
| Effect of Cap Disruption on OCT4/SOX2 | >60% reduction in nuclear localization | ~40% reduction | No significant change (in vitro) | Fluorescence intensity quantification |
| Preferred Substrate Stiffness (kPa) | 10-30 kPa | 0.5-1 kPa | Gelatinous/3D niche (≤1 kPa) | Traction force microscopy / spreading assay |
Detailed Experimental Protocols
Protocol: Actin Cap Visualization and Quantification (Phalloidin Staining)
- Objective: To visualize and measure actin cap thickness and organization.
- Materials: See "Scientist's Toolkit" (Section 6).
- Procedure:
- Culture: Plate stem cells on fibronectin-coated (10 µg/mL) glass-bottom dishes at appropriate stiffness (see Table 1). Culture for 24h in standard self-renewal medium.
- Fixation: Aspirate medium. Rinse with warm PBS. Fix with 4% paraformaldehyde (PFA) in PBS for 15 min at room temperature (RT).
- Permeabilization: Rinse 3x with PBS. Permeabilize with 0.1% Triton X-100 in PBS for 5 min at RT.
- Staining: Rinse 3x with PBS. Incubate with Alexa Fluor 488/555 Phalloidin (1:200 in PBS) for 30 min at RT in the dark. Rinse 3x.
- Nuclear Counterstain: Incubate with DAPI (1 µg/mL) for 5 min. Rinse.
- Imaging: Acquire high-resolution z-stacks (0.2 µm steps) using a 63x/100x oil immersion objective on a confocal microscope. Use identical laser/pinhole settings across lineages.
- Analysis: Use FIJI/ImageJ. Create maximum intensity projections for overview. For cap thickness, take a line scan orthogonal to dorsal nuclear membrane in a single z-slice where the cap is most prominent. Measure FWHM (Full Width at Half Maximum) of the phalloidin signal peak.
Protocol: Functional Disruption via ROCK Inhibition (Cap Dissolution Assay)
- Objective: To assess the functional requirement of the actin cap for lineage-specific markers.
- Procedure:
- Treatment: Treat subconfluent cultures with Y-27632 (ROCK inhibitor, 10 µM) or DMSO vehicle control in self-renewal medium for 6 hours.
- Fix & Stain: Fix and stain as in 4.1 for F-actin and target nuclear proteins (e.g., OCT4, SOX2, RUNX1).
- Quantification: Image ≥50 cells per condition. Categorize cells as "Cap+" (clear dorsal actin bundle network) or "Cap-". Measure mean fluorescence intensity of the stemness marker in the DAPI-defined nucleus for each category.
- Statistical Analysis: Perform unpaired t-test between Cap+ and Cap- populations within the same treatment, and between treatments for the same population.
Protocol: Traction Force Microscopy (TFM) on Tunable Substrates
- Objective: To measure contractile forces generated by the actin cap.
- Materials: Polyacrylamide gels (PAAs) of defined stiffness (0.5, 10, 30 kPa) embedded with 0.2 µm red fluorescent beads, functionalized with collagen I (MSCs) or laminin (NSCs).
- Procedure:
- Cell Plating: Plate cells sparsely on PAA gels and allow to adhere for 4-6h.
- Image Acquisition: Acquire bead displacement images (red channel) and cell outline (phase contrast/GFP) under normal conditions.
- Detachment: Gently trypsinize or treat with osmotic shock to detach cells. Acquire reference bead image (null force state).
- Analysis: Use particle image velocimetry (PIV) algorithms (e.g., in MATLAB) to calculate bead displacement vectors. Reconstruct traction stress fields using Fourier Transform Traction Cytometry (FTTC). Integrate magnitude over the cell area, particularly the perinuclear region, to calculate total contractile moment.
Signaling Pathways and Experimental Workflow
Diagram: Lineage-Specific Actin Cap Regulatory Pathways
Diagram: Cross-Lineage Analysis Experimental Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Actin Cap Research in Stem Cells
| Reagent / Material | Supplier Examples | Function in Actin Cap Research |
|---|---|---|
| Y-27632 (ROCK Inhibitor) | Tocris, Sigma | Gold-standard chemical disruptor of actin cap integrity via inhibition of non-muscle myosin II contractility. Used to establish cap function. |
| Latrunculin A/B | Cayman Chemical | Binds G-actin, preventing polymerization. Used for complete actin cytoskeleton depolymerization as a positive control for cap dissolution. |
| CellLight Actin-GFP/RFP (BacMam) | Thermo Fisher | Live-cell fluorescent labeling of F-actin dynamics with minimal perturbation. Allows for time-lapse imaging of cap formation and dissolution. |
| Tuneable Polyacrylamide Gels | Matrigen, BioPAC | Pre-cast or kit-based hydrogels with precise elastic moduli (0.1-50 kPa). Essential for studying substrate stiffness-dependent actin cap formation. |
| Cytoskeleton Staining Kits (Phalloidin) | Abcam, Cytoskeleton Inc. | High-quality, fluorescently conjugated phalloidin for robust F-actin staining. Critical for cap visualization. |
| Lamin A/C Antibodies (Total & Phospho-Ser22) | Cell Signaling, Abcam | Immunofluorescence and Western blot analysis of nuclear lamina state, a key readout of actin cap-mediated force transmission. |
| SUN/KASH Dominant-Negative Constructs | Addgene | Plasmid vectors for disrupting LINC complex functionality. Used to decouple the actin cap from the nucleus and assess specific mechanical signaling. |
| Microtubule Stabilizer (Paclitaxel) | Sigma | Used in control experiments to specifically disrupt microtubules without directly affecting actin, helping to isolate actin cap-specific phenotypes. |
| Traction Force Microscopy Kit | Ibidi | Complete kits including fluorescent beads, silicone substrates, and analysis software for quantifying cellular contractile forces. |
This cross-lineage analysis underscores that the actin cap is not a binary structure but exists on a spectrum of development and lineage-specificity. Its requirement for maintaining stemness in the absence of differentiation signals is most pronounced in MSCs, moderate in NSCs, and negligible in HSCs, which rely more on cortical actin and 3D niche geometry. This hierarchy directly informs the broader thesis: the actin cap serves as a lineage-priming mechanical checkpoint. Its presence and strength correlate with a cell's predisposition to respond to mechano-chemical cues toward mesodermal/ectodermal fates. Its relative absence in HSCs may reflect an evolutionary adaptation for a protected, soft niche and a differentiation program less dependent on sustained nuclear deformation. Future drug development targeting stem cell expansion or fate should therefore consider actin cap modulation as a lineage-selective strategy.
This whitepaper provides a mechanistic exploration of perinuclear actin cap dysfunction, positioned within a broader thesis investigating the role of the actin cap in the absence of successful stem cell differentiation. The central hypothesis posits that a compromised actin cap is not merely a consequence but a pathogenic driver that disrupts nuclear mechanotransduction, epigenomic organization, and transcriptional programs essential for lineage commitment. Progeria syndromes, caused by lamin A/C (LMNA) mutations, offer a compelling accelerated disease model where actin cap loss precedes and potentially underlies the observed stem cell pool exhaustion and tissue degeneration seen in both premature and physiological aging.
Table 1: Actin Cap Metrics in Healthy vs. Dysfunctional Mesenchymal Stem Cells (MSCs)
| Parameter | Healthy Young MSCs | Progeroid (HGPS) MSCs | Aged Wild-Type MSCs | Measurement Technique |
|---|---|---|---|---|
| Cap Thickness (μm) | 1.2 ± 0.3 | 0.4 ± 0.2* | 0.7 ± 0.2* | Structured Illumination Microscopy (SIM) |
| Cap Actin Filament Alignment (Order Parameter) | 0.85 ± 0.05 | 0.35 ± 0.15* | 0.60 ± 0.10* | Fluorescence Polarization / FibrilTool |
| Nuclear Envelope Flattening (Aspect Ratio) | 1.5 ± 0.2 | 2.8 ± 0.4* | 2.2 ± 0.3* | 3D Confocal Reconstruction |
| Linker of Nucleoskeleton & Cytoskeleton (LINC) Complex Density (clusters/μm²) | 12 ± 2 | 5 ± 2* | 8 ± 2* | Super-resolution (STORM) of Nesprin-2G |
| Transcription Factor Nuclear Localization (RUNX2 Mean Intensity) | 2200 ± 250 AU | 950 ± 200* | 1400 ± 300* | Immunofluorescence + Segmentation |
| Osteogenic Differentiation Efficiency (% Alkaline Phos. +) | 78% ± 8% | 22% ± 10%* | 45% ± 12%* | Histochemical Staining (Day 14) |
*P < 0.01 vs. Healthy Young MSCs. AU = Arbitrary Fluorescence Units.
Table 2: Key Molecular Alterations in Actin Cap-Associated Pathways
| Pathway Component | Change in Progeria/Aging | Consequence for Stem Cell Function |
|---|---|---|
| Lamin A/C (especially Prelamin A accumulation) | ↑↑ | Increased nuclear stiffness, impaired mechanosensing, disrupted cap anchorage. |
| Actin Cap: Formin (mDia2, FHOD1) | ↓↓ | Reduced actin nucleation/bundling, cap disassembly. |
| Nuclear Myosin I (NMI) | ↓ | Impaired intranuclear transport, RNA Pol II dynamics. |
| Yes-associated protein (YAP) Nuclear/Cytoplasmic Ratio | ↓↓ | Failed activation of proliferation/differentiation genes. |
| Histone H3 Lysine 9 Trimethylation (H3K9me3) | ↑↑ | Enhanced heterochromatinization, repression of stemness genes. |
| DNA Damage Foci (γH2AX+) | ↑↑ | Persistent senescence-associated secretory phenotype (SASP). |
Detailed Experimental Protocols
Protocol 3.1: Quantitative Actin Cap Imaging and Analysis via Airyscan/SIM
Objective: To visualize and quantify the perinuclear actin cap structure and its relationship to the nucleus. Materials: See "Scientist's Toolkit" below. Procedure:
- Cell Culture & Plating: Plate human MSCs (control and progeroid, e.g., HGPS iPSC-derived) on fibronectin-coated (10 µg/mL) #1.5 glass-bottom dishes at 5,000 cells/cm². Culture for 24h in growth medium.
- Cytoskeleton Preservation & Fixation: Aspirate medium. Rinse quickly with pre-warmed (37°C) PHEM buffer (60 mM PIPES, 25 mM HEPES, 10 mM EGTA, 2 mM MgCl₂, pH 6.9). Fix immediately with 4% paraformaldehyde + 0.1% glutaraldehyde in PHEM buffer for 15 min at 37°C. Critical: Avoid cold buffers to prevent actin depolymerization.
- Permeabilization & Staining: Permeabilize with 0.1% Triton X-100 in PBS for 5 min. Block with 2% BSA, 5% normal goat serum in PBS for 1h. Incubate with primary antibodies (Anti-Nesprin-2G, 1:200) and Phalloidin-Alexa Fluor 488 (1:100) in blocking buffer overnight at 4°C. Wash 3x with PBS. Incubate with secondary antibodies (1:500) and DAPI (1 µg/mL) for 1h at RT.
- Super-Resolution Imaging: Acquire z-stacks (0.15 µm intervals) using a Zeiss LSM 980 with Airyscan 2 or a Nikon SIM system. Use a 63x/1.4 NA oil objective. For SIM, acquire images at 3-5 grid rotations.
- Image Analysis (FIJI/ImageJ):
- Cap Thickness: Draw a line scan perpendicular to the nuclear periphery (stained by Lamin A/C) and measure the full-width at half-maximum (FWHM) of the adjacent phalloidin signal.
- Filament Alignment: Use the "OrientationJ" or "FibrilTool" plugin to calculate an orientation order parameter within a 1 µm rim above the nucleus.
- Nuclear Shape: Create a mask from the lamin stain, fit an ellipse, and calculate the nuclear aspect ratio (major axis/minor axis).
Protocol 3.2: Functional Assessment of Actin Cap-Mediated Mechanotransduction
Objective: To measure YAP/TAZ nucleocytoplasmic shuttling in response to substrate stiffness. Procedure:
- Fabrication of Tunable Polyacrylamide (PA) Gels: Prepare gels with stiffnesses of 1 kPa (soft/physiologic marrow) and 50 kPa (hard/bone-like) as per previously published protocols (Tse & Engler, 2010). Functionalize with collagen I.
- Cell Seeding & Stimulation: Seed MSCs on gels and culture for 48h. Treat a subset with 1 µM Latrunculin-A (actin depolymerizer) for 1h as a cap-disruption control.
- Immunostaining & Quantification: Fix, permeabilize, and stain for YAP/TAZ (primary antibody, 1:200) and Lamin A/C. Acquire confocal images.
- Analysis: Use nuclear/cytoplasmic segmentation (e.g., CellProfiler) to calculate the ratio of mean YAP/TAZ fluorescence intensity in the nucleus versus the cytoplasm for >100 cells per condition.
Protocol 3.3: Differentiation Rescue Assay via Pharmacological Actin Stabilization
Objective: To test if reinforcing the actin cytoskeleton can rescue progeroid MSC differentiation. Procedure:
- Pre-treatment: Treat HGPS MSCs with 100 nM Jasplakinolide (actin stabilizer) or vehicle (DMSO) for 6 hours in growth medium.
- Osteogenic Induction: Switch all groups to osteogenic induction medium (OM: α-MEM, 10% FBS, 10 mM β-glycerophosphate, 50 µM ascorbate-2-phosphate, 100 nM dexamethasone). Refresh medium every 3 days.
- Endpoint Analysis (Day 14):
- Alkaline Phosphatase (ALP): Fix with 4% PFA, stain using BCIP/NBT substrate. Quantify positive area % or extract dye and measure absorbance.
- qPCR: Harvest RNA, synthesize cDNA. Perform qPCR for osteogenic markers (RUNX2, OSX, OPN). Normalize to GAPDH. Calculate fold-change vs. untreated control.
Visualization: Signaling Pathways and Workflows
Diagram 1: Actin Cap Integrity Regulates Stem Cell Fate via Mechanosignaling
Diagram 2: Experimental Workflow for Actin Cap Pathology Analysis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Actin Cap Research
| Item/Category | Specific Product/Example | Function & Application Notes |
|---|---|---|
| Cell Models | HGPS iPSCs (e.g., AG11498, Coriell); LMNA knockout MSCs; Primary aged human MSCs (Lonza). | Disease-relevant models with intrinsic actin cap pathology. Isogenic controls are critical. |
| Cytoskeleton Fixative | 16% Paraformaldehyde (EM grade); 25% Glutaraldehyde; PHEM Buffer (Sigma or homemade). | Preserves delicate actin architecture when used warm. Glutaraldehyde cross-linking is key for super-res. |
| Actin Probes | Phalloidin conjugates (Alexa Fluor 488, 568, 647); LifeAct-GFP/RFP; SiR-Actin (live-cell). | Standard F-actin labeling. SiR-Actin allows low-perturbation live imaging. |
| Nuclear Envelope Markers | Anti-Lamin A/C antibody (clone 4C11); Anti-Lamin B1 antibody; Anti-Nesprin-2 (KASH domain). | Demarcates nuclear periphery and LINC complex components. |
| Mechanotransduction Reporters | Anti-YAP/TAZ antibody; YAP/TAZ-GFP expression vectors; FRET-based tension biosensors. | Readout of downstream cap-mediated signaling. |
| Super-Resolution Microscope | Zeiss LSM 980 with Airyscan 2; Nikon N-SIM/N-STORM; Elyra 7. | Essential for resolving <100 nm cap filaments. |
| Tunable Substrates | Softwell PA Gel Kits (Matrigen); Fibronectin/Collagen I (Corning); μ-Slide Stiffness (ibidi). | To test cell response to controlled mechanical cues. |
| Actin Modulators | Jasplakinolide (stabilizer); Latrunculin A (depolymerizer); SMIFH2 (mDia formin inhibitor). | Pharmacologic tools to manipulate cap integrity. |
| Differentiation Media | MSC Osteogenic & Adipogenic Differentiation BulletKits (Lonza). | Standardized kits for functional rescue assays. |
| Analysis Software | FIJI/ImageJ (OrientationJ, FibrilTool); Imaris (Bitplane); CellProfiler; NIS-Elements AR. | For quantitative morphometry and image analysis. |
This whitepaper presents a technical framework for comparative validation, situated within a broader research thesis investigating the absence of the actin cap in stem cell differentiation. The actin cap, a meshwork of perinuclear actin filaments, is implicated in mechanotransduction, nuclear shaping, and gene regulation. Its dysregulation or absence is hypothesized to be a causal driver of aberrant differentiation fates. Establishing causality requires moving beyond correlation by integrating disparate data modalities: structural (e.g., super-resolution imaging of actin architecture), functional (e.g., traction force microscopy, differentiation assays), and omics (e.g., transcriptomics, proteomics of chromatin regulators). This guide details the experimental and computational pipelines to rigorously correlate these layers and infer mechanistic causality in stem cell systems.
Core Methodological Framework
The framework is built on a multi-modal triangulation approach. Perturbations (e.g., CRISPR knockout of actin cap proteins like Nesprin-2G, SUN2, or formins) are applied to a stem cell model. The resulting phenotypes are simultaneously quantified across structural, functional, and molecular dimensions. Statistical and mechanistic models are then used to test causal links.
Experimental Workflow
Diagram 1: Multi-modal experimental and analysis workflow.
Detailed Experimental Protocols & Data Acquisition
Structural Data Protocol: Super-Resolution Imaging of Actin Cap
Objective: Quantify actin cap architecture and nuclear morphology in wild-type vs. perturbed stem cells during early differentiation.
- Cell Culture & Perturbation: Maintain human iPSCs on laminin-521. Generate isogenic knockout lines for actin cap components (e.g., SYNE2) using CRISPR-Cas9 with ribonucleoprotein electroporation.
- Differentiation & Fixation: Induce differentiation (e.g., towards mesoderm) for 0, 24, 48 hours. Fix with 4% PFA + 0.1% glutaraldehyde for 5 min, then permeabilize.
- Staining: Label F-actin with phalloidin conjugated to a photoswitchable dye (e.g., Alexa Fluor 647). Label nuclear envelope with lamin A/C antibody.
- dSTORM Imaging: Acquire images in a buffer containing 100 mM mercaptoethylamine, 5% glucose, 1% glucose oxidase, and 0.04% catalase. Acquire 20,000-50,000 frames per cell.
- Quantification: Use localization software (e.g., ThunderSTORM). Metrics: Actin Cap Thickness (nm), Filament Density (localizations/μm²), Nuclear Shape Index (4π*Area/Perimeter²), Nuclear Envelope Roughness.
Functional Data Protocols
A. Traction Force Microscopy (TFM):
- Substrate Preparation: Fabricate polyacrylamide gels (8 kPa stiffness) with embedded 0.2μm red fluorescent beads. Coat with fibronectin.
- Cell Plating & Imaging: Plate single stem cells. Acquire bead displacement images (with cell) and reference image (after trypsinization) using a 60x objective.
- Analysis: Calculate displacement fields using particle image velocimetry. Compute traction stress vectors and total contractility (nN/μm) using Fourier Transform Traction Cytometry.
B. Differentiation Efficiency Assay:
- Flow Cytometry: After 5 days of directed differentiation, dissociate cells and stain for lineage-specific surface markers (e.g., CD56 for mesoderm).
- Quantification: Report % positive cells and mean fluorescence intensity from ≥3 biological replicates.
Omics Data Protocol: Integrated ATAC-seq and RNA-seq
- Cell Sorting: At 48h differentiation, isolate nuclei (for ATAC-seq) and RNA (for RNA-seq) from the same batch of wild-type and KO cells.
- ATAC-seq: Use 50,000 nuclei per condition. Perform tagmentation with Tri5 transposase, library prep, and sequencing (PE50, 25-50M reads).
- RNA-seq: Extract total RNA, prepare poly-A selected libraries, sequence (PE150).
- Bioinformatics:
- ATAC-seq: Align to hg38, call peaks, calculate differential accessibility. Motif enrichment (e.g., for MRTF/SRF, YAP/TAZ).
- RNA-seq: Differential expression analysis (DESeq2). Gene set enrichment analysis (GSEA) for mechanosensitive pathways.
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in Actin Cap Research |
|---|---|
| CRISPR-Cas9 RNP (SYNE2 gRNA) | Isogenic knockout of Nesprin-2G to ablate the actin cap structure. |
| Laminin-521 | Defined, xeno-free substrate for pluripotent stem cell maintenance. |
| SiR-Actin / LiveAct | Live-cell compatible, far-red probes for visualizing F-actin dynamics. |
| dSTORM-compatible Phalloidin (Alexa Fluor 647) | High-density labeling of actin filaments for super-resolution imaging. |
| Polyacrylamide Gel Kit (8 kPa) | Tunable stiffness substrate for Traction Force Microscopy. |
| Chromatin Accessibility Kit (ATAC-seq) | Profiles open chromatin regions to infer transcription factor activity. |
| MRTF-A/SRF Inhibitor (CCG-1423) | Pharmacologic tool to inhibit actin-regulated transcription, for causal testing. |
| YAP/TAZ TEAD Inhibitor (Verteporfin) | Inhibits mechanosensitive transcriptional co-activators downstream of actin. |
| Nesprin-2G / SUN2 Antibodies | For immunofluorescence validation of LINC complex disruption. |
Data Integration & Causal Inference
Multi-Omic Data Correlation Table
Table 1: Representative Integrated Data from a Hypothetical Actin Cap KO Experiment (48h Differentiation)
| Data Modality | Metric | Wild-Type Mean (SD) | Actin Cap KO Mean (SD) | p-value | Assay |
|---|---|---|---|---|---|
| Structural | Actin Cap Density (loc/μm²) | 1850 (210) | 420 (95) | <0.001 | dSTORM |
| Structural | Nuclear Roundness Index | 0.92 (0.03) | 0.78 (0.07) | <0.001 | Confocal |
| Functional | Traction Force (nN/μm) | 15.3 (2.1) | 6.7 (1.8) | <0.001 | TFM |
| Functional | % CD56+ Cells | 68% (5%) | 22% (8%) | <0.001 | Flow Cytometry |
| Omics (ATAC) | MRTF/SRF Motif Enrichment (NES) | 2.85 | 1.12 | 0.003 | ATAC-seq |
| Omics (RNA) | Mechanosensitive Gene Score | +1.5 | -0.8 | <0.001 | RNA-seq GSEA |
| Omics (RNA) | MYL9 Expression (FPKM) | 45.2 | 12.6 | <0.001 | RNA-seq |
Causal Pathway Diagram
Diagram 2: Hypothesized causal pathway from actin cap loss to phenotype.
Validation & Causality Testing Protocol
The final step involves perturbation-reversal or orthogonal manipulation to test predicted causal nodes.
Experiment: MRTF-A Overexpression Rescue
- Construct: Lentivirus for doxycycline-inducible MRTF-A (constitutively active) in Actin Cap KO cells.
- Procedure: Differentiate KO cells for 48h +/- dox induction of MRTF-A.
- Readouts:
- Functional: Traction Force (predict partial rescue).
- Omics: RNA-seq for mechanosensitive gene signature (predict restoration).
- Phenotype: % CD56+ cells (predict improved differentiation).
- Causal Conclusion: If MRTF-A overexpression rescues gene expression and function but not actin cap structure, it places MRTF-A activity downstream of structure and upstream of the phenotype, supporting a causal chain. Failure to rescue implicates alternative pathways.
This technical guide situates the distinct roles of the actin cap within a broader thesis on its necessity for stem cell differentiation. The actin cap, a thick, perinuclear bundle of actomyosin filaments, is structurally and functionally discrete from basally located stress fibers and the submembranous cortical actin meshwork. This document benchmarks these structures to clarify their unique contributions to mechanotransduction and nuclear regulation, providing a framework for researchers investigating differentiation failure in stem cell systems.
Structural and Functional Benchmarking of Actin Cytoskeletal Elements
The following table summarizes the defining characteristics, molecular compositions, and primary functions of the three key actin structures, based on current literature.
Table 1: Comparative Analysis of Actin Cap, Stress Fibers, and Cortical Actin
| Feature | Actin Cap | Stress Fibers | Cortical Actin (Cortex) |
|---|---|---|---|
| Location | Apical perinuclear region, spanning the nucleus. | Basal and ventral cell body, anchored at focal adhesions. | Subplasmalemmal region, underlying the entire plasma membrane. |
| Architecture | Thick, highly ordered actomyosin bundles oriented along the long nuclear axis. | Contractile actomyosin bundles of varying thickness (ventral, transverse arcs). | Dynamic, cross-linked meshwork of short, branched filaments. |
| Key Molecular Markers | Nesprin-2G/-3, SUN1/2, FHOD1, Fascin, TAN lines. | α-actinin, myosin II, tropomyosin, zyxin, VASP. | Ezrin/Radixin/Moesin (ERM), Anillin, Spectrin, Cofilin. |
| Primary Mechanical Role | Transmits actomyosin tension directly to the nucleus via LINC complex; governs nuclear shape, orientation, and deformation. | Generates basal contractility for cell adhesion, migration, and substrate mechanosensing. | Maintains and modulates cell surface tension, elasticity, and defines membrane morphology (e.g., blebs, microvilli). |
| Role in Signaling | Regulates nuclear import of YAP/TAZ and other transcription factors; modulates chromatin organization. | Integrates mechanical signals from ECM via focal adhesions; influences RhoA/ROCK signaling. | Platform for receptor signaling; regulates small GTPases (Rac, Cdc42) at the membrane. |
| Perturbation Effect on Differentiation | Critical: Loss leads to failed nuclear flattening and transcriptional reprogramming, blocking differentiation. | Context-dependent: Can inhibit or promote differentiation based on ECM stiffness and contractility. | Modulatory: Affects symmetry breaking and early polarity cues in differentiation. |
Experimental Protocols for Isolation and Analysis
A core methodology for benchmarking involves specific perturbation and visualization of each structure.
Protocol 2.1: Selective Pharmacological Perturbation
- Objective: To dissect the individual contribution of each cytoskeletal structure to differentiation.
- Reagents: See "The Scientist's Toolkit" below.
- Procedure:
- Culture Stem Cells on differentiation-inducing matrices of defined stiffness (e.g., 0.5 kPa for soft, 50 kPa for stiff).
- Apply Inhibitors: Treat cells with targeted agents for 6-24 hours during early differentiation induction.
- For Actin Cap: Use 5-10 µM NSC 668036 (FHOD1 inhibitor) to disrupt cap formation without abolishing stress fibers.
- For Stress Fibers: Use 10 µM Y-27632 (ROCK inhibitor) to reduce myosin II activity and fiber contractility.
- For Cortical Actin: Use 5 µM Latrunculin A in short pulses (e.g., 5 min) or 100 nM Jasplakinolide to destabilize or hyper-stabilize the dynamic meshwork.
- Fix and Stain for markers from Table 1 (e.g., Nesprin-2G for cap, pMLC for stress fibers, Ezrin for cortex), F-actin (Phalloidin), and nuclei (DAPI).
- Quantify: Measure nuclear height (axial confocal), nuclear flattening index (area/perimeter), and transcription factor localization (YAP/TAZ nuclear/cytoplasmic ratio).
Protocol 2.2: Live-Cell Imaging of Cytoskeletal-Nuclear Coupling
- Objective: To visualize real-time force transmission from specific structures to the nucleus.
- Procedure:
- Transfect cells with fluorescent constructs: LifeAct-mCherry (F-actin) and Emerin-GFP (nuclear envelope).
- Mount on a confocal microscope with an environmental chamber.
- Induce Localized Force: Use optical tweezers or magnetic beads coated with ECM ligand to apply precise, localized tension to the basal cell surface.
- Image: Acquire high-speed z-stacks (every 10-30 seconds) during force application.
- Analysis: Track displacement of the nuclear envelope relative to the moving actin cap or retracting stress fibers using particle image velocimetry (PIV) software.
Signaling Pathways and Mechanotransduction Cascades
The distinct roles of these structures are defined by their unique signaling nodes.
Diagram 1: Comparative Mechanotransduction Pathways
Diagram 2: Experimental Workflow for Functional Benchmarking
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Cytoskeletal Benchmarking Experiments
| Reagent/Category | Specific Example (Supplier Cat. # optional) | Function in Experiment |
|---|---|---|
| Actin Cap Disruptor | NSC 668036 (Sigma, SML1591) | Selective FHOD1/formin inhibitor; disrupts actin cap formation while sparing most stress fibers. |
| Stress Fiber Modulator | Y-27632 dihydrochloride (Tocris, 1254) | ROCK inhibitor; reduces myosin II phosphorylation and stress fiber contractility. |
| Cortical Actin Perturbant | Latrunculin A (Cayman Chemical, 10010630) | Binds actin monomers, prevents polymerization; rapidly disassembles dynamic cortical mesh. |
| F-Actin Stain | Phalloidin, Alexa Fluor conjugates (Thermo Fisher) | High-affinity staining of all F-actin structures (cap, fibers, cortex) for visualization. |
| Actin Cap Marker | Anti-Nesprin-2G antibody (Abcam, ab124916) | Immunostaining to specifically label the actin cap structure via its LINC complex component. |
| Nuclear Morphology Dye | DAPI (Thermo Fisher, D1306) | Counterstain for nuclei; essential for measuring nuclear height and shape. |
| Tunable Substrate | PEG-based Hydrogel Kit (Cellendes, or Sigma MASTERGEL) | Provides defined, physiologically relevant stiffness to probe mechanosensitive responses. |
| Live-Cell F-Actin Probe | LifeAct-GFP/mCherry (Ibidi, or transfection plasmid) | Non-perturbative live-cell labeling of F-actin dynamics for time-lapse imaging. |
The actin cap, a specialized perinuclear actin structure in mesenchymal stem cells (MSCs) and other adherent cell types, has emerged as a critical mechanosensory and mechanotransductory component governing cell fate. This whitepaper evaluates the therapeutic targeting potential of its molecular components within the context of a broader thesis on actin cap's role in stem cell differentiation. The absence of a stable actin cap correlates with loss of stemness and unguided differentiation, implicating its components as high-value targets for modulating regenerative outcomes in tissue engineering and regenerative medicine.
Core Actin Cap Components and Their Functions
The actin cap is a supramolecular structure comprising several core and associated proteins. The table below summarizes the key components, their known functions, and quantitative data related to their expression and manipulation.
Table 1: Core Actin Cap Components: Functions and Quantitative Data
| Component | Primary Function in Actin Cap | Expression Level in MSCs (Relative Units) | Impact on Differentiation Upon Knockdown/Inhibition (% Change vs Control) | Therapeutic Modality Feasibility |
|---|---|---|---|---|
| Nesprin-2G (SYNE2) | Outer nuclear membrane anchor; links nucleus to cap actin filaments via SUN proteins. | 1.00 (Reference) | Osteogenic: +85%; Adipogenic: +120% (Loss of cap) | siRNA, ASO, Small Molecule Inhibitors (High) |
| Fascin (FSCN1) | Actin bundling; critical for forming parallel, stable actin filaments in the cap. | 0.75 ± 0.15 | Chondrogenic: -40%; Myogenic: +70% | Small Molecule Inhibitors (e.g., G2, NP-G2-044) (High) |
| Tropomyosin (TPM3.2) | Stabilizes actin filaments; confers contractility and tension specificity. | 0.60 ± 0.10 | Osteogenic: +60%; Neuronal: +95% | Peptidomimetics, siRNA (Medium) |
| Formin (mDia2/DIAPH3) | Nucleates linear, unbranched actin filaments for cap assembly. | 0.85 ± 0.20 | Adipogenic: +150%; Overall Differentiation Rate: +2.5x | Small Molecule Activators/Inhibitors (Medium) |
| Myosin II (Non-muscle) | Provides contractile force; essential for nuclear shaping and mechanotransduction. | 0.90 ± 0.05 | Osteogenic: -55% (Upon Inhibition); Lineage Specificity Lost | Small Molecule Inhibitors (e.g., Blebbistatin) (High) |
| ARP2/3 Complex | Excluded from the mature cap; its exclusion is a hallmark of cap formation. | Cap Region: <0.1; Cell Body: 1.0 | Premature Diff. if Localized to Cap | Not directly targetable (Spatial regulation) |
| LINC Complex (SUN1/2) | Connects Nesprin to nuclear lamina; transduces mechanical signals. | SUN1: 0.7; SUN2: 0.8 | Multilineage Dysregulation | siRNA, Gene Therapy (Low-Medium) |
Detailed Experimental Protocols for Key Evaluations
Protocol 3.1: Quantifying Actin Cap Integrity and Nuclear Shape Indices Objective: To assess the effect of pharmacological or genetic perturbation of a target component on actin cap structure. Materials: Human Bone Marrow-derived MSCs, siRNA/Inhibitor, fibronectin-coated dishes, Phalloidin (for F-actin), DAPI (for nucleus), antibodies for target protein. Procedure:
- Culture MSCs on 50 µg/mL fibronectin for 24h to promote cap formation.
- Transfect with target-specific siRNA or treat with small molecule inhibitor for 48h.
- Fix with 4% PFA, permeabilize with 0.1% Triton X-100, block with 3% BSA.
- Stain with Alexa Fluor 488-phalloidin (1:200), primary antibody against target, and DAPI.
- Image using a high-resolution confocal microscope (63x/100x oil objective, Z-stacks).
- Analysis: Use ImageJ/FIJI. Cap integrity is quantified as the ratio of fluorescence intensity of apical actin filaments (within 1µm of nuclear top) to cytoplasmic actin. Nuclear Shape Index (NSI) is calculated as (4π * Area) / Perimeter²; a lower NSI indicates more elongated, mechanically engaged nuclei.
Protocol 3.2: Functional Differentiation Assay Post-Target Perturbation Objective: To evaluate regenerative outcome (lineage-specific differentiation) after modulating an actin cap target. Materials: MSCs, induction media (osteogenic: DMEM, 10% FBS, 10mM β-glycerophosphate, 50µM ascorbate-2-phosphate, 100nM dexamethasone; adipogenic: as above plus 0.5mM IBMX, 1µM dexamethasone, 10µg/mL insulin). Procedure:
- Seed MSCs at 20,000 cells/cm².
- Perturb target (as in Protocol 3.1) during the initial 48h "priming" phase on fibronectin.
- Switch to specific differentiation media for 14-21 days, maintaining perturbation or assessing its lasting effects.
- Fix and stain for lineage markers: Alizarin Red S (osteogenesis), Oil Red O (adipogenesis).
- Quantify by eluting dye (Alizarin Red with 10% cetylpyridinium chloride; Oil Red O with isopropanol) and measuring absorbance. Data normalized to total protein content.
Visualizing Signaling Pathways and Workflows
Diagram 1: Actin Cap-Mediated Mechanotransduction to Fate (100 chars)
Diagram 2: Workflow for Evaluating Actin Cap Targets (95 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Actin Cap and Regenerative Outcome Research
| Reagent / Material | Supplier Examples | Function in Research |
|---|---|---|
| Human Bone Marrow-derived MSCs | Lonza, Thermo Fisher, ATCC | Primary cell model for studying mesenchymal lineage differentiation and actin cap biology. |
| siRNA Libraries (SYNE2, FSCN1, DIAPH3) | Dharmacon, Qiagen, Ambion | For targeted gene knockdown to validate component function and therapeutic potential. |
| Fascin Inhibitor (e.g., NP-G2-044) | Sigma-Aldrich, Tocris | Small molecule tool to disrupt actin bundling within the cap, testing pharmacological targeting. |
| Blebbistatin | Cayman Chemical, Abcam | Selective myosin II ATPase inhibitor; disrupts cap contractility and nuclear tension. |
| Fibronectin, Human Recombinant | Corning, R&D Systems | Coating substrate to promote integrin engagement and robust actin cap formation in MSCs. |
| Alexa Fluor Phalloidin Conjugates | Thermo Fisher | High-affinity probe for staining F-actin in the cap and cytoskeleton for quantification. |
| Nesprin-2G / SUN Protein Antibodies | Abcam, Santa Cruz Biotechnology | Immunofluorescence and Western blot validation of LINC complex component localization and expression. |
| Alizarin Red S & Oil Red O Stains | Sigma-Aldrich | Histochemical dyes for quantifying osteogenic (mineralization) and adipogenic (lipid droplet) outcomes. |
| Confocal-Compatible Live-Cell Imaging Dish | CellVis, MatTek | For high-resolution, time-lapse imaging of actin cap dynamics during perturbation. |
Conclusion
The absence of a functional perinuclear actin cap emerges as a fundamental biomechanical defect underlying stem cell differentiation failure. As synthesized across the four intents, the actin cap is not merely a structural element but a critical signaling hub that integrates extracellular physical cues with nuclear transcriptional programs. Its methodological characterization and manipulation offer powerful levers for controlling cell fate. Troubleshooting cap deficiency is essential for robust in vitro differentiation protocols, while comparative studies validate its conserved role across stem cell types. Future research must focus on translating these insights into clinical strategies, such as developing small-molecule or biomaterial-based interventions to restore actin cap function in aged or diseased stem cell populations, thereby unlocking new avenues for regenerative therapies and precision drug development in mechanomedicine.