From Structural Collapse to Malignant Transformation: Unraveling Actin Cap Disruption in Laminopathy-Driven Cancer Mechanisms
This article provides a comprehensive analysis of the emerging mechanistic link between laminopathies, actin cap disruption, and oncogenesis.
From Structural Collapse to Malignant Transformation: Unraveling Actin Cap Disruption in Laminopathy-Driven Cancer Mechanisms
Abstract
This article provides a comprehensive analysis of the emerging mechanistic link between laminopathies, actin cap disruption, and oncogenesis. Targeting researchers and drug development professionals, we explore the foundational biology of nuclear-cytoskeletal coupling via the LINC complex, detailing how lamin A/C mutations destabilize the perinuclear actin cap. We then examine cutting-edge methodologies for modeling and quantifying these disruptions in vitro and in vivo, followed by a troubleshooting guide for common experimental challenges. Finally, we validate these mechanisms by comparing them with other oncogenic pathways and evaluating potential therapeutic targets. This synthesis aims to bridge fundamental cell biology with translational oncology, highlighting novel intervention points for cancers with underlying nuclear envelope defects.
The Structural Link: How Lamin A/C Mutations Destabilize the Perinuclear Actin Cap and Prime the Cell for Transformation
Nuclear mechanotransduction is the process by which mechanical forces from the extracellular matrix (ECM) and cellular microenvironment are transmitted across the cytoskeleton to the nucleus, resulting in biochemical signals and changes in gene expression. This pathway is fundamental to cell differentiation, migration, and tissue homeostasis. Disruption of this force transmission apparatus is a central hypothesis in the pathology of laminopathies (e.g., Hutchinson-Gilford Progeria Syndrome, muscular dystrophies) and in cancer progression, where altered nuclear mechanics can promote metastasis and drug resistance. This whitepaper defines the three core structural players—Lamin A/C, the LINC complex, and the Actin Cap—that form the primary physical link for mechanotransduction.
Core Molecular Players & Quantitative Data
Table 1: Core Components of the Nuclear Mechanotransduction Pathway
| Component | Primary Molecular Constituents | Key Function | Quantitative Pertinence |
|---|---|---|---|
| Nuclear Lamina | Lamin A, Lamin C, B-type lamins | Provides structural scaffold for nucleus; determines nuclear stiffness and shape. | Lamin A/C expression correlates with nuclear stiffness (~0.1-10 kPa range). Progerin expression reduces stiffness by >50%. |
| LINC Complex | SUN1/2 (inner nuclear membrane), Nesprin-1/2/3/4 (outer nuclear membrane), Sad1/UNC-84 domain. | Spanning the nuclear envelope, connects cytoskeletal filaments to nuclear lamina. | Knockdown of SUN1/2 reduces force transmission efficiency by ~70-80% in magnetic bead twisting assays. |
| Actin Cap | Apical, perinuclear bundled F-actin, Myosin II, Formins (mDia1/2), Tropomyosin. | A stiff, contractile actin structure that applies direct, anisotropic stress to the nucleus via the LINC complex. | Cap actin bundles sustain forces of ~1-10 nN. Disruption (e.g., Latrunculin A) reduces nuclear deformation by >90% under substrate strain. |
Table 2: Pathological Correlations in Actin Cap Disruption, Laminopathies, and Cancer
| Condition/Model | Lamin A/C State | Actin Cap/LINC Integrity | Functional Consequence |
|---|---|---|---|
| Hutchinson-Gilford Progeria Syndrome | Accumulation of farnesylated Progerin (mutant LMNA). | Severely disrupted cap architecture; mislocalized Nesprins. | Impaired mechanosignaling, nuclear blebbing, premature senescence. |
| Emery-Dreifuss Muscular Dystrophy | Mutations in LMNA or EMD (emerin). | Weakened LINC complex linkage. | Reduced nuclear stability in muscle contraction, apoptosis. |
| Metastatic Cancer Cells | Often downregulated or mislocalized Lamin A/C. | Frequently absent or disorganized actin cap; altered Nesprin expression. | Increased nuclear deformability, enhanced migration through confined spaces, chemoresistance. |
| Cell Senescence | Increased Lamin A/C expression and nuclear stiffening. | Enhanced actin cap formation and stress fiber organization. | Resistance to deformation, altered YAP/TAZ signaling. |
Detailed Experimental Protocols
Protocol 1: Quantifying Nuclear Deformation and Actin Cap Integrity Objective: To measure real-time nuclear shape changes in response to substrate strain and correlate with actin cap structure.
- Cell Plating: Plate NIH/3T3 fibroblasts or MCF-10A epithelial cells on silicone elastomer membranes (Flexcell plates) coated with 10 µg/mL fibronectin.
- Transfection/Staining: Transfect with GFP-Nesprin-2G or stain fixed samples with:
- Phalloidin (Actin): 1:1000 in PBS for 1 hr.
- DAPI (Nucleus): 300 nM for 5 min.
- Anti-Lamin A/C Antibody: 1:500, overnight at 4°C.
- Mechanical Stimulation: Apply cyclic uniaxial strain (10% elongation, 0.5 Hz) using a Flexcell FX system. Maintain control stations without strain.
- Live/Endpoint Imaging: Use confocal microscopy (63x oil objective) to image cells at 0, 15, 30, and 60 mins post-strain onset. For fixed endpoints, permeabilize with 0.1% Triton X-100 for 10 min before staining.
- Analysis: Quantify nuclear aspect ratio (major/minor axis) using ImageJ. Score actin cap presence as apical, perinuclear F-actin bundles overlapping the nucleus in >50% of z-sections.
Protocol 2: FRET-based Tension Sensor Across the LINC Complex Objective: To directly measure mechanical tension across specific LINC complex components in live cells.
- Sensor Construct: Utilize a Nesprin-2 tension sensor module (TSMod) inserted between the spectrin repeats and the SUN-binding KASH domain.
- Cell Transfection: Transfect U2OS cells with Nesprin-2G-TSMod using Lipofectamine 3000.
- Mechanical Perturbation:
- Inhibition: Treat with 10 µM Y-27632 (ROCK inhibitor) or 2 µM Latrunculin B for 1 hr.
- Activation: Seed cells on 50 kPa vs. 1 kPa polyacrylamide hydrogels to modulate substrate stiffness.
- FRET Imaging: Acquire images using a confocal microscope with FRET capabilities. Calculate the FRET ratio (acceptor emission/donor emission) after background subtraction.
- Interpretation: A decreased FRET ratio indicates increased tension stretching the sensor module. Compare ratios between soft/stiff substrates or before/after drug treatment.
Protocol 3: Chromatin Immunoprecipitation (ChIP) for Mechano-Induced Transcription Objective: To assess changes in transcription factor binding to promoters of mechanosensitive genes upon actin cap disruption.
- Cell Treatment: Subject human mesenchymal stem cells (hMSCs) to cyclic tensile strain (10%, 1 Hz) for 24 hrs. Include a control group treated with 100 nM siRNAs against SUN1/2 to disrupt the LINC complex.
- Crosslinking & Lysis: Fix cells with 1% formaldehyde for 10 min. Quench with 125 mM glycine. Lyse cells and isolate nuclei.
- Chromatin Shearing: Sonicate chromatin to an average size of 200-500 bp.
- Immunoprecipitation: Incubate sheared chromatin overnight with an antibody against RNA Polymerase II or a mechanoresponsive factor (e.g., YAP/TAZ). Use IgG as a negative control.
- qPCR Analysis: Analyze purified DNA by qPCR using primers for promoters of known mechanoresponsive genes (CYR61, CTGF, ANKRD1). Enrichment is calculated as % input relative to static controls.
Visualization of Pathways and Workflows
Title: Nuclear Mechanotransduction Signaling Pathway
Title: Core Experimental Workflow for Nuclear Mechanobiology
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Investigating Nuclear Mechanotransduction
| Reagent / Tool | Category | Example Product/Identifier | Primary Function in Research |
|---|---|---|---|
| Flexcell Tension System | Hardware | FX-6000T Series | Applies precise, cyclic, or static strain to cells cultured on elastic membranes. |
| Polyacrylamide Hydrogels | Tunable Substrate | Soft, Medium, Stiff (1-50 kPa) kits | Presents defined mechanical stiffness to cells to probe stiffness-dependent responses. |
| Lamin A/C Antibody | Antibody | Abcam ab8984, Santa Cruz sc-7292 | Detects and localizes Lamin A/C by immunofluorescence or Western blot; assesses expression/mislocalization. |
| Phalloidin Conjugates | Actin Stain | Alexa Fluor 488/568/647 Phalloidin | Visualizes F-actin architecture, specifically the actin cap, with high specificity and signal. |
| SUN1/2 siRNA Pool | Gene Knockdown | ON-TARGETplus SMARTpool (Dharmacon) | Efficiently depletes LINC complex core components to disrupt nucleo-cytoskeletal coupling. |
| Y-27632 Dihydrochloride | Small Molecule Inhibitor | ROCK Inhibitor (ROCK1/2) | Inhibits actomyosin contractility, leading to actin cap dissolution and reduced nuclear tension. |
| Nesprin-2G Tension Sensor | Molecular Biosensor | Nesprin-2G-TSMod (Addgene) | FRET-based live-cell reporter for direct measurement of tension across the LINC complex. |
| Progerin cDNA Construct | Disease Model | pBABE-puro LMNA G608G (Addgene) | Induces expression of progerin to model Hutchinson-Gilford Progeria Syndrome in vitro. |
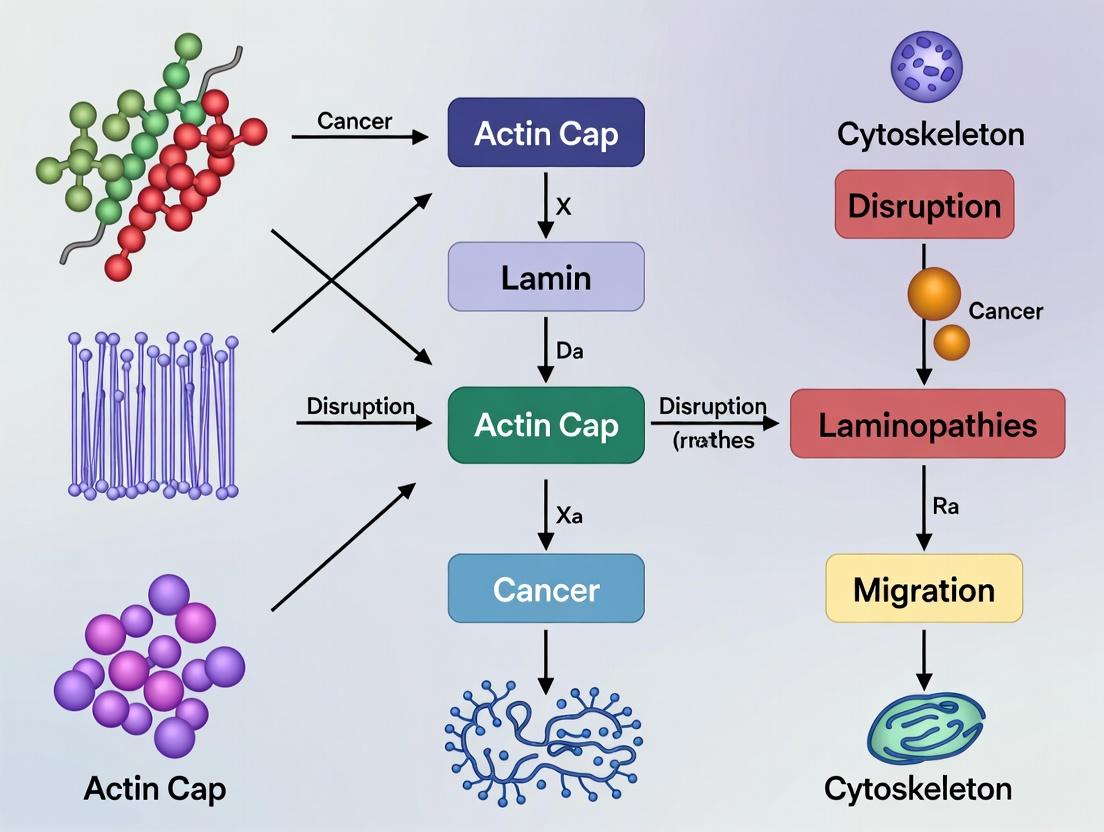
The integrated system of the actin cap, LINC complex, and Lamin A/C forms the definitive mechanical pathway from the cell surface to the genome. In the context of actin cap disruption laminopathies cancer mechanisms, research must focus on:
- Dynamic Mapping: Using advanced biosensors to visualize force propagation in real-time across this pathway in disease models.
- Chromatin-Specificity: Determining how specific nuclear deformations alter the epigenome and transcriptional programs differentially in cancer versus laminopathies.
- Therapeutic Targeting: Identifying small molecules or gene therapies that can specifically modulate the stiffness of this pathway—softening nuclei in fibrosis or stiffening them in metastatic cancer—to restore homeostatic mechanosignaling.
The precise definition and interrogation of these players provide a robust framework for developing novel mechano-based therapeutics.
1. Introduction and Thesis Context Disruption of the nuclear lamina, a meshwork of A- and B-type lamins, is a hallmark of laminopathies and is increasingly implicated in cancer progression. Within the broader thesis of actin cap disruption in laminopathies and cancer, this whitepaper investigates the precise molecular mechanisms by which laminopathy-associated mutations cause disassembly of the perinuclear actin cap—a critical structure that governs nuclear shape, mechanotransduction, and gene expression. We dissect two primary mechanistic paradigms: direct physical uncoupling of the nucleus from the cytoskeleton versus indirect signaling-mediated disassembly.
2. Quantitative Data Summary
Table 1: Key Quantitative Findings on Actin Cap Parameters in Laminopathy Models
| Parameter Measured | Wild-Type / Control | Lamin A/C Mutant (e.g., R482W) | Lamin B1 Depletion | Measurement Technique |
|---|---|---|---|---|
| Actin Cap Fiber Thickness | 1.0 ± 0.1 μm | 0.4 ± 0.15 μm | 0.7 ± 0.1 μm | Structured Illumination Microscopy (SIM) |
| Nuclear Height/Width Aspect Ratio | 0.65 ± 0.05 | 0.35 ± 0.08 | 0.45 ± 0.06 | Confocal 3D Reconstruction |
| Transmembrane Actin-associated Nuclear (TAN) Line Lifespan | > 60 min | < 15 min | ~ 30 min | Live-cell TIRF Microscopy |
| LINC Complex (Nesprin-2G/SUN2) Co-localization at Nuclear Envelope | High (Pearson's r > 0.8) | Very Low (Pearson's r ~ 0.2) | Moderate (Pearson's r ~ 0.5) | Super-Resolution Microscopy |
| Nuclear Stiffness (Young's Modulus) | ~4.5 kPa | ~1.8 kPa | ~2.9 kPa | Atomic Force Microscopy (AFM) |
| pERK/ERK Ratio (Signaling Readout) | 1.0 (basal) | 2.8 ± 0.4 | 1.5 ± 0.3 | Western Blot Quantification |
Table 2: Summary of Proposed Mechanisms and Supporting Evidence
| Proposed Mechanism | Primary Evidence | Key Mutations Exemplified | Therapeutic Implication |
|---|---|---|---|
| Direct Mechano-uncoupling: Mutant lamins disrupt LINC complex binding. | Loss of Nesprin-2G/SUN2 clustering; AFM shows mechanical decoupling. | R482W (FPLD), H222P (EDMD) | Stabilize LINC interactions. |
| Indirect Signaling Dysregulation: Altered MAPK/ERK or Rho/ROCK signaling. | Elevated pERK, rescued cap by MEK inhibition; Altered RhoA activity. | ΔK32 (Prelamin A accumulation), R133L (Heart disease) | Target upstream kinases (e.g., MEK, ROCK). |
| Chromatin-Based Alterations: Changed histone modifications affecting cap gene expression. | H3K9me3 mislocalization; altered expression of TPM1, TPM2. | G232E, R386K (Progeroid syndromes) | Epigenetic modulators (HDAC inhibitors). |
3. Experimental Protocols
Protocol 1: Quantifying Actin Cap Integrity via Structured Illumination Microscopy (SIM)
- Cell Culture & Transfection: Seed human dermal fibroblasts (WT and LMNA mutant) on fibronectin-coated #1.5 coverslips. Transfect with LifeAct-GFP or stain endogenous F-actin.
- Fixation & Staining: At 80% confluency, fix with 4% PFA for 15 min, permeabilize with 0.1% Triton X-100, and block with 3% BSA. Stain F-actin with Phalloidin-Alexa Fluor 568 (1:200). Counterstain nuclei with DAPI.
- SIM Imaging: Acquire z-stacks (0.15 μm steps) on a Nikon N-SIM or equivalent system using a 100x oil-immersion TIRF objective (NA 1.49). Use 561 nm laser for phalloidin.
- Image Reconstruction & Analysis: Reconstruct raw SIM images using manufacturer's software. Use FIJI/ImageJ to create maximum intensity projections. Measure actin fiber thickness and density in the perinuclear region using the "Analyze Particles" and "Line Scan" tools. Quantify the percentage of cells with a clearly defined, dorsally located actin cap.
Protocol 2: Assessing LINC Complex Tethering by Proximity Ligation Assay (PLA)
- Sample Preparation: Culture cells on coverslips as above. Fix with ice-cold methanol for 10 min at -20°C for better antigen preservation of nuclear envelope proteins.
- PLA Procedure: Perform Duolink PLA according to manufacturer's protocol. Use primary antibodies: mouse anti-Nesprin-2G (K20) and rabbit anti-SUN2. Incubate with PLA PLUS and MINUS probes.
- Ligation & Amplification: Perform ligation (30 min at 37°C) and amplification (100 min at 37°C) using a fluorescent detection kit (Orange, 594 nm).
- Imaging & Quantification: Acquire confocal images at 63x. Count PLA puncta (representing <40 nm proximity) at the nuclear periphery using an automated spot detection algorithm (e.g., in FIJI). Normalize puncta count to nuclear perimeter.
Protocol 3: Nuclear Mechanophenotyping by Atomic Force Microscopy (AFM)
- AFM Probe Preparation: Use silicon nitride cantilevers with a 5 μm spherical silica tip. Calibrate spring constant (k ≈ 0.1 N/m) via thermal tuning.
- Sample Preparation: Plate cells at low density on 35 mm Petri dishes. Perform experiments in live-cell imaging medium at 37°C/5% CO2.
- Force Mapping: Using a Bruker Bio-AFM or similar, perform a 10x10 force map over the center of the nucleus. Apply a maximum force of 2 nN at 1 Hz approach/retract rate.
- Data Analysis: Fit the retract curve of each force-indentation curve to the Hertz model for a spherical indenter to calculate the apparent Young's modulus (stiffness). Average values per nucleus and across cell populations.
4. Signaling Pathways and Logical Relationships
Diagram 1: Mechanisms of Actin Cap Disassembly in Laminopathies
5. The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Materials
| Reagent / Material | Supplier Examples | Function in Research |
|---|---|---|
| LMNA Mutant Fibroblasts | Coriell Institute, ATCC | Disease-relevant cellular models for mechanistic studies. |
| LifeAct-GFP/RFP BacMam 2.0 | Cytoskeleton, Inc. | Live-cell visualization of F-actin dynamics with low cytotoxicity. |
| Duolink PLA Kit | Sigma-Aldrich | Detects protein-protein interactions (<40 nm) at LINC complex. |
| Phalloidin Conjugates (e.g., Alexa Fluor 568) | Thermo Fisher Scientific | High-affinity staining of filamentous actin for fixed-cell imaging. |
| MEK Inhibitor (Selumetinib) | Selleckchem | Tool to inhibit MAPK/ERK signaling and test indirect mechanisms. |
| ROCK Inhibitor (Y-27632) | Tocris Bioscience | Inhibits ROCK kinase to probe Rho pathway role in cap regulation. |
| Lamin A/C siRNA Pool | Dharmacon | For knock-down studies to model lamin loss-of-function. |
| Nesprin-2G GFP Expression Plasmid | Addgene | Overexpression to test rescue of LINC complex tethering (plasmid #125552). |
| Bioactive G-LISA RhoA Activation Assay | Cytoskeleton, Inc. | Quantifies GTP-bound RhoA levels to assess pathway activity. |
| Nuclear Isolation Kit | Cell Biolabs, Inc. | Isolates clean nuclei for biochemical analysis of lamina components. |
1. Introduction & Thesis Context
Within the broader mechanistic research into laminopathies and oncogenesis, the disruption of the perinuclear actin cap (a dense, contractile F-actin meshwork anchored to the nuclear envelope via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes) represents a critical point of convergence. This whitepaper details the causal pathway from actin cap destabilization to nuclear envelope failure, subsequent DNA damage, and the establishment of genomic instability—a hallmark of cancer and a feature of many lamin-associated diseases.
2. Core Mechanisms and Quantitative Data
The collapse of the actin cap exerts direct mechanical stress on the nuclear envelope. This stress is transduced via nesprin-2G/ SUN protein LINC complexes, leading to the aberrant recruitment and activation of mechanosensitive effectors.
Table 1: Consequences of Actin Cap Disruption on Nuclear Integrity
| Parameter Measured | Control (Wild-Type/Stable Cap) | Condition (Cap Disrupted/Collapsed) | Experimental Method |
|---|---|---|---|
| Nuclear Height/Shape | Tall, elongated morphology | Flattened, rounded morphology | Confocal microscopy + 3D reconstruction |
| Nuclear Envelope Rupture Frequency | <5% of cells over 24h | 35-60% of cells over 24h | Live-cell imaging with cGAS-GFP or NLS-mCherry leakage assay |
| Cytosolic DNA Fragment Presence | Low baseline (≤100 AU) | High accumulation (≥1000 AU) | ELISA for cytosolic dsDNA or cGAS activity assay |
| Persistent 53BP1 Nuclear Foci (>6h) | 0-2 foci/nucleus | 8-15 foci/nucleus | Immunofluorescence, automated quantification |
| Chromothripsis-like Rearrangements | Rare (<0.5% of clones) | Frequent (8-12% of clones) | Karyotyping & whole-genome sequencing |
3. Detailed Experimental Protocols
Protocol 3.1: Induction and Validation of Actin Cap Collapse
- Method: siRNA-mediated knockdown of CapG or TOCA-1, or pharmacological inhibition using Latrunculin A (low-dose, 100 nM).
- Procedure:
- Plate NIH/3T3 or U2OS cells on fibronectin-coated (2 µg/cm²) glass-bottom dishes.
- At 60% confluency, transfect with 25 nM siRNA targeting CapG or scramble control using a lipid-based transfection reagent.
- Incubate for 48-72 hours.
- Fix with 4% PFA for 15 min, permeabilize with 0.2% Triton X-100, and stain with Phalloidin-Alexa Fluor 488 (1:1000) and anti-Nesprin-2G antibody (1:500).
- Image using a confocal microscope with a 63x oil objective. Z-stacks (0.5 µm slices) are required. Cap collapse is defined by the loss of dorsal, perinuclear F-actin fibers and concomitant dispersion of nesprin-2G puncta.
Protocol 3.2: Live-Cell Imaging of Nuclear Rupture
- Method: Co-expression of a nuclear localization signal (NLS)-tagged fluorescent protein (NLS-mCherry, 3xNLS-GFP) and a cytosolic marker.
- Procedure:
- Generate stable cell line expressing NLS-mCherry or transfect 24h prior to imaging.
- Mount dish on a live-cell imaging system (37°C, 5% CO₂).
- Acquire images every 10 minutes for 24-48 hours using a 40x objective.
- Rupture events are identified by the rapid diffusion of NLS-mCherry from the nucleus into the cytoplasm, quantified as a >50% decrease in nuclear-to-cytoplasmic fluorescence ratio.
Protocol 3.3: Quantification of DNA Damage Response Activation
- Method: Immunofluorescence for 53BP1 or γH2AX foci combined with lamin B1 staining to identify ruptured nuclei.
- Procedure:
- After experimental treatment, fix and permeabilize cells as in 3.1.
- Block with 5% BSA for 1 hour.
- Incubate with primary antibodies: mouse anti-lamin B1 (1:1000) and rabbit anti-53BP1 (1:800) overnight at 4°C.
- Incubate with species-appropriate secondary antibodies (Alexa Fluor 488, 568) for 1 hour at RT.
- Image 50-100 nuclei per condition. Score nuclei with lamin B1 loss/gaps as "ruptured." Count 53BP1 foci in both intact and ruptured nuclei populations.
4. Signaling Pathway and Experimental Workflow Diagrams
Title: Mechanistic Pathway from Cap Collapse to Genomic Instability
Title: Integrated Experimental Workflow for Cap Collapse Studies
5. The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents and Materials
| Reagent/Material | Supplier Examples | Function in Research |
|---|---|---|
| siRNA Pools (CapG, TOCA-1, Nesprin-2G) | Dharmacon, Qiagen | Targeted knockdown of actin cap and LINC complex components to induce collapse. |
| Latrunculin A (Low-Dose) | Cayman Chemical, Tocris | Actin polymerization inhibitor; used at low dose (100-250 nM) to specifically disrupt cap fibers. |
| Fibronectin, Human Plasma | Corning, MilliporeSigma | Coating substrate to promote robust actin cap formation in adherent cells. |
| Anti-Nesprin-2G Antibody | Abcam, Santa Cruz Biotechnology | Validates LINC complex localization and integrity via immunofluorescence. |
| Phalloidin Conjugates (Alexa Fluor 488/568) | Thermo Fisher, Cytoskeleton | High-affinity F-actin stain for visualizing actin cap architecture. |
| cGAS-GFP / NLS-mCherry Reporters | Addgene (plasmids) | Live-cell reporters for nuclear rupture (leakage) and cytosolic DNA sensing. |
| Anti-Lamin B1 Antibody | Proteintech, Abcam | Marks nuclear envelope; loss/gaps indicate rupture events in fixed samples. |
| Anti-53BP1 / Anti-γH2AX Antibodies | Cell Signaling, Novus | Gold-standard markers for DNA double-strand breaks and damage response foci. |
| Nuclear Dye (Hoechst 33342 or DAPI) | Thermo Fisher | Nuclear counterstain for segmentation and focus colocalization analysis. |
| Long-Read Sequencing Service (PacBio, Nanopore) | PacBio, Oxford Nanopore | Detects large-scale genomic rearrangements and structural variants post-rupture. |
The transition from a normal to a precancerous state involves a complex rewiring of cellular architecture and signaling. Within the broader thesis of actin cap disruption and laminopathy-mediated oncogenesis, the shift from a stiff to a soft cellular mechanophenotype emerges as a critical, quantifiable early indicator. The actin cap, a perinuclear bundle of actomyosin stress fibers anchored to the nucleus via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes, is a primary determinant of nuclear morphology and cellular stiffness. Mutations in nuclear envelope proteins, notably lamins (laminopathies), disrupt this anchoring, leading to actin cap disassembly, nuclear envelope blebbing, and aberrant mechanotransduction. This breakdown compromises the cell's ability to sense and respond to extracellular matrix cues, promoting a soft phenotype that favors uncontrolled proliferation, evasion of apoptosis, and early invasion. This whitepaper details the technical assessment of this mechanophenotypic shift as a precancerous biomarker.
Quantitative Data: Hallmarks of the Precancerous Softening
Table 1: Comparative Mechanophenotypic Metrics in Normal vs. Precancerous Cells
| Metric | Normal Cell (Typical Range) | Precancerous Cell (Typical Range) | Measurement Technique | Key Implication |
|---|---|---|---|---|
| Young's Modulus (Stiffness) | 2 - 5 kPa | 0.5 - 1.5 kPa | Atomic Force Microscopy (AFM) | ~60-75% reduction in cell stiffness. |
| Actin Cap Integrity Score | 85 - 100% | 20 - 50% | Fluorescence Microscopy / F-actin Morphometry | Severe disruption of perinuclear actin architecture. |
| Nuclear Area / Circularity | Low Area, High Circularity | Increased Area, Low Circularity | Image Analysis (DAPI staining) | Nuclear deformation and blebbing evident. |
| Traction Force (per cell) | 50 - 200 nN | 10 - 50 nN | Traction Force Microscopy (TFM) | Reduced force generation on substrate. |
| Lamin A/C Expression Level | 1.0 (relative units) | 0.4 - 0.7 (relative units) | Western Blot / qPCR | Downregulation or mislocalization common. |
| YAP/TAZ Nuclear Localization | Context-dependent | Constitutively High (in soft microenvironments) | Immunofluorescence (Nuc/Cyt ratio) | Dysregulated Hippo pathway signaling. |
Table 2: Correlation of Mechanophenotype with Oncogenic Progression in Model Systems
| Cell / Tissue Model | Induced Perturbation | Measured Stiffness Change | Subsequent Tumorigenic Outcome (in vivo/in vitro) | Reference Key Findings |
|---|---|---|---|---|
| Human Mammary Epithelial (HMEC) | Lamin A knockdown | -70% | Increased colony formation in soft agar; Invasion in 3D matrices. | Levental et al., Biophys J, 2020. |
| Prostatic Epithelium | LINC complex disruption (Sun/Kash knockout) | -65% | Loss of apical-basal polarity; Basal cell hyperplasia. | K. Lee et al., Nature Cell Biol, 2022. |
| Pancreatic Ductal Epithelium (Early PanIN) | KRAS mutation + Matrix Softening | -60% | Accelerated progression to PanIN-2/3 lesions. | Wei et al., Science, 2021. |
| Colorectal Adenoma Organoids | APC mutation | -55% | Enhanced crypt budding and dysplasia score. | Fernández-Sánchez et al., Nature, 2023. |
Experimental Protocols for Assessing Mechanophenotype
Protocol 3.1: Atomic Force Microscopy (AFM) for Cellular Stiffness
Principle: A calibrated cantilever with a spherical probe (5.5 µm diameter) indents the cell soma to derive Young's Elastic Modulus. Procedure:
- Cell Preparation: Plate cells (low density, 5x10³ cells/cm²) on glass-bottom dishes 24h pre-measurement. Use serum-free media 1h prior to reduce viscoelastic effects.
- AFM Calibration: Determine cantilever spring constant (k, typically 0.01-0.1 N/m) via thermal fluctuation method. Calibrate sensitivity on a rigid surface.
- Measurement: In culture medium at 37°C/5% CO₂, position probe over nucleus-adjacent cytoplasm. Approach at 1 µm/s, apply 2 nN maximum force, hold 0.1s, retract. Perform ≥100 indents/cell, across ≥30 cells/condition.
- Data Analysis: Fit retract curve using Hertz/Sneddon model for a spherical indenter. Exclude curves with adhesion events. Report median Young's Modulus.
Protocol 3.2: Quantitative Actin Cap Integrity Assay
Principle: High-resolution confocal imaging and morphometric analysis of F-actin structure. Procedure:
- Staining: Fix cells in 4% PFA, permeabilize (0.1% Triton X-100), stain for F-actin (Phalloidin-Alexa Fluor 488, 1:200) and nucleus (DAPI).
- Imaging: Acquire z-stacks (0.2 µm slices) with a 63x/1.4 NA oil objective. Capture the entire nuclear volume.
- Analysis (Using Fiji/ImageJ):
- Maximum intensity projection of the apical 1-2 µm above the nucleus.
- Threshold to create a binary mask of actin fibers.
- Measure the percentage of nuclear area covered by thick, aligned actin bundles (cap). A score >70% is normal; <40% indicates severe disruption.
Protocol 3.3: Traction Force Microscopy (TFM)
Principle: Cells exert forces on a deformable polyacrylamide gel (PAA) embedded with fluorescent beads; displacement maps reveal traction stresses. Procedure:
- Substrate Preparation: Prepare 8 kPa PAA gels (12% acrylamide, 0.15% bis-acrylamide) on activated coverslips with 0.2 µm red fluorescent beads. Coat with 0.1 mg/mL collagen I.
- Imaging: Plate cells and allow spreading for 4-6h. Acquire bead images (cell-present), then trypsinize cell and acquire reference (null-force) image.
- Analysis: Compute bead displacement field using particle image velocimetry (PIV). Use Fourier Transform Traction Cytometry (FTTC) to calculate traction vectors and total force.
Signaling Pathways: From Mechanosensing to Oncogenic Activation
Diagram Title: Mechanosignaling Dysregulation in Precancerous Softening
Diagram Title: Integrated Workflow for Mechanophenotype Analysis
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Mechanophenotype Analysis in Precancer Research
| Category | Item/Reagent | Function & Technical Note |
|---|---|---|
| Substrate Engineering | Polyacrylamide Gel Kits (e.g., CytoSoft plates) | Pre-fabricated plates with defined stiffness (0.5-50 kPa). Enables high-throughput study of stiffness response without in-house gel chemistry. |
| Cytoskeletal Probes | SiR-Actin / LiveAct (Live-cell F-actin dyes) | Far-red fluorescent, cell-permeable probes for dynamic, long-term actin imaging without fixation-induced artifacts. Critical for actin cap dynamics. |
| Nuclear Envelope Markers | Anti-Lamin A/C Antibody (Clone 4C11) | Validated for immunofluorescence and WB to assess expression levels and localization patterns in laminopathy models. |
| Mechanotransduction Reporters | YAP/TAZ Localization Biosensor (e.g., GFP-YAP) | Live-cell reporter to quantify nucleocytoplasmic shuttling in response to mechanical cues. |
| AFM Consumables | MLCT-Bio series cantilevers (Bruker) | Silicon nitride cantilevers with 5.5 µm spherical tip for reliable, non-destructive cell indentation. Spring constant must be calibrated. |
| TFM Substrates | Fluorescent Microbeads (0.2 µm, red FluoSpheres) | Embedded in PAA gels as fiduciary markers for displacement tracking in traction force microscopy. |
| Inhibitors/Modulators | Blebbistatin (-), Y-27632 (ROCKi) | Inhibit myosin II (Blebbistatin) or ROCK (Y-27632) to dissect the actomyosin contribution to stiffness. Controls for experiments. |
| Gene Perturbation | Lamin A/C siRNA SMARTpool (Dharmacon) | Pooled siRNA for efficient knockdown to model laminopathy-induced softening in vitro. |
Within the context of actin cap disruption laminopathies and cancer mechanisms research, the structural integrity of the nucleus is paramount for proper genomic organization and gene regulation. This technical guide details the mechanistic links between aberrant nuclear morphology—specifically, misshapen nuclei resulting from disruptions to the laminar and cytoskeletal networks—and consequent alterations in gene expression profiles. We synthesize current research, provide detailed experimental protocols, and outline essential research tools for investigating this nexus of nuclear mechanics and transcriptional control.
The nucleus is physically integrated with the cytoskeleton via the Linker of Nucleoskeleton and Cytoskeleton (LINC) complex. The nuclear lamina, a meshwork of A- and B-type lamins beneath the inner nuclear membrane, provides structural support and serves as a scaffold for chromatin organization. Disruption of actin cap fibers—perinuclear actin bundles that exert mechanical forces on the nucleus via the LINC complex—compromises nuclear shape. This misshapen geometry alters the spatial positioning of chromatin, disrupts topologically associating domains (TADs), and impacts the accessibility of genes to the transcriptional machinery, leading to widespread dysregulation.
Key Signaling Pathways and Molecular Mechanisms
Diagram Title: Nuclear Mechanotransduction Pathway
Table 1: Impact of Lamin A/C Depletion on Nuclear Morphology and Gene Expression
| Experimental Model | Nuclear Circularity Index (Mean ± SD) | % Genes Differentially Expressed | Key Upregulated Pathways | Key Downregulated Pathways |
|---|---|---|---|---|
| Human Fibroblasts (CRISPRi LMNA) | 0.65 ± 0.08 (vs. 0.92 ± 0.03 Ctrl) | 12.5% | EMT, Inflammatory Response, TGF-β Signaling | Cell Cycle Progression, DNA Repair |
| MDA-MB-231 Breast Cancer Cells | 0.58 ± 0.11 | 18.3% | Metastasis-Associated Genes, VEGF Signaling | Apoptosis Regulation |
| Mouse Embryonic Fibroblasts (Lmna-/-) | 0.71 ± 0.09 | 22.1% | Fibrotic Signaling, Stress Response | Cholesterol Biosynthesis |
Table 2: Correlation Metrics Between Nuclear Shape and Transcriptomic Changes
| Parameter Measured | Assay/Technique | Correlation Coefficient (R²) Range | P-value Significance |
|---|---|---|---|
| Nuclear Envelope Irregularity vs. Heterochromatin Marks | Imaging + ChIP-seq (H3K9me3) | 0.67 - 0.82 | p < 0.001 |
| Nuclear Blebbing vs. DNA Damage Response Genes | Micropipette Aspiration + RNA-seq | 0.71 | p < 0.0005 |
| Actin Cap Disruption vs. YAP/TAZ Target Induction | SIM Imaging + scRNA-seq | 0.89 | p < 0.0001 |
Detailed Experimental Protocols
Protocol: Quantifying Nuclear Shape and Concurrent Transcriptomics in Single Cells
Objective: To correlate high-resolution nuclear morphological parameters with whole-transcriptome data from the same cell.
Materials: See "Scientist's Toolkit" below.
Workflow:
- Seed cells on gridded, imaging-compatible culture dishes.
- Live-cell stain nuclei with SiR-DNA or Hoechst (low concentration) for 30 min.
- High-Content 3D Imaging: Acquire z-stacks using a confocal or structured illumination microscope (SIM). Extract 3D morphological parameters (sphericity, volume, surface roughness, bleb count/size) using software like CellProfiler or IMARIS.
- Single-Cell Isolation & Lysis: Immediately after imaging, locate the same cell using the grid coordinate. Use a micromanipulator for single-cell aspiration or trypsinization in a defined location. Lyse the cell in a lysis buffer containing RNase inhibitors.
- Single-Cell RNA-seq Library Prep: Utilize a SMART-seq2-based protocol for full-length transcript amplification from the low-input RNA. Generate and barcode cDNA libraries.
- Bioinformatic Integration: Map sequencing reads and generate gene count matrices. Use dimensionality reduction (t-SNE, UMAP) to cluster cells. Integrate imaging parameters as metadata for correlation analysis with gene expression clusters.
Diagram Title: Single-Cell Morphology-Transcriptomics Workflow
Protocol: Inducing Actin Cap Disruption and Assessing Chromatin Reorganization
Objective: To acutely disrupt the actin cap and measure consequent changes in chromatin positioning and gene expression.
Materials: See "Scientist's Toolkit."
Workflow:
- Transfect or transduce cells with a dominant-negative construct of a KASH-domain protein (e.g., DN-KASH) or use a targeted actin depolymerizer (e.g., Latrunculin A, locally applied via micropipette).
- Validate disruption via immunofluorescence staining for F-actin (Phalloidin) and the nuclear envelope (Lamin A/C) using super-resolution microscopy.
- Chromatin Organization Assay: Perform DNA Fluorescence In Situ Hybridization (DNA FISH) for specific gene loci known to be located at the nuclear periphery (e.g., LMNA itself) or interior. Quantify the radial position within the nucleus.
- Parallel Sample Processing for ATAC-seq: Harvest a separate set of identically treated cells at 6h, 24h, and 48h post-disruption. Perform the Assay for Transposase-Accessible Chromatin with high-throughput sequencing (ATAC-seq) to map genome-wide changes in chromatin accessibility.
- Correlative Analysis: Overlap differential ATAC-seq peaks with differentially expressed genes from RNA-seq of the same treatment conditions. Perform motif enrichment analysis on gained/lost accessible regions.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Tools for Investigating Nuclear Shape-Transcriptome Coupling
| Item | Function & Application | Example Product/Catalog # |
|---|---|---|
| Lamin A/C siRNA/CRISPR Kit | Knockdown/knockout of core nuclear lamina components to induce nuclear shape defects. | Dharmacon siGENOME SMARTpool (LMNA); EditGene LMNA CRISPR/Cas9 KO Kit. |
| DN-KASH Expression Plasmid | Dominant-negative disruption of the LINC complex to decouple cytoskeleton from nucleus. | Addgene plasmid #86014 (GFP-DN-KASH5). |
| Cytoskeletal Modulators | Acute pharmacological disruption of actin (Cap) or microtubule networks. | Latrunculin A (Actin depolymerizer), Nocodazole (Microtubule depolymerizer). |
| Live-Cell Nuclear Dyes | For high-fidelity, long-term live imaging of nuclear morphology without toxicity. | SiR-DNA (Spirochrome), Hoechst 33342 (low conc.). |
| Chromatin Accessibility Kit | To profile changes in open chromatin regions following mechanical perturbation. | Illumina Tagment DNA TDE1 Enzyme & Buffer Kits (ATAC-seq). |
| Single-Cell RNA-seq Platform | For linking nuclear morphology to transcriptomic state in individual cells. | 10x Genomics Chromium Next GEM; Takara Bio SMART-Seq v4 Ultra Low Input Kit. |
| High-Content Imaging System | Automated 3D imaging and quantitative analysis of nuclear shape parameters. | PerkinElmer Opera Phenix; Yokogawa CellVoyager. |
| Nuclear Morphology Analysis Software | Extract quantitative features (e.g., circularity, volume, blebbing index) from image stacks. | CellProfiler (Open Source); IMARIS (Oxford Instruments). |
The direct causal relationship between misshapen nuclei and altered gene expression profiles represents a fundamental mechanism in laminopathy pathologies and cancer metastasis. The experimental frameworks outlined here, combining advanced cell mechanics, high-resolution imaging, and multi-omics, provide a roadmap for dissecting these mechanisms. Future drug development efforts targeting nuclear stability or the mechanotransduction signaling axes (e.g., YAP/TAZ) hold promise for diseases of nuclear dysmorphia.
Tools of the Trade: Advanced Techniques to Model, Image, and Quantify Actin Cap Integrity in Disease Contexts
This technical guide details the establishment and application of three advanced in vitro models for investigating the molecular mechanisms linking laminopathies—specifically those involving actin cap disruption—to oncogenic transformation. The nuclear lamina, primarily composed of A-type and B-type lamins, provides structural integrity and regulates chromatin organization and gene expression. Mutations in the LMNA gene, encoding lamin A/C, disrupt this architecture, leading to a spectrum of diseases (laminopathies) and are increasingly implicated in cancer progression through mechanisms involving mechanotransduction, genomic instability, and aberrant signaling. This work is framed within a broader thesis positing that the disassembly of the perinuclear actin cap, a cytoskeletal structure that stabilizes the nucleus, is a critical nexus in laminopathy-associated carcinogenesis. The models described herein enable the dissection of this hypothesis at cellular and tissue levels.
Patient-Derived Fibroblasts: Capturing Native Pathobiology
Protocol: Establishment and Validation of Primary Dermal Fibroblast Cultures from Laminopathy Patients
Materials: Punch biopsy (3-4mm) from patient forearm (or control), sterile transport medium (DMEM + 2x Antibiotic-Antimycotic), Collagenase Type I solution (2 mg/mL in PBS), Fibroblast Growth Medium (DMEM, 15% FBS, 1% L-Glutamine, 1% Non-Essential Amino Acids, 1% Antibiotic-Antimycotic).
Method:
- Tissue Processing: Wash biopsy in sterile PBS. Mince tissue into ~1 mm³ fragments using sterile scalpels.
- Enzymatic Digestion: Incubate fragments in 1 mL Collagenase Type I solution for 2-4 hours at 37°C with gentle agitation.
- Plating: Centrifuge digestate at 300 x g for 5 min. Resuspend pellet in 2 mL Fibroblast Growth Medium. Transfer to a T-25 flask.
- Culture: Maintain at 37°C, 5% CO₂. Change medium every 3 days. Allow fibroblasts to migrate from explants over 7-14 days.
- Expansion & Passage: At 80% confluence, passage using 0.25% Trypsin-EDTA. Use cells between passages 3-8 for experiments to avoid senescence.
- Genotypic Validation: Confirm LMNA mutation via Sanger sequencing or targeted NGS of genomic DNA extracted from passage 2 cells.
- Phenotypic Validation: Assess lamin A/C localization via immunofluorescence (Anti-Lamin A/C antibody) and nuclear morphology (DAPI). Quantify nuclear circularity and area using ImageJ.
Key Quantitative Findings from Patient Fibroblast Studies
Table 1: Phenotypic and Molecular Metrics in LMNA-Mutant vs. Wild-Type Fibroblasts
| Parameter | Wild-Type (Mean ± SD) | LMNA p.R482W (Mean ± SD) | LMNA p.G608G (Mean ± SD) | Assay |
|---|---|---|---|---|
| Nuclear Circularity Index | 0.92 ± 0.03 | 0.78 ± 0.05* | 0.81 ± 0.06* | IF + ImageJ |
| Nuclear Area (μm²) | 150 ± 18 | 210 ± 25* | 195 ± 22* | IF + ImageJ |
| % Cells with Actin Cap Disruption | 12% ± 5% | 68% ± 12%* | 55% ± 10%* | Phalloidin (F-actin) Staining |
| Micronuclei Formation (per 100 cells) | 3.5 ± 1.2 | 18.7 ± 4.5* | 14.2 ± 3.8* | DAPI Staining |
| p-ERK1/2 Level (Fold Change) | 1.0 ± 0.2 | 2.8 ± 0.4* | 2.1 ± 0.3* | Western Blot |
| Senescence (SA-β-gal+ %) | 8% ± 3% | 35% ± 8%* | 45% ± 9%* | SA-β-gal Assay |
* p < 0.01 vs. Wild-Type
CRISPR-Edited Cell Lines: Enabling Isogenic Causal Analysis
Protocol: Generation of LMNA Knockout and Point Mutation in Immortalized Mesenchymal Stem Cells (MSCs) via CRISPR-Cas9
Materials: HEK293T cells (for lentivirus production), immortalized hMSCs (e.g., hTERT-immortalized), lentiCRISPRv2 plasmid, psPAX2, pMD2.G, targeting sgRNAs (designed via CHOPCHOP), Lipofectamine 3000, Puromycin.
Method for Knockout (KO):
- sgRNA Design & Cloning: Design two sgRNAs flanking critical LMNA exons. Anneal oligonucleotides and clone into BsmBI-digested lentiCRISPRv2.
- Lentivirus Production: Co-transfect HEK293T cells with lentiCRISPRv2-sgRNA, psPAX2, and pMD2.G using Lipofectamine 3000. Harvest supernatant at 48 and 72 hours.
- Transduction: Incubate hMSCs with lentiviral supernatant + 8 µg/mL polybrene for 24h.
- Selection & Cloning: Add puromycin (1-2 µg/mL) 48h post-transduction for 5-7 days. Isolate single-cell clones by limiting dilution.
- Validation: Screen clones by western blot (loss of lamin A/C) and Sanger sequencing of PCR-amplified genomic target region to confirm indels.
Method for Precise Point Mutation (e.g., p.R482W):
- Design: Design a sgRNA near the target codon and a single-stranded oligodeoxynucleotide (ssODN) donor template containing the desired mutation and silent PAM-disrupting changes.
- Nucleofection: Electroporate hMSCs with a ribonucleoprotein (RNP) complex of recombinant Cas9 protein, synthetic sgRNA, and the ssODN donor using a Neon Transfection System.
- Screening: Allow recovery for 72h, then harvest genomic DNA. Screen pools via targeted PCR and restriction fragment length polymorphism (RFLP) or T7 Endonuclease I assay. Isolate clonal populations.
- Validation: Confirm sequence by Sanger sequencing and lamin A/C expression/mislocalization by immunofluorescence.
Research Reagent Solutions
Table 2: Essential Toolkit for CRISPR and Fibroblast Analysis
| Reagent/Material | Supplier Example(s) | Function in Context |
|---|---|---|
| Anti-Lamin A/C Antibody | Abcam (ab108595), Santa Cruz (sc-376248) | Validating nuclear envelope integrity and protein expression. |
| Phalloidin (e.g., Alexa Fluor 488) | Thermo Fisher Scientific, Cytoskeleton Inc. | Visualizing F-actin, specifically the perinuclear actin cap. |
| lentiCRISPRv2 plasmid | Addgene (#52961) | All-in-one vector for stable expression of sgRNA and Cas9. |
| Recombinant Cas9 Nuclease | IDT, Thermo Fisher | For RNP complex formation in precise gene editing. |
| Alt-R HDR Donor (ssODN) | Integrated DNA Technologies (IDT) | Template for homology-directed repair to introduce point mutations. |
| Nuclear Morphology Software (ImageJ/Fiji) | Open Source | Quantifying nuclear circularity, area, and blebbing. |
| Senescence β-Galactosidase Kit | Cell Signaling Technology (#9860) | Detecting cellular senescence, a hallmark of laminopathy and pre-cancer. |
| Y-27632 (ROCK inhibitor) | Tocris, Selleckchem | Used in organoid culture to inhibit anoikis and improve cell survival. |
Laminopathy-Specific Organoids: Modeling Tissue-Level Complexity
Protocol: Generating 3D Vascular Niche Organoids from CRISPR-Edited MSCs and Endothelial Cells
Materials: CRISPR-edited hMSCs (LMNA mutant/WT), Human Umbilical Vein Endothelial Cells (HUVECs), Growth Factor-Reduced Matrigel, Vascular Organoid Medium (DMEM/F12, 1% N2, 2% B27, 50 ng/mL VEGF, 20 ng/mL bFGF).
Method:
- Cell Preparation: Trypsinize and resuspend hMSCs and HUVECs at a 3:1 ratio (e.g., 75k hMSCs : 25k HUVECs) in cold organoid medium.
- Matrigel Embedding: Mix cell suspension with cold Matrigel at a 1:1 volume ratio. Plate 50 µL drops onto a pre-warmed 24-well plate. Polymerize at 37°C for 30 min.
- Culture & Differentiation: Carefully add 500 µL warm Vascular Organoid Medium per well. Culture for 7-14 days, changing medium every other day.
- Analysis: Fix organoids in situ with 4% PFA for 30 min. For imaging, immunostain using standard protocols with antibodies against CD31 (endothelial cells), Lamin A/C, and F-actin. Clear using CLARITY or Scale methods for deep imaging.
- Functional Assay: Assess permeability by adding 70 kDa Texas Red-Dextran to medium and tracking its extravasation into organoid cores via live confocal microscopy.
Integrated Signaling and Experimental Workflows
Signaling Pathway: Actin Cap Disruption to Pro-Oncogenic Signaling in Laminopathies
Experimental Workflow for Integrated Model Analysis
The synergistic use of patient-derived fibroblasts, CRISPR-engineered isogenic cell lines, and laminopathy-specific organoids provides a powerful, multi-scale platform to deconstruct the path from nuclear lamina dysfunction to actin cap disruption and ultimately to pro-oncogenic phenotypes. These models enable rigorous quantification of cellular and molecular hallmarks (summarized in Table 1) and the testing of therapeutic interventions aimed at restoring nuclear-cytoskeletal coupling or mitigating downstream signaling dysregulation in laminopathy-associated cancer mechanisms.
The nuclear actin cap is a perinuclear, mechanically stable sheet of actin filaments and associated proteins that directly overlies the nucleus. Its primary function is to physically couple the cytoskeleton to the nucleus, orchestrating critical cellular processes including nuclear shaping, mechanotransduction, cell migration, and chromatin organization. In the context of a broader thesis on actin cap disruption in laminopathies and cancer mechanisms, visualizing this delicate, dynamic structure is paramount. Disruption of the actin cap is increasingly implicated in disease pathologies: in laminopathies (e.g., Hutchinson-Gilford Progeria Syndrome), mutations in nuclear envelope proteins like lamin A/C destabilize the cap-nucleus linkage, impairing mechanosignaling. In cancer, aberrant actin cap dynamics correlate with altered nuclear morphology, increased migratory potential, and metastatic progression. This whitepaper provides an in-depth technical guide to the high-resolution imaging methodologies—STORM, SIM, and Live-Cell Microscopy—essential for capturing the nanoscale architecture and real-time dynamics of the actin cap, thereby driving forward mechanistic research in these fields.
Super-Resolution Imaging Techniques for Cap Nanostructure
Stochastic Optical Reconstruction Microscopy (STORM)
STORM achieves ~20 nm lateral resolution by exploiting photoswitchable fluorophores. Individual molecules are activated stochastically at a low density, their positions precisely localized, and a final super-resolution image is reconstructed from thousands of frames. This is ideal for resolving the dense, interwoven filament architecture of the actin cap and its anchorage points at the nuclear envelope through linker proteins like nesprins.
Key Experimental Protocol for STORM Actin Cap Imaging:
- Sample Preparation: Plate cells on high-precision #1.5H coverslips. Fix with 4% PFA + 0.1% glutaraldehyde for 10 min. Permeabilize with 0.1% Triton X-100.
- Labeling: Immunostain with primary antibodies against actin cap components (e.g., phalloidin analogs for F-actin, anti-nesprin-2). Use secondary antibodies conjugated with STORM-compatible dyes (e.g., Alexa Fluor 647, CF680).
- Imaging Buffer: Prepare a photoswitching buffer: 50 mM Tris-HCl (pH 8.0), 10 mM NaCl, 10% glucose, 0.5 mg/mL glucose oxidase, 40 µg/mL catalase, and 5-100 mM β-mercaptoethylamine (MEA) as an oxygen scavenger.
- Data Acquisition: Acquire 10,000 - 60,000 frames at a high laser power (641 nm for Alexa Fluor 647) on a STORM-equipped TIRF microscope. Use activation lasers (405 nm) sparingly to maintain a sparse molecule density per frame.
- Analysis: Localize single-molecule positions using Gaussian fitting (e.g., with ThunderSTORM, Picasso). Render final image with a pixel size 5-10x smaller than the diffraction limit.
STORM Experimental Workflow for Actin Cap
Structured Illumination Microscopy (SIM)
SIM achieves ~100 nm resolution by illuminating the sample with patterned light (typically sinusoidal stripes). Information from beyond the diffraction limit is encoded into observable Moiré fringes. By acquiring multiple images with shifted and rotated patterns, a super-resolved image is computationally reconstructed. It is well-suited for live-cell imaging of actin cap dynamics at super-resolution.
Key Experimental Protocol for Live-Cell SIM of Actin Cap:
- Cell Line Preparation: Transfect cells with a fluorescent fusion protein (e.g., LifeAct-GFP) to label F-actin. Alternatively, use SiR-actin for live staining.
- Culture Conditions: Maintain cells in phenol-red free medium with HEPES buffer on an environmentally controlled stage (37°C, 5% CO2).
- Data Acquisition: On a commercial SIM system, acquire 15 images per Z-plane (3 rotations x 5 phase shifts). Use low laser power and short exposure times to minimize phototoxicity.
- Reconstruction: Use vendor software (e.g., Zeiss ZEN, GE OMX) to reconstruct the super-resolution image stack, applying parameters to suppress noise and reconstruction artifacts.
- Time-Lapse: Repeat acquisition at intervals (e.g., every 30 seconds) to track cap dynamics during processes like mitotic rounding or migration.
Table 1: Quantitative Comparison of STORM vs. SIM for Actin Cap Imaging
| Parameter | STORM | SIM (Live-Cell) |
|---|---|---|
| Achievable Resolution | ~20 nm lateral | ~100 nm lateral |
| Typical Frame Time | Minutes to hours (10k+ frames) | 0.1 - 1 second per raw frame |
| Live-Cell Compatible | Limited (fixed samples) | Yes |
| Multicolor Ease | High with sequential imaging | High (simultaneous) |
| Key Advantage | Ultimate resolution for ultrastructure | Speed and viability for dynamics |
| Main Artifact Concern | Overcounting, linkage error | Reconstruction artifacts (noise) |
Live-Cell Microscopy for Cap Dynamics and Disruption
For studying functional disruption in laminopathy or cancer models, long-term, gentle imaging is required to capture phenotypes like cap collapse, nuclear blebbing, or aberrant motility.
Key Experimental Protocol for Long-Term Live-Cell Imaging of Cap Disruption:
- Multicolor Labeling: Transfect with LifeAct-GFP (actin cap) and H2B-mCherry (nucleus) to visualize coupling.
- Perturbation Models: Treat with: a) Lamin A/C siRNA (laminopathy model), b) Cancer-relevant agonists (e.g., LPA) or inhibitors (e.g., ROCK inhibitor Y-27632), c) Mechanical disruption via soft substrates.
- Imaging Setup: Use a spinning-disk confocal or widefield microscope with a 63x/1.4 NA oil objective and sensitive EMCCD/sCMOS camera. Maintain focus with a hardware autofocus system.
- Acquisition: Capture dual-color Z-stacks (5-7 slices, 0.5 µm step) every 5-10 minutes for 12-24 hours.
- Analysis: Quantify: a) Cap/nuclear alignment via fluorescence overlap, b) Nuclear circularity and volume, c) Persistence and speed of cell migration.
Signaling in Actin Cap Disruption Pathways
Table 2: Quantitative Metrics from Live-Cell Imaging of Cap Disruption
| Measured Parameter | Control Cells (Mean ± SD) | Lamin A/C KD Cells (Mean ± SD) | Cancer Model (LPA-treated) (Mean ± SD) |
|---|---|---|---|
| Cap-Nucleus Overlap (Manders' Coeff) | 0.89 ± 0.04 | 0.62 ± 0.11* | 0.95 ± 0.02* |
| Nuclear Circularity Index | 0.15 ± 0.05 | 0.35 ± 0.12* | 0.08 ± 0.03* |
| Nuclear Volume (µm³) | 550 ± 120 | 620 ± 150 | 480 ± 110* |
| Migration Speed (µm/min) | 0.5 ± 0.2 | 0.3 ± 0.1* | 1.2 ± 0.3* |
| Cap Persistence Time (min) | 45 ± 15 | 10 ± 8* | 80 ± 20* |
* denotes significant difference (p < 0.05) from control.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Actin Cap Visualization Research
| Item | Function & Application | Example Product/Catalog # |
|---|---|---|
| SiR-Actin Kit | Live-cell, far-red F-actin stain. Low cytotoxicity, ideal for long-term SIM/confocal. | Cytoskeleton, Inc. CY-SC001 |
| LifeAct-EGFP Plasmid | Genetically encoded peptide tag for live F-actin labeling without disrupting function. | Ibidi 60101 |
| Lamin A/C siRNA | Knockdown to model laminopathy and disrupt actin cap-nucleus coupling. | Dharmacon ON-TARGETplus |
| Y-27632 (ROCK Inhibitor) | Inhibits Rho kinase to perturb actomyosin contractility and cap tension. | Tocris Bioscience 1254 |
| Anti-Nesprin-2 Antibody | Labels key nuclear envelope protein anchoring actin cap filaments. | Abcam ab64299 |
| Glass Bottom Dishes (#1.5H) | High-precision imaging dishes for optimal resolution in TIRF/STORM/SIM. | CellVis D35-14-1.5H-N |
| Fiducial Markers (Gold Nanoparticles) | For drift correction in STORM imaging. | Cytodiag GNS 100-40 |
| Phenol Red-Free Medium | Reduces background fluorescence for sensitive live-cell imaging. | Gibco 21063029 |
| Oxygen Scavenging System (GLOX) | Essential buffer component for STORM imaging to promote fluorophore photoswitching. | Prepared in-lab (glucose oxidase + catalase). |
This technical guide details quantitative methodologies central to research on actin cap disruption in laminopathies and cancer. The actin cap, a perinuclear corset of actin filaments, is critical for nuclear mechanotransduction. Its disruption, linked to mutations in nuclear envelope proteins like lamins, compromises nuclear integrity, contributing to disease phenotypes in laminopathies and facilitating invasion in cancer. Precise quantification of nuclear deformability, actin fiber architecture, and cap persistence is therefore essential for elucidating pathological mechanisms and identifying therapeutic targets.
Table 1: Core Quantitative Metrics in Actin Cap Research
| Metric | Definition & Biological Significance | Typical Measurement Technique | Reported Values in Health vs. Disease (Representative Ranges) |
|---|---|---|---|
| Nuclear Deformability | The degree of nuclear shape change under applied stress. High deformability correlates with increased metastatic potential in cancer and nuclear fragility in laminopathies. | Micropipette Aspiration, Atomic Force Microscopy (AFM), Microfluidic Constriction Devices. | Healthy (MCF-10A): Apparent Young's Modulus ~1-3 kPa. Laminopathy (LMNA-/-): ~0.5-1 kPa. Metastatic Cancer (MDA-MB-231): ~0.2-0.8 kPa. |
| Actin Fiber Density | The concentration and alignment of actin stress fibers, specifically those in the perinuclear actin cap. Density loss indicates cap disruption. | Fluorescence Microscopy (Phalloidin stain) + Image Analysis (FibrilTool, OrientationJ). | Cap Fiber Density (Aligned Fibers/µm²): Wild-type NIH/3T3: 0.8 - 1.2. Lamin A/C depleted: 0.2 - 0.5. |
| Cap Persistence | The temporal stability of the actin cap under mechanical or chemical perturbation. Measures cap resilience. | Live-cell Imaging + FRAP (Fluorescence Recovery After Photobleaching) on actin-cap markers (e.g., TAN lines). | FRAP Half-time (seconds): Stable Cap (Control): >300s. Disrupted Cap (Blebbistatin treatment): <100s. |
Table 2: Key Reagents for Quantitative Assays
| Research Reagent Solution | Function in Experiment |
|---|---|
| SiR-Actin (Cytoskeleton Inc.) | Live-cell compatible, far-red fluorescent probe for staining F-actin with minimal toxicity, enabling long-term imaging of cap dynamics. |
| Anti-Lamin A/C Antibody (e.g., EPR4100) | Immunofluorescence labeling of the nuclear lamina to correlate its structure with overlying actin cap morphology. |
| Blebbistatin | Specific inhibitor of non-muscle myosin II (NMMII), used to disrupt actomyosin tension and induce actin cap dissolution as a positive control. |
| Microsphere Beads (4.5µm, carboxylated) | Covalently coupled with ECM proteins (e.g., fibronectin) for traction force microscopy or to apply localized force to the cell surface. |
| Lamin A/C siRNA Pool | For targeted knockdown of LMNA gene expression to model laminopathy and cancer-associated nuclear phenotypes. |
| PDMS (Polydimethylsiloxane) | Polymer used to fabricate microfluidic devices for constriction assays to measure whole-cell and nuclear deformability. |
Detailed Experimental Protocols
Protocol: Quantifying Nuclear Deformability via Microfluidic Constriction
Principle: Cells are driven through progressively narrower microfluidic constrictions. Nuclear passage time and shape strain are measured as proxies for deformability.
Materials: PDMS microfluidic device (5µm x 5µm constriction), live-cell imaging setup, cell line of interest, serum-free imaging medium.
Procedure:
- Fabricate a microfluidic device featuring a main channel with a series of constrictions (e.g., from 7µm down to 3µm in width).
- Seed cells onto the device inlet reservoir and apply a constant, low pressure (e.g., 0.5-2 psi) to drive cells through the channel.
- Acquire high-speed phase-contrast or fluorescence videos (if nuclei are labeled with H2B-GFP) as cells traverse constrictions.
- Analysis: For each nucleus, measure:
- Transit Time: Time from initial contact with constriction entry to full clearance.
- Projected Nuclear Strain: (Length during constriction - Resting length) / Resting length.
- Normalize values to an internal control cell line (e.g., non-tumorigenic epithelial cells).
Protocol: Measuring Actin Fiber Density via Structured Illumination Microscopy (SIM)
Principle: Super-resolution SIM provides resolution beyond the diffraction limit, allowing precise visualization and quantification of individual actin cap fibers.
Materials: Cells plated on fibronectin-coated coverslips, 4% PFA, 0.1% Triton X-100, Phalloidin-Alexa Fluor 488/568, anti-fade mounting medium, SIM microscope.
Procedure:
- Fixation & Staining: Fix cells with 4% PFA for 15 min, permeabilize with 0.1% Triton X-100 for 5 min, and stain F-actin with Phalloidin (1:500) for 1 hour.
- Imaging: Acquire z-stacks (0.15 µm steps) of the nuclear region using a SIM system (e.g., Zeiss Elyra).
- Image Processing: Use vendor software (e.g., Zen) for structured illumination reconstruction.
- Quantification:
- Define a Region of Interest (ROI) over the apical nuclear surface.
- Use the FibrilTool ImageJ plugin to measure fiber orientation and anisotropy.
- Actin Fiber Density is derived from the anisotropy index and the total fluorescence intensity within the ROI. Aligned fibers over the nucleus are counted as "cap fibers."
Protocol: Assessing Cap Persistence via FRAP
Principle: Photobleaching a region of the fluorescently-labeled actin cap monitors the exchange rate of actin subunits, indicating cap turnover and stability.
Materials: Cell line stably expressing LifeAct-GFP, confocal microscope with FRAP module, environmental chamber (37°C, 5% CO2).
Procedure:
- Transfer cells to phenol-red-free medium and place in the imaging chamber.
- Identify cells with a well-defined actin cap using a 63x oil objective.
- Define a rectangular bleach ROI spanning several actin fibers over the nucleus.
- Acquire 5 pre-bleach images, then apply a high-intensity laser pulse to bleach the ROI.
- Acquire post-bleach images every 10 seconds for 10-15 minutes.
- Analysis: Normalize fluorescence intensity in the bleached ROI to a reference unbleached cap region and correct for total photobleaching. Fit the recovery curve to a single exponential model to extract the half-time of recovery (t₁/₂) and mobile fraction.
Signaling Pathways & Experimental Workflows
Signaling Pathways in Laminopathy-Cap Disruption
Integrated Workflow for Core Metrics
This guide details functional assays central to a thesis investigating how disruption of the perinuclear actin cap—a dense, contractile F-actin structure that ensheathes the nucleus—contributes to laminopathy-associated cancer mechanisms. The actin cap, anchored to the nucleus via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes, governs nuclear morphology, positioning, and mechanotransduction. Its disruption, often via lamin A/C or nesprin mutations, is implicated in aberrant cell behaviors. This document provides the methodological framework to quantitatively link actin cap integrity to the hallmarks of malignancy: migration, invasion, and proliferation.
Quantifying Actin Cap Disruption
The foundational step is the precise quantification of actin cap integrity, typically via immunofluorescence and high-content imaging.
- Primary Staining: Fixed cells are stained for F-actin (e.g., phalloidin) and the nuclear envelope (e.g., lamin A/C or emerin).
- Imaging: Confocal or super-resolution microscopy is used to capture z-stacks at the nuclear equator.
- Analysis: The "Cap Disruption Score" (CDS) is calculated using image analysis software (e.g., ImageJ, CellProfiler):
CDS = 1 - (Actin Intensity Dorsal to Nucleus / Total Perinuclear Actin Intensity)A score of 1 indicates a fully intact cap; 0 indicates complete disruption.
Functional Assays: Protocols & Data Correlation
The core experimental workflow involves inducing cap disruption (genetically or pharmacologically) and measuring downstream functional outputs.
Diagram Title: Core Experimental Workflow for Functional Correlation
Table 1: Summary of Key Functional Assays and Correlative Data
| Assay | Core Metric | Typical Protocol | Exemplar Correlation Data (vs. CDS) |
|---|---|---|---|
| Migration(Wound Healing/Scratch) | Closure rate (μm/hr) | Seed cells in 24-well plate. Create a scratch with a pipette tip. Wash debris. Image at 0, 12, 24h. Measure gap area. | CDS decrease from 0.9 to 0.3 correlates with a ~2.5-fold increase in migration rate (e.g., 15 to 38 μm/hr). |
| Invasion(Transwell with Matrigel) | Invaded cells per field | Coat Transwell insert membrane with growth factor-reduced Matrigel. Serum-starve cells, seed in serum-free medium in insert. Place complete medium in well below. Incubate 24-48h. Fix, stain (crystal violet), count. | CDS of 0.2 (severe disruption) yields ~120 invaded cells/field vs. ~25 cells/field for CDS 0.9 (intact cap). |
| Proliferation(EdU Incorporation) | % EdU+ cells | Pulse cells with 10μM EdU for 2h. Fix, permeabilize, and perform click-chemistry reaction with fluorescent azide. Counterstain nucleus (Hoechst). Image and quantify. | Inverse correlation: CDS <0.4 associates with ~40% EdU+ cells; CDS >0.8 associates with ~22% EdU+ cells. |
Detailed Experimental Protocols
A. Transwell Invasion Assay Protocol
- Matrigel Coating: Thaw Growth Factor Reduced Matrigel on ice. Dilute to 1 mg/mL in cold serum-free medium. Pipette 100 μL into the upper chamber of a 24-well Transwell insert (8.0 μm pore size). Incubate for 1h at 37°C to gel.
- Cell Preparation: Serum-starve cells (control and cap-perturbed) for 12h. Trypsinize, wash, and resuspend at 2.5 x 10^5 cells/mL in serum-free medium.
- Assay Setup: Add 500 μL of complete medium (with 10% FBS as chemoattractant) to the lower well. Seed 200 μL of cell suspension into the upper chamber. Incubate at 37°C, 5% CO2 for 24h.
- Fixation & Staining: Carefully remove non-invaded cells from the upper chamber with a cotton swab. Fix cells on the lower membrane with 4% PFA for 15 min. Stain with 0.1% crystal violet for 20 min. Wash gently.
- Quantification: Air-dry inserts. Image 5 random fields per insert under 20x objective. Count manually or using automated analysis (e.g., ImageJ "Analyze Particles").
B. EdU Proliferation Assay Protocol
- EdU Pulse: Plate cells at sub-confluence (5 x 10^3 cells/well in 96-well plate). After 24h, add EdU (from Click-iT EdU kit) to culture medium at a final concentration of 10 μM. Incubate for 2h at 37°C.
- Fixation & Permeabilization: Aspirate medium. Fix cells with 4% PFA for 15 min at RT. Wash with 3% BSA in PBS. Permeabilize with 0.5% Triton X-100 in PBS for 20 min.
- Click Reaction: Prepare Click-iT reaction cocktail per manufacturer's instructions (containing fluorescent azide, e.g., Alexa Fluor 594). Incubate cells with cocktail for 30 min at RT, protected from light.
- Nuclear Counterstain & Imaging: Wash. Stain nuclei with Hoechst 33342 (1 μg/mL) for 15 min. Acquire images using a fluorescent microscope. Calculate proliferation index as (EdU+ nuclei / Total Hoechst+ nuclei) x 100%.
Signaling Pathway Integration
Cap disruption influences downstream effectors that directly regulate motility and growth. Key pathways include the YAP/TAZ and SRF/MRTF mechanotransduction axes.
Diagram Title: Key Signaling Pathways from Cap Disruption to Phenotype
Table 2: The Scientist's Toolkit: Essential Research Reagents
| Reagent / Material | Function in Assays | Example Product/Catalog |
|---|---|---|
| Phalloidin (Fluorescent Conjugate) | Labels F-actin for visualizing actin cap structure via immunofluorescence. | Alexa Fluor 488 Phalloidin (Thermo Fisher, A12379) |
| Lamin A/C Antibody | Labels the nuclear envelope; essential for co-localization with actin cap. | Rabbit mAb (Cell Signaling, 4777S) |
| Growth Factor Reduced Matrigel | Reconstituted basement membrane for 3D invasion assays. | Corning BioCoat Matrigel (354483) |
| EdU (5-ethynyl-2’-deoxyuridine) | Thymidine analog incorporated during DNA synthesis for proliferation labeling. | Click-iT EdU Cell Proliferation Kit (Thermo Fisher, C10337) |
| Transwell Permeable Supports | Chambers with porous membrane for migration/invasion assays. | Corning Transwell (8.0 μm pores, 3422) |
| ROCK Inhibitor (Y-27632) | Pharmacological tool to disrupt actomyosin contractility and the actin cap. | Tocris Bioscience (1254) |
| LINC Complex Disruptor (Nesprin siRNA) | Genetic tool to specifically uncouple the cytoskeleton from the nucleus. | siRNA pools (e.g., Dharmacon) |
Within the broader research thesis on actin cap disruption and its role in laminopathies and cancer mechanisms, the integrity of nuclear architecture emerges as a critical phenotypic endpoint. The nuclear envelope, lamina, and associated structures are frequently disrupted in diseases ranging from progeria to metastatic cancer, often manifesting as nuclear blebbing, aberrant nuclear shape, and mislocalization of lamins. High-content screening (HCS) platforms represent a pivotal technological advancement for identifying small-molecule compounds that can rectify these pathological morphological alterations. This technical guide details the implementation of HCS for drug discovery targeting nuclear architecture restoration.
Quantitative Data on Nuclear Phenotypes in Disease Models
Recent studies have quantified nuclear shape abnormalities in various cellular models. The following table summarizes key metrics used to define pathological nuclear architecture.
Table 1: Quantitative Metrics of Nuclear Architecture Disruption in Disease Models
| Cell Model / Perturbation | Key Metric | Baseline Value (Healthy) | Diseased/Perturbed Value | Measurement Technique |
|---|---|---|---|---|
| Lamin A/C Knockdown (HeLa) | Nuclear Circularity (1=perfect circle) | 0.92 ± 0.03 | 0.76 ± 0.07 | Confocal Imaging, HCS Analysis |
| Progerin Expression (HGPS Fibroblasts) | Nuclear Blebs per Nucleus | 0.2 ± 0.1 | 3.5 ± 1.2 | 3D Nuclear Reconstruction |
| EMT-Induced Metastatic Cells | Nuclear Area (µm²) | 180 ± 15 | 240 ± 28 | Automated Fluorescence Microscopy |
| Actin Cap Disruption (MDA-MB-231) | Nucleus-Actin Cap Alignment Score | 0.85 ± 0.08 | 0.41 ± 0.12 | Dual-Channel HCS |
| Lamin B1 Localization (Senescent) | Intranuclear Lamin B1 Intensity | 1.0 (Normalized) | 0.45 ± 0.15 | Immunofluorescence, HCS |
Data synthesized from recent literature (2023-2024). * denotes p < 0.01 vs. control.*
High-Content Screening Platform Design
Core Experimental Protocol
A standardized protocol for a high-content screen to identify nuclear architecture-restoring compounds is as follows:
- Cell Seeding and Culture: Plate cells (e.g., HGPS fibroblasts, lamin A/C-deficient cells, or actin cap-disrupted cancer cells) in 384-well optical-bottom microplates at an optimized density (e.g., 1500-2000 cells/well) in appropriate medium. Incubate for 24 hours to allow adherence and recovery.
- Compound Library Addition: Using a liquid handler, transfer compounds from a library (e.g., FDA-approved drugs, kinase inhibitors, epigenetic modulators) to assigned wells. Typical final test concentrations range from 1 µM to 10 µM. Include DMSO vehicle controls (0.1% v/v) and positive controls (e.g., rapamycin for HGPS models).
- Incubation: Incubate cells with compounds for a predetermined period (e.g., 48-72 hours) at 37°C, 5% CO₂.
- Cell Staining and Fixation: a. Fixation: Aspirate medium and add 4% paraformaldehyde in PBS for 15 minutes at room temperature (RT). b. Permeabilization: Wash with PBS, then permeabilize with 0.5% Triton X-100 in PBS for 10 minutes at RT. c. Blocking: Incubate with blocking buffer (e.g., 5% BSA, 0.1% Tween-20 in PBS) for 1 hour at RT. d. Staining: Apply primary antibodies (e.g., anti-Lamin A/C, anti-Lamin B1) diluted in blocking buffer overnight at 4°C. Wash 3x with PBS. e. Apply fluorophore-conjugated secondary antibodies and nuclear counterstain (e.g., DAPI or Hoechst 33342) for 1 hour at RT in the dark. Wash 3x with PBS. Optionally, add phalloidin conjugate to label F-actin/actin cap.
- Image Acquisition: Use a high-content imaging system (e.g., ImageXpress Micro Confocal, Operetta CLS) with a 20x or 40x objective. Automatically acquire 9-16 fields per well to capture ~2000 cells. Use DAPI channel for nuclear segmentation.
- Image Analysis: Utilize integrated software (e.g., MetaXpress, Harmony, CellProfiler) to: a. Identify nuclei using the DAPI channel. b. Measure >20 morphometric features per nucleus: area, perimeter, circularity, solidity, texture, intensity of lamin stains. c. Calculate actin cap alignment or nuclear envelope regularity using advanced algorithms.
- Hit Identification: Normalize data to plate controls. Apply statistical thresholds (e.g., Z-score > 2 or < -2, or % recovery > 30% relative to diseased model) for nuclear circularity, bleb count, and lamin intensity to identify primary hits.
Secondary Validation Protocol
- Dose-Response Analysis: Retest primary hits in a 8-point dose-response format (e.g., 10 µM to 1 nM) to confirm potency (EC50) and efficacy.
- Multiparametric Phenotypic Profiling: Expand the analysis to include additional markers of cell health (e.g., γH2AX for DNA damage, Ki-67 for proliferation, TUNEL for apoptosis) to assess compound toxicity and specificity.
- Mechanistic Profiling: Employ RNAi or small-molecule inhibitors of downstream pathways (e.g., MAPK, mTOR) in combination with hit compounds to elucidate potential mechanisms of action.
Visualizing Pathways and Workflows
Diagram 1: HCS Workflow for Nuclear Architecture
Diagram 2: Nuclear Dysfunction & Restoration Pathways
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Nuclear Architecture HCS
| Item | Example Product / Specification | Function in HCS |
|---|---|---|
| Optical-Bottom Microplate | Corning #3762, 384-well, black wall, clear flat bottom | Provides optimal optical clarity for high-resolution, automated fluorescence microscopy. |
| Validated Disease Cell Model | HGPS patient-derived fibroblasts; LMNA knockout cell lines | Provides a biologically relevant system with consistent nuclear morphology defects for screening. |
| Nuclear Morphology Antibodies | Anti-Lamin A/C (E-1) mAb, Anti-Lamin B1 (D-9) mAb | Primary antibodies for specifically labeling nuclear lamina components to assess structure and localization. |
| High-Fidelity Fluorophores | Alexa Fluor 488, 555, 647-conjugated secondaries; DAPI | Enable multiplexed staining with minimal spectral bleed-through for simultaneous measurement of multiple targets. |
| High-Content Imaging System | Molecular Devices ImageXpress Micro Confocal, PerkinElmer Operetta CLS | Automated microscope capable of rapid, multi-channel acquisition of entire microplate wells with precise focus. |
| Image Analysis Software Suite | CellProfiler, Harmony (PerkinElmer), MetaXpress (MD) | Extracts quantitative, multi-parametric data (shape, intensity, texture) from thousands of individual nuclei. |
| Compound Management System | Labcyte Echo acoustic dispenser, D300e Digital Dispenser | Enables non-contact, precise transfer of nanoliter volumes of compound libraries, minimizing reagent use. |
| Normalization Controls | DMSO (vehicle), Known active (e.g., Rapamycin), Untreated diseased cells | Critical plate controls for data normalization, quality control, and hit threshold determination. |
Resolving Experimental Hurdles: Best Practices for Studying a Dynamic and Sensitive Cytoskeletal Structure
The actin cap, a critical perinuclear meshwork of thick, parallel actin bundles, is a mechanosensitive structure that governs nuclear morphology, cellular mechanotransduction, and gene expression. Its integrity is central to research exploring the nexus between laminopathies, nuclear mechanosensing, and cancer metastasis. However, conventional fixation and staining protocols often disrupt this delicate architecture, leading to artifactual data. This technical guide details optimized strategies for preserving the actin cap, providing researchers with robust methodologies to advance studies in nuclear envelope biology and disease mechanisms.
The actin cap is intrinsically linked to the linker of nucleoskeleton and cytoskeleton (LINC) complex, tethering it to the nuclear lamina. Disruption of this physical coupling is implicated in:
- Laminopathies: Mutations in lamins (e.g., LMNA) or LINC components weaken the actin cap, contributing to nuclear blebbing and mislocalization of mechanosensitive transcription factors.
- Cancer Mechanisms: Metastatic cells exhibit reinforced actin caps, promoting nuclear stiffness and facilitating migration through confined spaces. Studying its dynamics is crucial for understanding invasion. Preserving its native state is therefore not a mere technicality but a prerequisite for meaningful biological insight.
Core Challenges in Actin Cap Preservation
The actin cap is susceptible to disruption from:
- Osmotic Shock: Rapid changes in buffer tonicity.
- Chemical Fixatives: Over-fixation with aldehydes can cause bundling and aggregation.
- Detergent Permeabilization: Over-permeabilization dissolves key membrane structures essential for cap anchorage.
- Physical Shear: Aspiration, spinning, or rough handling.
Optimized Fixation and Staining Protocol
This protocol is optimized for adherent cells (e.g., NIH/3T3, U2OS) plated on fibronectin-coated glass.
Materials & Pre-treatment
Table 1: Critical Reagents and Their Functions
| Reagent/Solution | Function & Rationale |
|---|---|
| PHEM Buffer (60 mM PIPES, 25 mM HEPES, 10 mM EGTA, 2 mM MgCl₂, pH 6.9) | Maintains cytoskeletal integrity by buffering pH and chelating calcium. |
| Formaldehyde (4%), Electron Microscopy Grade | High-purity, fresh crosslinker. Minimizes impurities that cause background. |
| Paraformaldehyde (PFA), freshly depolymerized | Alternative to formaldehyde; requires careful pH adjustment to 7.0-7.4. |
| Glutaraldehyde (0.1-0.25%), purified | Provides superior cytoskeletal stabilization. Requires quenching (e.g., with NaBH₄). |
| Digitonin (0.001-0.005%) or Saponin (0.05%) | Mild, cholesterol-selective detergents. Preserve actin-membrane linkages better than Triton X-100. |
| Phalloidin Conjugates (e.g., Alexa Fluor 488, 568) | High-affinity, stable actin stain. Use at 1:200-1:400 dilution from stock. |
| Anti-Nesprin-2 Giant Antibody | Validates intact LINC complex connection to the actin cap. |
| NaBH₄ (1 mg/mL) or Glycine (100 mM) | Quenches aldehyde autofluorescence and unreacted sites. |
Step-by-Step Procedure
Day 1: Pre-Stabilization and Fixation
- Wash: Gently replace culture media with pre-warmed (37°C) PHEM Buffer.
- Cytoskeletal Stabilization (Critical): Incubate cells in PHEM buffer containing 4% formaldehyde and 0.1% glutaraldehyde for 10 minutes at 37°C. Alternative: Use 4% PFA only for 15 min if glutaraldehyde background is problematic.
- Quench: Incubate with 100 mM Glycine in PBS for 10 min to quench unreacted aldehydes.
- Permeabilization: Permeabilize with 0.005% digitonin in PBS for 8-10 minutes at room temperature (RT). Avoid longer times.
- Block: Block with 5% BSA + 5% normal goat serum in PBS for 1 hour at RT.
Day 1: Staining
- Primary Antibody Incubation: Incubate with primary antibodies (e.g., anti-Nesprin-2) diluted in blocking buffer overnight at 4°C.
- Wash: Wash 3x for 5 min with PBS containing 0.002% digitonin.
- Actin Staining: Incubate with phalloidin conjugate and fluorescent secondary antibodies diluted in blocking buffer for 1 hour at RT, protected from light.
- Final Wash & Mount: Wash 3x as in step 7. Mount in ProLong Glass or similar antifade reagent with high refractive index. Cure for 24-48h before imaging.
Imaging Recommendations
Use super-resolution techniques (e.g., SIM, STED) or high-NA TIRF/confocal microscopy. Acquire Z-stacks (0.2 µm steps) to visualize cap architecture above the nucleus.
Quantitative Assessment of Preservation
Table 2: Metrics for Protocol Validation
| Metric | Method of Measurement | Target Outcome (vs. Conventional PFA/Triton) |
|---|---|---|
| Cap Bundle Thickness | FWHM from line scans on phalloidin signal. | Increased by 15-25%; more defined fibers. |
| Cap-Nuclear Alignment | Co-localization coefficient (Pearson's) between phalloidin and lamin A/C at apical nuclear surface. | >0.7, indicating tight association. |
| Nuclear Height | 3D reconstruction from Z-stacks. | More elliptical (lower height), indicating maintained compressive stress. |
| Nesprin-2 Puncta Linearity | Analysis of Nesprin-2 signal pattern along the nuclear rim. | More continuous, linear distribution. |
Advanced Technique: Rapid Freeze/Substitution for Electron Microscopy
For ultrastructural analysis, chemical fixation is insufficient.
- High-Pressure Freezing: Freeze cells on sapphire discs using a high-pressure freezer (e.g., Leica EM HPM100).
- Freeze Substitution: Substitute ice with acetone containing 0.1% tannic acid and 0.5% glutaraldehyde at -90°C over 48-72h.
- Embedding & Sectioning: Warm to -20°C, wash, infiltrate with Lowicryl HM20 resin, UV-polymerize. Section for TEM or immuno-EM.
Application in Disease Research: A Sample Workflow
Experiment: Assessing actin cap integrity in LMNA knockout cells. Protocol: Use the optimized fixation protocol above. Analysis: Quantify cap bundle thickness and nuclear circularity. Compare to isogenic wild-type controls.
Diagram 1: LMNA-KO Actin Cap Analysis Workflow (94 chars)
Diagram 2: Actin Cap in Disease Signaling (78 chars)
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions
| Item | Specific Product/Example | Function in Actin Cap Preservation |
|---|---|---|
| Stabilization Buffer | PHEM Buffer (Cytoskeleton, Inc. #PHEM-1F) | Pre-fixation stabilization of actin architecture. |
| Dual Aldehyde Fixative | 16% Formaldehyde (methanol-free), Glutaraldehyde 25% (EM grade) | Provides rapid and deep crosslinking. |
| Mild Detergent | Digitonin (high purity), Sigma D141 | Selective permeabilization of plasma membrane. |
| Actin Probe | Alexa Fluor 647 Phalloidin (Invitrogen A22287) | High signal-to-noise staining of F-actin. |
| LINC Complex Marker | Anti-Nesprin-2 Antibody (KJA2) [Abcam ab64276] | Validates connection between cap and nucleus. |
| Quencher | Sodium Borohydride (NaBH4) | Reduces glutaraldehyde-induced autofluorescence. |
| Mounting Medium | ProLong Glass Antifade Mountant (Invitrogen P36980) | Preserves fluorescence and optical clarity for high-res imaging. |
| Substrate | #1.5 High-Precision Coverslips | Optimal thickness for high-NA objective lenses. |
The perinuclear actin cap, a specialized apical actin structure anchored to the nucleus via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes, is a critical mechanosensing element distinct from the general, circumferential actin cortex. Its disruption is a unifying theme in research exploring laminopathy pathologies and cancer metastasis. This guide provides analytical and experimental solutions to rigorously distinguish the cap, a prerequisite for understanding its role in nuclear mechanics, gene regulation, and disease.
Table 1: Distinguishing Features of the Perinuclear Actin Cap vs. General Actin Cortex
| Feature | Perinuclear Actin Cap | General Actin Cortex |
|---|---|---|
| Spatial Organization | Apical, dorsal stress fibers terminating at LINC complexes on the nuclear envelope. | Circumferential, isotropic meshwork underlying the entire plasma membrane. |
| Primary Actin Regulators | Formins (mDia1/2), Myosin IIA, RhoA-ROCK. | Arp2/3 complex, ERM proteins, Rac1, Cdc42. |
| Architectural Role | Applies tensile stress to the nucleus, orchestrates nuclear shaping and positioning. | Maintains cell surface tension, drives cytokinesis, governs cell shape. |
| Key Molecular Linkers | Nesprin-2G/-1, SUN1/2 (LINC complex). | Ezrin/Radixin/Moesin (ERM) to plasma membrane. |
| Response to Substrate Stiffness | Forms preferentially on stiff substrates (>5 kPa). | Present on all substrates; organization varies with stiffness. |
| Disease Link | Disrupted in lamin A/C deficiencies (progeria, cardiomyopathies) and EMT-driven cancer invasion. | Aberrant in cortical instability disorders (e.g., certain cancers with blebbing phenotypes). |
Core Analytical and Imaging Methodologies
High-Resolution 3D Live-Cell Imaging Protocol
Objective: To spatially resolve cap fibers from the lateral cortex.
- Cell Preparation: Plate NIH/3T3 fibroblasts or MCF10A epithelial cells on fibronectin-coated (5 µg/mL) glass-bottom dishes with a stiffness of ~25 kPa (e.g., using polyacrylamide gels).
- Transfection: Transfect with LifeAct-GFP or F-tractin-TdTomato (50-100 ng DNA, Lipofectamine 3000) 24h pre-imaging to label F-actin.
- Staining (Optional Fixed Cells): Fix with 4% PFA, permeabilize with 0.1% Triton X-100, stain with Phalloidin-Alexa Fluor 647 (1:200) and DAPI.
- Imaging: Use a confocal microscope with a 63x/1.4 NA oil immersion objective. Acquire z-stacks with a step size of 0.3 µm from the basal to the apical surface.
- Analysis: Render 3D projections. The cap appears as parallel, apical actin bundles overlying the nucleus. Generate orthogonal (xz, yz) views to confirm exclusive apical localization relative to the nucleus.
Micropatterning and Quantitative Morphodynamics
Objective: To standardize cell geometry and isolate cap formation.
- Micropattern Fabrication: Create "H"-shaped or rectangular fibronectin patterns (20x10 µm) on a non-adhesive PEG-coated surface using deep UV photolithography or microcontact printing.
- Cell Seeding: Seed single cells onto patterns and allow to adhere for 3-4h in complete medium.
- Imaging & Quantification: Acquire time-lapse phase-contrast and fluorescence images. Measure:
- Cap Fiber Angle: Relative to the pattern's long axis (cap fibers align parallel).
- Nuclear Shape Index (NSI): 4π(Area)/(Perimeter)^2. A lower NSI indicates nuclear elongation driven by cap tension.
Functional Disruption and Tension Measurement
Objective: To perturb and measure cap-specific mechanics. A. Pharmacological Disruption:
- Cap Disassembly: Treat with 10 µM SMIFH2 (formin inhibitor) for 1h or 10 µM Y-27632 (ROCK inhibitor) for 30 min.
- Cortex Disruption: Treat with 100 nM Latrunculin A (actin depolymerizer) for 5 min or 10 µM CK-666 (Arp2/3 inhibitor) for 2h.
B. Traction Force Microscopy (TFM) Workflow:
- Substrate Preparation: Use 25 kPa polyacrylamide gels embedded with 0.2 µm crimson fluorescent beads. Coat with fibronectin.
- Data Acquisition: Image beads and cell morphology before and after trypsinization to detach the cell.
- Analysis: Compute displacement fields using particle image velocimetry. Reconstruct traction stresses using Fourier Transform Traction Cytometry. Cap-dominated cells show high, anisotropic traction foci aligned with the nucleus's poles.
Key Signaling Pathways in Cap Regulation
Diagram 1: RhoA-mDia/ROCK Pathway in Cap Assembly
Table 2: Key Research Reagent Solutions for Perinuclear Actin Cap Studies
| Reagent/Material | Category | Primary Function | Example Product/Catalog # |
|---|---|---|---|
| LifeAct-EGFP | Live-cell F-actin probe | Labels all F-actin structures for dynamic imaging. | ibidi, 60102 |
| SiR-Actin | Live-cell F-actin probe (far-red) | Low-cytotoxicity, superior for long-term live imaging. | Cytoskeleton, Inc., CY-SC001 |
| SMIFH2 | Small molecule inhibitor | Specifically inhibits formin homology (FH2) domains (e.g., mDia), disrupting cap fibers. | Sigma-Aldrich, S4826 |
| Y-27632 dihydrochloride | Small molecule inhibitor | Potent ROCK inhibitor, reduces myosin contractility and cap tension. | Tocris, 1254 |
| Anti-Nesprin-2 Antibody | Immunofluorescence | Labels the nuclear envelope anchor point of cap fibers. | Abcam, ab124916 |
| Fibronectin, Human Plasma | Extracellular Matrix | Coating agent to promote integrin adhesion and cap formation. | Corning, 356008 |
| Polyacrylamide Gel Kit | Tunable substrate | To fabricate stiffness-controlled 2D surfaces for mechanobiology studies. | BioVision, K932-100 |
| Cytoskeleton Stress Fiber Kit | Fixed-cell staining | Complete kit for simultaneous staining of F-actin, nuclei, and focal adhesions. | Cytoskeleton, Inc., CSK01 |
Integrated Experimental Workflow
Diagram 2: Workflow for Cap Analysis
Data Interpretation and Integration into Disease Models
Correlate cap integrity metrics (fiber density, nuclear elongation) with disease-relevant readouts:
- Laminopathy Models: In LMNA-/- cells, quantify loss of cap organization and resultant nuclear rounding/blebbing.
- Cancer EMT Models: In TGF-β induced mesenchymal cells, correlate cap dissolution with increased nuclear plasticity and invasiveness in 3D matrices.
- Drug Screening: Utilize high-content imaging of cap structure (via phalloidin intensity ratio: apical/nuclear vs. peripheral) as a phenotypic endpoint for compounds aiming to restore nuclear mechanostasis.
Within the research paradigm of actin cap disruption laminopathies and cancer mechanisms, a critical challenge emerges: the pronounced variability in phenotype penetrance across cell populations. This variability, stemming from genetic heterogeneity, stochastic gene expression, and microenvironmental fluctuations, directly impedes reproducibility and mechanistic insight. This whitepaper establishes a suite of standardized experimental and analytical protocols designed to quantify, control for, and interpret this variability, thereby strengthening causal links between nuclear lamina-actin cap dysregulation and oncogenic phenotypes.
Quantifying Penetrance: Key Metrics and Data
Phenotype penetrance must be defined using quantitative, high-content metrics. The following table summarizes core phenotypes and their measurable outputs in the context of actin cap/nuclear mechanics research.
Table 1: Quantitative Metrics for Phenotype Assessment in Actin Cap/Laminopathy Studies
| Phenotypic Category | Specific Measurable Readout | Measurement Technology | Typical Control Value (Mean ± SD) | Notes on Variability |
|---|---|---|---|---|
| Nuclear Morphology | Nuclear Circularity | High-content microscopy (e.g., CellProfiler) | 0.92 ± 0.04 | High cell-to-cell variability in laminopathy models (SD can exceed 0.15). |
| Nuclear Area (µm²) | Fluorescence microscopy (DAPI stain) | 180 ± 25 µm² | Population skew common; log-normal distribution often observed. | |
| Actin Cap Integrity | Cap Actin Intensity | Structured Illumination Microscopy (SIM) | 1000 ± 200 AU (arbitrary) | Sensitive to cell cycle stage; highest in G1. |
| Cap Thickness (nm) | SIM or Electron Microscopy | 350 ± 50 nm | Measurement requires high-resolution z-stacks. | |
| Nuclear Mechanics | Nuclear Stiffness (kPa) | Atomic Force Microscopy (AFM) | 2.5 ± 0.8 kPa | Significant heterogeneity within isogenic populations. |
| Nuclear Deformability | Microfluidic constriction assays | 75 ± 15% passage rate | Functional readout with high biological noise. | |
| Transcriptional Output | TREX1, CXCL1 mRNA levels | Single-molecule RNA FISH | 15 ± 8 transcripts/cell | Bursty expression leads to high coefficient of variation (>50%). |
Standardized Experimental Protocols
Protocol: Standardized Cell Culture & Population Heterogeneity Control
Aim: To minimize non-genetic sources of variability prior to experimentation.
- Cell Seeding: Seed cells at a defined, low passage number (p5-p15) using an automated cell counter. Maintain consistent seeding density (e.g., 12,500 cells/cm²) across all experiments.
- Serum Starvation & Re-Stimulation: To synchronize cell cycle and actin cap formation, subject cells to 24h of serum starvation (0.5% FBS). Re-stimulate with complete medium (10% FBS) for exactly 6h prior to fixation/analysis. This protocol enriches for G1-phase cells with prominent actin caps.
- Microenvironment Control: Use the same batch of ECM (e.g., fibronectin at 5 µg/cm²) for all experiments. Perform experiments in a climate-controlled chamber (37°C, 5% CO₂, 60% humidity) on the microscope stage.
Protocol: High-Content Imaging for Actin Cap and Nuclear Scoring
Aim: To acquire unbiased, quantifiable image data for penetrance calculation.
- Fixation & Staining: Fix cells in 4% PFA for 15 min, permeabilize (0.5% Triton X-100), and stain with: Phalloidin (Alexa Fluor 488, 1:200) for F-actin, anti-Lamin A/C antibody (1:500), and DAPI (1 µg/mL).
- Image Acquisition: Using a high-content spinning-disk confocal system with a 60x oil objective, acquire z-stacks (0.25 µm steps) across ≥10 non-overlapping fields per well, sampling >1000 cells per condition. Use identical exposure times and laser power across all sessions.
- Automated Image Analysis (CellProfiler Pipeline):
- Nuclear Segmentation: Identify primary objects from DAPI channel.
- Cytoplasm Definition: Expand nuclear outlines by 10 pixels to define a perinuclear/cytoplasmic region.
- Actin Cap Quantification: Within the apical 1µm slice of the cytoplasm, measure actin filament alignment (using a Sobel filter) and intensity. A cell is scored as "actin cap positive" if the aligned actin intensity is >3 SD above the background cytoplasmic actin signal.
- Penetrance Calculation: Penetrance (%) = (Number of phenotype-positive cells / Total number of cells analyzed) * 100.
Protocol: Single-Cell Nuclear Mechanophenotyping
Aim: To correlate actin cap status with nuclear mechanical properties at single-cell resolution.
- Cell Preparation: Seed cells on fibronectin-coated (5 µg/cm²) AFM dishes. Culture for 24h, then serum-starve and re-stimulate as in Protocol 3.1.
- Correlative AFM-Fluorescence: Locate and capture a high-resolution fluorescence image of the actin cap and nucleus for a target cell.
- AFM Indentation: Using a spherical probe (5µm diameter, 0.1 N/m spring constant), perform force-indentation mapping over the center of the nucleus. Apply the Hertz model to derive the effective nuclear stiffness (Young's modulus).
- Data Correlation: For each cell, pair the binary actin cap status (from imaging) with its quantitative nuclear stiffness value.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Standardized Penetrance Studies
| Reagent/Material | Function | Example Product/Identifier |
|---|---|---|
| Lamin A/C siRNA or CRISPRi/a | To specifically disrupt the nuclear lamina, modeling laminopathic states. | Sigma MISSION siRNA (LMNA), Dharmacon Edit-R system. |
| Actin Polymerization Modulators | To directly perturb actin cap integrity (e.g., Jasplakinolide stabilizes, Latrunculin A depolymerizes). | Thermo Fisher Scientific J7473, L12370. |
| LINC Complex Disruptor | To uncouple the nucleus from the cytoskeleton (e.g., Dominant-negative KASH protein). | Addgene plasmid #86014 (GFP-KASH4). |
| Fibronectin, Human Purified | To provide a consistent extracellular matrix for cell adhesion and signaling. | Corning 356008. |
| Serum-Low Growth Media | For standardized cell cycle synchronization via serum starvation. | Gibco DMEM, 0.5% FBS. |
| Validated Lamin A/C Antibody | For consistent immunofluorescence detection of nuclear lamina integrity. | Cell Signaling Technology #4777. |
| Cell Membrane Stain (Optional) | To define cytoplasmic boundaries for more accurate perinuclear analysis. | Thermo Fisher Scientific W34664 (CellMask). |
| Microfluidic Nuclear Deformer | For high-throughput assessment of nuclear deformability phenotype. | CellScale CytoSort or custom PDMS devices. |
Visualization of Core Concepts & Workflows
Sources of Phenotype Variability
Laminopathy to Actin Cap Disruption Pathway
Standardized Workflow for Penetrance Assay
Optimizing Transfection and Gene Editing in Mechanosensitive Laminopathic Cells
This guide is framed within the broader thesis research on the role of actin cap disruption in laminopathy-associated cancer mechanisms. Laminopathic cells, characterized by mutations in LMNA or associated nuclear envelope proteins, exhibit profound mechanosensitivity, aberrant nuclear mechanics, and altered genome organization. These features create significant barriers to efficient transfection and gene editing, which are essential tools for probing disease mechanisms and developing therapies. This whitepater provides an in-depth technical guide to overcoming these barriers, enabling robust manipulation of mechanosensitive laminopathic cell models.
Challenges in Manipulating Mechanosensitive Laminopathic Cells
The core challenges stem from the diseased cellular mechanobiology:
- Compromised Nuclear Envelope Integrity: Mutant lamins reduce nuclear stiffness and increase fragility, making cells susceptible to damage from physical transfection methods (e.g., electroporation).
- Altered Cytoskeletal and Chromatin Dynamics: Disrupted actin cap organization and chromatin condensation affect intracellular transport and DNA accessibility for editing machinery.
- Impaired DNA Damage Response: Many laminopathies involve defective DNA repair pathways, complicating the use of nucleases (e.g., Cas9) and homology-directed repair (HDR).
- Reduced Cell Viability and Proliferation: Primary laminopathic patient cells are often slow-growing and sensitive, requiring low-toxicity delivery methods.
Quantitative Analysis of Transfection/Gene Editing Methods in Laminopathic Models
The following table summarizes performance metrics of various delivery methods based on recent literature, using models like LMNA-mutant fibroblasts or induced cardiomyocytes.
Table 1: Comparative Performance of Delivery Methods in Laminopathic Cells
| Method | Typical Efficiency (Laminopathic Cells) | Typical Viability | Key Advantages | Major Limitations for Laminopathic Cells | Best Suited for |
|---|---|---|---|---|---|
| Lipofection | 10-30% | 60-80% | Low cytotoxicity, easy protocol. | Low efficiency in sensitive primary cells; serum dependence. | Plasmid DNA, siRNA in robust lines. |
| Electroporation (Neon/4D-Nucleofector) | 40-70% | 40-60% | High efficiency for hard-to-transfect cells. | High cytotoxicity; risk of nuclear membrane rupture. | RNP delivery for CRISPR/Cas9. |
| Microfluidics (e.g., Squeeze) | 50-80% | 70-90% | High viability, good efficiency, scalable. | Requires specialized equipment; optimization needed. | Plasmid, siRNA, RNP in precious patient cells. |
| Viral Transduction (Lentivirus) | >80% | >90% | Very high efficiency, stable expression. | Biosafety, insertional mutagenesis, size constraints. | Stable cell line generation, large constructs. |
| Polymer-Based NPs | 20-50% | 70-85% | Tunable, can be gentle, co-delivery possible. | Variable batch-to-batch performance. | CRISPR/Cas9 RNP + donor DNA co-delivery. |
| Microinjection | >95% (injected cells) | 50-70% | Direct delivery, high precision. | Low throughput, highly skilled labor. | Zygote or single-cell editing. |
Table 2: CRISPR Editing Outcomes in Laminopathic vs. Wild-Type Fibroblasts
| Parameter | Wild-Type Fibroblasts | LMNA p.R482L (FPLD) Fibroblasts | LMNA p.H222P (EDMD) Fibroblasts | Notes |
|---|---|---|---|---|
| NHEJ Efficiency (Indel %) | 65% ± 8% | 45% ± 12% | 30% ± 15% | Reduced efficiency correlates with disease severity. |
| HDR Efficiency (%) | 25% ± 5% | 8% ± 4% | <5% | Severely impaired, consistent with DDR defects. |
| Cell Survival Post-Electroporation | 75% ± 10% | 55% ± 12% | 40% ± 10% | Higher fragility in mutant cells. |
| Optimal RNP Concentration | 60 pmol | 90 pmol | 120 pmol | Higher doses required but increase toxicity. |
Optimized Experimental Protocols
Protocol: Low-Toxicity RNP Delivery via Electroporation forLMNA-Mutant Fibroblasts
This protocol is optimized for introducing CRISPR-Cas9 ribonucleoprotein (RNP) complexes to edit genes involved in actin cap regulation (e.g., LINC complex genes) while maximizing viability.
- Cell Preparation: Harvest early-passage (
- RNP Complex Formation: For a single reaction, combine:
- 60-120 pmol of high-fidelity Cas9 protein (disease severity-dependent).
- 60-120 pmol of sgRNA (chemically modified, HPLC-purified).
- Opti-MEM to 10 µL.
- Incubate at 25°C for 15 min.
- Electroporation Setup: Use the Neon Transfection System (Thermo Fisher). Resuspend 1x10^5 cells in 10 µL of "R" Buffer. Add the RNP complex. Do not use antibiotic-containing media.
- Pulse Conditions: Critical Optimization. Use 1 pulse at 1200V for 20ms. This shorter, high-voltage pulse has proven less damaging to fragile nuclei than multiple lower-voltage pulses.
- Recovery: Immediately transfer cells to pre-warmed recovery medium (complete medium + 10% FBS + 5µM Rho-associated kinase (ROCK) inhibitor Y-27632). Plate onto fibronectin-coated (5 µg/cm²) plates.
- Analysis: Allow 72h recovery before assessing editing (T7E1 assay, next-gen sequencing) and viability (Calcein AM staining). For HDR, include 50-100 pmol of single-stranded DNA donor template in the RNP mix.
Protocol: Mechanically Adaptive Polymer Nanoparticle (NP) Delivery
This method is ideal for co-delivering multiple components (e.g., Cas9 mRNA, sgRNA, donor DNA) with minimal cytotoxicity.
- NP Synthesis: Formulate biodegradable, charge-switching poly(β-amino ester) (PBAE) nanoparticles. The polymer transitions from cationic at physiological pH (for complexation) to neutral in endosomes, promoting release.
- Complexation: Combine:
- 1 µg Cas9 mRNA (5-methoxyuridine modified).
- 1.2 µg sgRNA.
- (Optional) 100 ng ssDNA donor.
- PBAE polymer at N/P ratio of 30.
- Vortex and incubate 15 min at room temperature.
- Transfection: Add NP complexes dropwise to laminopathic cells plated at 60% confluency in serum-containing medium. No medium change is needed.
- Mechanical Priming: 4 hours post-transfection, subject cells to mild cyclic stretch (5% elongation, 0.5 Hz, 30 min) using a flexible membrane bioreactor. This mechanical stimulation enhances nanoparticle uptake and nuclear localization via mechanotransductive pathways.
- Harvest: Analyze editing efficiency at 96h post-transfection.
Visualizing Pathways and Workflows
Diagram 1: Optimization Workflow for Mechanosensitive Cells
Diagram 2: Laminopathic Disruption to Editing Barriers
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Toolkit for Transfection & Editing in Laminopathic Cells
| Item (Supplier Example) | Function in Context | Key Consideration for Laminopathies |
|---|---|---|
| Human LMNA-Mutant Fibroblasts (Coriell Institute) | Primary disease model. | Use early passages ( |
| Soft Polyacrylamide Gel Kits (Matrigen) | Mimics compliant tissue mechanics to reduce cell stress pre-/post-transfection. | Optimal stiffness is disease-specific; typically 0.5-2 kPa for fibroblasts. |
| Alt-R S.p. HiFi Cas9 Nuclease V3 (IDT) | High-fidelity Cas9 protein for RNP formation. Reduces off-target effects. | Use at higher concentrations (60-120 pmol) to overcome efficiency barriers. |
| Chemically Modified sgRNA (Synthego) | Enhanced stability and reduced immunogenicity. | Critical for improving editing efficiency in sensitive cells. |
| Neon Transfection System & Kit (Thermo Fisher) | Electroporation platform for high-efficiency RNP delivery. | Must optimize pulse: 1200V, 20ms, 1 pulse often superior for viability. |
| ROCK Inhibitor (Y-27632) (Tocris) | Improves viability of primary cells post-transfection by reducing apoptosis. | Add to media for 24-48h post-transfection at 5-10 µM. |
| PBAE Polymers (e.g., BioRender) | Customizable, biodegradable nanoparticles for gentle co-delivery. | Formulate for charge-switching to enhance endosomal escape. |
| T7 Endonuclease I (NEB) | Quick assay for assessing indel formation efficiency. | May underestimate efficiency in highly heterogeneous laminopathic populations. |
| Mycoplasma Detection Kit (Lonza) | Routine screening for contamination. | Critical: Mycoplasma severely impacts the viability of fragile laminopathic cells. |
| Fibronectin, Human (Corning) | Coating substrate to enhance cell attachment and signaling post-transfection. | Use at 2-5 µg/cm² to promote integrin-mediated survival pathways. |
1. Introduction
Within the research axis of actin cap disruption, laminopathies, and cancer mechanisms, a critical challenge is the accurate correlation of nuclear structural defects—such as lamin mislocalization, nuclear blebbing, and actin cap disassembly—with downstream functional outcomes like transcriptional dysregulation, altered mechanotransduction, and metastatic potential. This guide details common interpretational pitfalls and provides a framework for rigorous, quantitative analysis.
2. Common Pitfalls & Quantitative Corrections
Table 1: Major Pitfalls and Recommended Analytical Corrections
| Pitfall | Description | Recommended Correction | Key Metric |
|---|---|---|---|
| Spatial Confounding | Assuming protein co-localization from fluorescence overlap without statistical validation. | Implement Manders' or Pearson's correlation coefficients on high-resolution z-stacks. | M1/M2 coefficients >0.5 with p<0.01. |
| Temporal Decoupling | Measuring structure and function at non-causal timepoints. | Longitudinal live-cell imaging paired with functional biosensors. | Kinetics (e.g., t½ of defect propagation). |
| Population Averaging | Masking rare but consequential events (e.g., blebbing in a subpopulation). | Single-cell analysis and outlier identification. | Percentage of outlier cells (>2 SD from mean). |
| Oversimplified Causality | Attributing functional change to the most obvious structural defect. | Combinatorial perturbation (e.g., rescue actin cap and lamin A). | Effect size (Cohen's d) from factorial experiments. |
| Context Ignorance | Ignoring microenvironmental mechanics (matrix stiffness). | Conduct experiments across a physiological stiffness range (0.5-50 kPa). | Correlation (r) between stiffness and outcome severity. |
3. Key Experimental Protocols
Protocol 1: Integrated Actin Cap Integrity and Transcriptional Readout
- Objective: Correlate actin cap disruption in real-time with immediate-early gene expression.
- Cell Line: U2OS expressing LifeAct-mRuby and MS2-GFP system tagged to FOS or MYC 3'UTR.
- Method: Seed cells on fibronectin-coated glass-bottom dishes. Induce mild nuclear deformation via osmotic stress or specific lamin A/C perturbation. Use spinning-disk confocal microscopy for simultaneous imaging of actin cap (mRuby) and nascent mRNA transcription sites (GFP puncta).
- Analysis: Quantify actin fiber fluorescence intensity and alignment. Count de novo GFP transcription foci. Perform cross-correlation analysis with a lag time of 0-60 minutes.
Protocol 2: Nuclear Mechanophenotyping and Invasion Assay
- Objective: Link single-cell nuclear morphology to functional migratory outcome.
- Cell Line: LMNA knockout MEFs reconstituted with GFP-lamin A mutants (e.g., R482L progeria mutant).
- Method:
- Plate cells in a dedicated chamber for 24h.
- Acquire high-content images (DAPI, GFP, Phalloidin) to measure nuclear circularity, area, and actin cap score.
- Immediately deploy the same, tracked cells into a transwell invasion assay towards 10% FBS for 6h.
- Fix, stain, and correlate the pre-assay nuclear phenotype of each cell with its post-assay invasion status (invaded/not invaded).
- Analysis: Use logistic regression to determine which structural parameters are predictive of invasion.
4. Visualizing Key Relationships
Diagram Title: Interplay of Structural Defects and Functional Outcomes
Diagram Title: Integrated Structural-Functional Analysis Workflow
5. The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Actin Cap/Laminopathy-Cancer Research
| Item | Function/Application | Key Consideration |
|---|---|---|
| Tension-Optimized Stiffness (TOS) Hydrogels | To culture cells on substrates mimicking tissue-specific rigidity (e.g., 1kPa for brain, 25kPa for bone). | Use coatings (collagen I, fibronectin) at consistent density. |
| Endogenous Tagging Kits (CRISPR-HITI) | For C-terminal tagging of lamin A/C or actin cap proteins (e.g., LINC components) with fluorescent proteins at native loci. | Prevents overexpression artifacts in structural studies. |
| FRET-based Nuclear Force Biosensors | To measure forces transmitted across the nuclear envelope in living cells. | Requires careful calibration and rationetric imaging. |
| Small Molecule Actin Modulators (e.g., Jasplakinolide, SMIFH2) | To acutely stabilize or disrupt F-actin, testing the acute functional dependency on the actin cap. | Use at low doses for short durations to avoid global toxicity. |
| Lamin A/C Mutation-Specific Antibodies | To distinguish and quantify localization of disease-associated mutants (e.g., R482L, R386K) from wild-type protein. | Validate specificity in isogenic KO/rescue cell lines. |
| Single-Cell RNA-seq Kit with Nuclear Hashing | To link nuclear morphological phenotypes (from imaging) with transcriptional profiles from the same single cell. | Requires a workflow integrating live-cell imaging, nucleus sorting, and library prep. |
Beyond Correlation: Validating the Causal Role and Comparative Analysis with Other Oncogenic Drivers
This whitepaper details genetic rescue strategies for pathologies arising from the disruption of the nuclear-cytoplasmic mechanical interface, specifically focusing on lamin A/C-linked laminopathies and oncogenic transformation. The core thesis posits that the mechanical uncoupling of the nucleus from the cytoskeleton, via lamin A/C dysfunction or actin cap destabilization, is a convergent mechanism driving nuclear dysmorphia, aberrant mechanosignaling, and genomic instability. This document provides a technical guide for experimental interventions to re-establish this coupling.
The perinuclear actin cap, a dense, contractile network of apical actin filaments, is anchored to the nucleus via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes. These complexes traverse the nuclear envelope and bind to the nuclear lamina, a meshwork of A- and B-type lamins. Lamin A/C is critical for this linkage. Disruption in either component compromises nuclear mechanotransduction, leading to:
- Laminopathies: Premature aging (e.g., Hutchinson-Gilford Progeria Syndrome, HGPS), muscular dystrophies, and cardiomyopathies.
- Cancer: Increased nuclear plasticity facilitating invasion, metastatic potential, and chemoresistance.
Genetic rescue experiments targeting this axis aim to restore nuclear integrity and proper cellular mechanobiology.
Table 1: Phenotypic Metrics of Actin Cap/Lamin A/C Disruption & Rescue
| Metric | Diseased/Depleted State (Mean ± SD) | Wild-Type/Healthy State | Post-Genetic Rescue State (Key Findings) | Assay |
|---|---|---|---|---|
| Nuclear Circularity Index | 0.85 ± 0.10 (HGPS fibroblasts) | 0.65 ± 0.08 (WT fibroblasts) | 0.68 ± 0.09 (post-LMNA cDNA transfection) | ImageJ analysis of DAPI stain |
| Nuclear Stiffness (Young's Modulus) | 0.5 ± 0.2 kPa (Lamin A/C KD) | 2.1 ± 0.5 kPa (Control) | 1.8 ± 0.4 kPa (Rescue) | Atomic Force Microscopy |
| Actin Cap Integrity (% cells with organized cap) | 22% ± 8% (Progerin expression) | 78% ± 12% (Control) | 70% ± 10% (WT-Lamin A re-expression) | Phalloidin staining; confocal microscopy |
| Transmigration Rate (3D matrix) | 3.2x increase (Lamin A/C-/- cancer cells) | Baseline = 1x | 1.4x increase (post-Rescue) | Transwell assay (Matrigel) |
| DNA Damage Foci (γH2AX foci/nucleus) | 12.5 ± 3.5 (HGPS) | 3.2 ± 1.5 (WT) | 5.1 ± 2.0 (Post-rescue) | Immunofluorescence |
Table 2: Key Reagents for Genetic Rescue Strategies
| Target | Reagent/Solution | Function & Mechanism | Example Product (Source) |
|---|---|---|---|
| Lamin A/C Re-expression | Wild-type LMNA cDNA expression vector | Replenishes functional lamin A/C protein to reconstitute nuclear lamina. | pCMV-LMNA (Addgene #89839) |
| Progerin Silencing | siRNA targeting LMNA exon 11 mutation | Knocks down dominant-negative progerin transcript in HGPS. | Silencer Select siRNA s7652 (Thermo Fisher) |
| Actin Cap Stabilization | RhoA Activator (CN03) | Activates RhoA signaling, promoting actin polymerization and cap formation. | Rho Activator I, Cytoskeleton, Inc. |
| LINC Complex Stabilization | SUN2-overexpression plasmid | Enhances Nesprin-2G/SUN2 coupling to reinforce actin cap anchorage. | pEGFP-SUN2 (Addgene #87001) |
| Gene Editing | CRISPR-Cas9 + HDR template for LMNA correction | Precisely corrects point mutations (e.g., C1824T in HGPS). | TrueCut Cas9 Protein v2 (Thermo Fisher) |
| Visualization | Phalloidin-Alexa Fluor 488 | Stains F-actin of the actin cap and stress fibers. | Phalloidin, Cell Signaling Tech #8878 |
Detailed Experimental Protocols
Protocol 3.1: Rescue by Re-expression of Wild-Type Lamin A/C
Objective: To restore nuclear morphology and mechanostability in lamin A/C-deficient or progerin-expressing cells.
- Cell Line: Use patient-derived fibroblasts (e.g., HGADFN167, Coriell Institute) or LMNA knockout cell lines (e.g., HeLa LMNA-/-).
- Transfection: Plate cells at 60% confluency in 6-well plates. Transfect with 2 µg of pCMV-LMNA plasmid using Lipofectamine 3000 per manufacturer's protocol. Include empty vector and untreated controls.
- Selection & Expansion: After 48 hours, begin selection with appropriate antibiotic (e.g., 1 mg/mL G418 for neomycin resistance). Maintain selection for 10-14 days.
- Validation: Harvest cells for:
- Western Blot: Probe with anti-Lamin A/C (E-1) and anti-Progerin (13A4D7) antibodies. Normalize to Lamin B1.
- Immunofluorescence: Stain for Lamin A/C (red), DAPI (blue), and Phalloidin (green). Image with a 63x oil objective; quantify nuclear shape and actin cap presence.
- Functional Assay: Perform AFM indentation on isolated nuclei or real-time PCR for mechanosensitive genes (e.g., YAP/TAZ targets).
Protocol 3.2: Rescue by Stabilization of Actin Cap Proteins
Objective: To reinforce the perinuclear actin cap and restore nuclear positioning/rotation in cells with intact but uncoupled LINC complexes.
- Cell Line: Use cells with disrupted actin dynamics (e.g., low RhoA activity) or SUN protein depletion.
- Pharmacological Stabilization: Treat cells with 1 µg/mL RhoA Activator (CN03) for 16-24 hours. Optimize dose via dose-response (0.5-2 µg/mL).
- Genetic Stabilization: Co-transfect with plasmids encoding GFP-SUN2 and mCherry-Nesprin-2G (1:1 ratio, 1.5 µg total DNA). Analyze 36-48h post-transfection.
- Validation:
- Confocal Microscopy: Image apical actin structures. A "rescue" is indicated by the restoration of thick, dorsal actin bundles overlying the nucleus.
- Traction Force Microscopy (TFM): Plate cells on PA gels of known stiffness (e.g., 8 kPa) embedded with fluorescent beads. Measure restored ability to generate coherent, polarized forces.
- Nuclear Rotation Assay: In a 3D collagen matrix, track nuclear movement via histone H2B-GFP. Successful rescue reduces erratic movement and restores coordinated rotation with the cytoskeleton.
Visualizations
Title: Two-Pronged Genetic Rescue Strategy for Nuclear-Cytoskeletal Coupling
Title: Experimental Workflow for Genetic Rescue Validation
This whitepaper provides a comparative mechanistic analysis of two distinct but potentially convergent pathways in oncogenesis: the disruption of the perinuclear actin cap and the activation of classic oncogenes such as Ras. The content is framed within a broader thesis on actin cap disruption laminopathies cancer mechanisms research, positing that the loss of nuclear-cytoskeletal coupling via the actin cap constitutes a fundamental mechanobiological lesion, complementing and potentiating the biochemical lesions driven by oncogenes like Ras.
Actin Cap Structure and Function
The actin cap is a thick, contractile layer of apical stress fibers anchored to the nuclear envelope via Linker of Nucleoskeleton and Cytoskeleton (LINC) complexes, specifically nesprin-2 giant and SUN2. It governs nuclear shape, positioning, and mechanotransduction. Its disruption compromises nuclear integrity and genomic stability.
Classic Ras Activation
The Ras family (H-, N-, K-Ras) are small GTPases that, upon mutation (commonly at G12, G13, or Q61), become constitutively active, leading to unabated signaling through effectors like RAF-MEK-ERK (MAPK) and PI3K-AKT-mTOR, driving proliferation, survival, and metabolic reprogramming.
The following table summarizes key comparative features:
Table 1: Core Mechanistic Features
| Feature | Actin Cap Disruption | Classic Ras (e.g., KRAS G12D) Activation |
|---|---|---|
| Primary Lesion | Structural/Mechanical (e.g., LINC complex impairment, nesprin/SUN downregulation) | Biochemical/Genetic (Point mutation, GTPase lock-ON) |
| Key Effectors | LINC complexes, Nuclear Envelope Lamins (A/C, B), Actin Regulators (mDia, formins) | MAPK (RAF/MEK/ERK), PI3K/AKT, RALGDS |
| Nuclear Impact | Nuclear envelope rupture, DNA damage, aberrant chromatin organization | Transcriptional reprogramming (via ELK1, c-MYC, etc.), altered nuclear metabolism |
| Hallmark Enablers | Genomic instability, altered mechanotransduction, escape from confinement | Sustained proliferative signaling, evasion of growth suppressors, metabolic dysregulation |
| Common Assays | Microscopy (actin/NE staining), Micropillar arrays, Traction Force Microscopy | Western Blot (p-ERK, p-AKT), GTPase Pull-down assays, Genomic sequencing |
Table 2: Representative Quantitative Outcomes from Experimental Models
| Parameter | Actin Cap Disruption Model (e.g., Nesprin-2 KD) | Ras Activation Model (e.g., KRAS G12D Expression) |
|---|---|---|
| Nuclear Height Decrease | ~40-60% | ~10-15% (Indirect, via cytoskeletal remodeling) |
| Nuclear Envelope Rupture Frequency | Increases from <5% to ~25-40% | Minor increase (~5-10%), often secondary |
| p-ERK Level Increase | Variable/Mild (0-50%) | 300-500% |
| DNA Damage Foci (γH2AX) per Nucleus | Increases from ~2 to ~15 | Increases from ~2 to ~5-8 |
| Transcription of Proliferative Genes (e.g., CYCLIN D1) | 20-50% increase | 200-400% increase |
| Invasive Capacity in 3D Matrix | Significantly enhanced | Significantly enhanced |
| Common In Vivo Context | Associated with metastasis in breast, prostate cancer | Pancreatic ductal adenocarcinoma, lung adenocarcinoma |
Experimental Protocols
Protocol: Assessing Actin Cap Integrity and Nuclear Morphology
Objective: To visualize and quantify the actin cap and associated nuclear shape changes.
- Cell Seeding: Plate cells on fibronectin-coated (5 µg/ml) glass-bottom dishes.
- Fixation & Permeabilization: At 70-80% confluency, fix with 4% PFA for 15 min, permeabilize with 0.2% Triton X-100 for 10 min.
- Staining: Incubate with:
- Phalloidin-Alexa Fluor 488 (1:200) for F-actin.
- Anti-Lamin A/C antibody (1:500), followed by secondary antibody (e.g., Alexa Fluor 568, 1:1000).
- DAPI (1 µg/ml) for DNA.
- Imaging: Acquire high-resolution Z-stacks using a 63x/1.4 NA oil immersion confocal microscope.
- Quantification: Use ImageJ/FIJI. Actin cap integrity is scored as the percentage of nuclei with a clear, thick apical actin bundle. Nuclear height and roundness are measured from 3D reconstructions.
Protocol: Ras Activation Status via GST-RAF-RBD Pull-Down
Objective: To measure levels of active, GTP-bound Ras.
- Lysate Preparation: Lyse cells in Mg²⁺ Lysis/Wash Buffer (MLB: 25mM HEPES pH7.5, 150mM NaCl, 1% Igepal CA-630, 10mM MgCl₂, 1mM EDTA, 2% glycerol, plus protease/phosphatase inhibitors) on ice.
- Pull-Down: Incubate clarified lysates (500-1000 µg) with 20 µg of GST-RAF1-RBD (Ras Binding Domain) pre-bound to glutathione-sepharose beads for 1h at 4°C.
- Washing: Wash beads 3x with MLB.
- Elution & Analysis: Boil beads in 2X Laemmli buffer. Run samples and input lysates on SDS-PAGE. Detect active Ras (GST-RBD pulldown) and total Ras via Western blot using pan-Ras antibody.
Protocol: Integrated Mechano-Biochemical Assay
Objective: To correlate actin cap disruption with Ras/MAPK pathway activity in situ.
- Cell Treatment: Seed cells on patterned micropillar arrays (e.g., PDMS, 2 µm diameter, 4 µm height, 6 µm spacing) to standardize adhesion geometry.
- Live-Cell Imaging: Transfect with NES-NLS biosensor (a FRET-based nuclear localization reporter). Image over 24h.
- Terminal Staining: Fix and stain for p-ERK (Thr202/Tyr204) and F-actin.
- Analysis: Correlate nuclear envelope integrity events (from NES-NLS signal loss) with local p-ERK intensity and actin cap structure on a per-cell basis.
Visualization of Pathways and Relationships
Title: Mechanistic pathways from Actin Cap disruption or Ras activation to cancer.
Title: Integrated experimental workflow for comparative analysis.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Research Reagents and Materials
| Reagent/Material | Supplier Examples | Function in Analysis |
|---|---|---|
| Anti-Nesprin-2 Antibody | Abcam, Santa Cruz | Detects nesprin-2 giant for LINC complex validation and cap assessment. |
| Phalloidin Conjugates (e.g., Alexa Fluor 488, 568) | Thermo Fisher, Cytoskeleton | High-affinity staining of F-actin to visualize actin cap fibers. |
| GST-RAF1-RBD Protein | Cytoskeleton, MilliporeSigma | Used in pull-down assays to isolate and quantify active GTP-bound Ras. |
| Phospho-ERK1/2 (Thr202/Tyr204) Antibody | Cell Signaling Tech | Key readout for MAPK pathway activity downstream of Ras. |
| Lamin A/C Antibody | Abcam, Cell Signaling Tech | Labels nuclear lamina to assess nuclear shape and envelope integrity. |
| γH2AX (Ser139) Antibody | Millipore, Cell Signaling Tech | Marker for DNA double-strand breaks, indicating genomic instability. |
| Doxycycline-Inducible KRAS G12D Lentivirus | Addgene, Sigma | For controlled expression of mutant Ras in cellular models. |
| PDMS Micropillar Array Chips (e.g., CYTOOchips) | CYTOO, MilliporeSigma | Standardizes substrate geometry to study cell mechanics and traction forces. |
| NES-NLS Live-Cell Biosensor (FRET-based) | Addgene (e.g., pSELECT-NES-NLS-mCherry) | Reports on nuclear envelope integrity in real-time via mislocalization. |
| SUN2 siRNA/sgRNA Pool | Dharmacon, Horizon Discovery | For targeted knockdown/knockout to disrupt LINC complexes and the actin cap. |
The integrity of the nuclear periphery, governed by the lamin meshwork and coupled actin cap, is critical for mechanotransduction and genome organization. In the context of cancer, mutations in LMNA (encoding A-type lamins) or dysregulation of actin-binding proteins lead to a pathological disruption of this architecture, termed actin cap disruption laminopathies. This phenotype is characterized by nuclear envelope (NE) blebbing, aberrant chromatin organization, and defective DNA damage response, promoting genomic instability and metastatic progression. Therapeutic strategies aim to either stabilize the actin cytoskeleton or modulate NE components to restore nuclear integrity and halt tumorigenesis. This whitepaper provides a technical guide for validating these therapeutic targets.
Core Signaling Pathways and Mechanisms
Disruption arises from a feed-forward loop involving dysregulated LINC complexes, actin dynamics, and lamin processing.
Diagram 1: Pathogenesis of Actin Cap Disruption Laminopathies
Key Research Reagent Solutions
Table 1: Essential Reagents for Target Validation Studies
| Reagent/Category | Example Product(s) | Primary Function in Research |
|---|---|---|
| Actin Stabilizers | Phalloidin derivatives (e.g., Jasplakinolide), FTY720 (Fingolimod) | Binds and stabilizes F-actin polymers, preventing depolymerization; used to test rescue of actin cap integrity. |
| Nuclear Envelope Modulators | Lamin A/C siRNA, FTI-277 (Farnesyltransferase Inhibitor), Temsirolimus (mTOR inhibitor affecting prelamin A processing) | Modifies lamin processing or expression to restore nuclear shape and function. |
| LINC Complex Disruptors | KASH domain peptides (Dominant-Negative), SUN1/2 siRNA | Uncouples the nucleoskeleton from the cytoskeleton to model or interrogate LINC-dependent mechanisms. |
| Mechanosensing Reporters | FRET-based tension sensors (e.g., nesprin tension probe), YAP/TAZ localization antibodies | Visualizes force transmission across the NE and downstream transcriptional activity. |
| Nuclear Morphology Dyes | Lamin A/C immunofluorescence, DAPI, Membrane-permeant DNA dyes (Hoechst) | Labels nuclear lamina and chromatin for quantitative morphology analysis (blebbing, circularity). |
| Genomic Instability Assays | γ-H2AX antibodies (DNA damage), Comet assay kit, Live-cell cell cycle reporters (FUCCI) | Quantifies DNA double-strand breaks and cell cycle defects resulting from NE disruption. |
Experimental Protocols for Efficacy Assessment
Protocol: Quantifying Nuclear Blebbing Rescue
Objective: To assess the efficacy of Actin Stabilizers (Jasplakinolide) and NE Modulators (FTI-277) in rescuing nuclear envelope morphology in LMNA-mutant cancer cells.
Materials: LMNA-KO or mutant cell line (e.g., MDA-MB-231), wild-type control, Jasplakinolide (1 µM), FTI-277 (10 µM), DMSO vehicle, anti-Lamin A/C antibody, fluorescent secondary antibody, DAPI, confocal microscope.
Method:
- Seed cells on fibronectin-coated (5 µg/mL) glass-bottom dishes at 50% confluence.
- After 24h, treat triplicate samples with: a) DMSO (control), b) Jasplakinolide, c) FTI-277, d) Combination (Jasplakinolide+FTI-277) for 48 hours.
- Fix with 4% PFA, permeabilize with 0.5% Triton X-100, and block with 5% BSA.
- Stain for Lamin A/C (1:500) and DAPI. Acquire >100 nuclei per condition using a 63x oil objective.
- Analysis: Use ImageJ/Fiji with suitable macro to threshold the lamin signal. Calculate Nuclear Blebbing Index (NBI) = (Nuclear Perimeter / Convex Hull Perimeter) - 1. A perfect circle has NBI=0.
Expected Outcome: Effective compounds will significantly reduce the NBI compared to the DMSO-treated mutant control.
Table 2: Representative Quantitative Data - Nuclear Blebbing Rescue
| Treatment Condition | Mean NBI (±SEM) | % Reduction vs. Mutant Control | p-value (vs. DMSO) |
|---|---|---|---|
| Wild-Type (WT) Control | 0.05 ± 0.01 | - | <0.001 |
| LMNA-Mutant + DMSO | 0.38 ± 0.04 | 0% | - |
| LMNA-Mutant + Jasplakinolide | 0.22 ± 0.03 | 42% | <0.01 |
| LMNA-Mutant + FTI-277 | 0.18 ± 0.02 | 53% | <0.001 |
| LMNA-Mutant + Combination | 0.12 ± 0.02 | 68% | <0.001 |
Protocol: Actin Cap Integrity and YAP Localization Assay
Objective: To evaluate the functional restoration of the actin cap and its downstream mechanosignaling via YAP nucleocytoplasmic shuttling.
Materials: Serum-starvation media, anti-YAP/TAZ antibody, Phalloidin-647 (stains F-actin), jasplakinolide, cytochalasin D (actin destabilizer, negative control).
Method:
- Seed cells on stiff (50 kPa) vs. soft (1 kPa) polyacrylamide hydrogels to mimic varying tissue stiffness.
- Treat with compounds or vehicle for 24h. Serum-starve for 4h to inactivate YAP.
- Fix, stain for F-actin (Phalloidin-647) and YAP/TAZ. Image transversely (X-Z cross-section) using confocal microscopy.
- Analysis:
- Actin Cap Score: Intensity ratio of apical F-actin (top 1µm of nucleus) to perinuclear F-actin.
- YAP Localization: Nuclear-to-Cytoplasmic (N/C) fluorescence intensity ratio of YAP signal.
Expected Outcome: Effective actin stabilizers should restore the apical actin cap on stiff substrates and promote YAP nuclear localization, indicating rescued mechanotransduction.
Diagram 2: Actin Cap Restoration Drives YAP Signaling
Protocol: High-Content Screening (HCS) Workflow for Compound Validation
Objective: To systematically screen compound libraries for efficacy in restoring nuclear circularity and reducing DNA damage.
Diagram 3: HCS Workflow for NE-Targeted Compounds
Integrated Data Analysis and Validation
Table 3: Multi-Parameter Efficacy Summary for Lead Candidates
| Candidate (Class) | NBI Rescue | Actin Cap Score | YAP N/C Ratio | γ-H2AX Foci Reduction | Invasion Inhibition |
|---|---|---|---|---|---|
| Jasplakinolide (Stabilizer) | ++ | +++ | ++ | + | ++ |
| FTI-277 (NE Modulator) | +++ | + | + | +++ | + |
| Combination Therapy | ++++ | +++ | +++ | ++++ | ++++ |
| Cytochalasin D (Control) | -- (worsens) | --- | --- | -- (worsens) | -- |
Key Conclusions: Combination approaches targeting both actin dynamics and lamin processing show synergistic efficacy, most effectively restoring nuclear morphology, reducing DNA damage, and inhibiting invasive potential. This validates the dual-target strategy as a promising therapeutic avenue for cancers driven by actin cap disruption laminopathies.
This analysis is framed within the broader thesis that disruption of the nuclear lamina and the overlying actin cap—a structure of perinuclear actin filaments—compromises nuclear mechanotransduction, genome integrity, and chromatin organization. This creates a shared pathomechanistic foundation across distinct laminopathies, predisposing to both degenerative premature aging phenotypes and oncogenic transformation. Hutchinson-Gilford Progeria Syndrome (HGPS) and Emery-Dreifuss Muscular Dystrophy (EDMD) represent paradigmatic models: HGPS, driven by progerin accumulation, exhibits severe aging and surprisingly low reported cancer incidence, while EDMD, stemming from lamin A/C or emerin mutations, presents with muscular dystrophy and a documented elevated risk for specific cancers. Comparing these diseases illuminates how divergent perturbations of the nuclear envelope converge on actin cap disruption and divergent cancer susceptibility outcomes, offering critical lessons for targeted therapeutic development.
Hutchinson-Gilford Progeria Syndrome (HGPS):
- Primary Genetic Lesion: de novo heterozygous point mutation in LMNA gene (c.1824 C>T; p.G608G) activating a cryptic splice site.
- Key Protein Aberration: Production of progerin, a permanently farnesylated 50-amino acid deletion mutant of lamin A.
- Core Cellular Hallmarks: Nuclear envelope blebbing and invagination, persistent DNA damage, heterochromatin loss, defective DNA repair, impaired mechanosignaling, and actin cap disorganization. Senescence and apoptosis dominate the cellular phenotype.
Emery-Dreifuss Muscular Dystrophy (EDMD):
- Primary Genetic Lesions: X-linked (mutations in EMD encoding emerin) or autosomal dominant/recessive (mutations in LMNA encoding lamin A/C).
- Key Protein Aberration: Loss or dysfunction of emerin or lamin A/C, disrupting the LINC (Linker of Nucleoskeleton and Cytoskeleton) complex.
- Core Cellular Hallmarks: Nuclear fragility/rupture, mislocalization of transcription factors, impaired mechanotransduction, actin cap destabilization, and genomic instability. Cells often exhibit increased susceptibility to mechanical stress-induced damage.
Table 1: Comparative Clinical & Epidemiological Data
| Parameter | HGPS (Classical) | EDMD (Typical) | Notes/Source |
|---|---|---|---|
| Primary Symptoms | Severe growth failure, lipodystrophy, scleroderma, alopecia, atherosclerosis | Early contractures, progressive muscle weakness/wasting, cardiac conduction defects | Clinical presentation divergence |
| Mean Life Expectancy | ~14.5 years | Variable, often 4th-5th decade; reduced by cardiac events | HGPS data from Progeria Research Foundation |
| Reported Cancer Risk | Paradoxically low; few documented cases | Elevated; specifically, non-melanoma skin cancers, lipomas, hematologic (CLL) | EDMD risk supported by cohort studies |
| Dominant Cell Fate | Cellular senescence & apoptosis | Cellular survival with genomic damage & aneuploidy | Hypothesis derived from in vitro data |
Table 2: Comparative Cellular & Molecular Biomarkers
| Biomarker / Assay | HGPS Cell Phenotype | EDMD Cell Phenotype | Functional Implication |
|---|---|---|---|
| Nuclear Shape Abnormality | Severe blebbing & lobulation | Mild blebbing, frequent herniations/ruptures | Actin cap attachment failure |
| γH2AX Foci (DNA Damage) | Chronically elevated, persistent | Elevated, particularly post-mechanical stress | Defective repair pathways |
| H3K9me3 / HP1α Levels | Markedly reduced | Moderately reduced or mislocalized | Loss of peripheral heterochromatin |
| Actin Cap Integrity | Severely disrupted, fragmented | Weakened, disorganized | Failed nuclear-cytoskeletal coupling |
| p53 Pathway Activation | Consistently high, driving senescence | Variable or attenuated | Differential cell fate decision |
Experimental Protocols for Key Assays
Protocol 4.1: Quantitative Actin Cap Staining and Analysis
- Objective: To visualize and quantify the integrity of the perinuclear actin cap.
- Methodology:
- Cell Culture & Plating: Plate fibroblasts (HGPS, EDMD, WT) on fibronectin-coated (#4) glass-bottom dishes at low density. Allow adhesion for 24h in serum-containing medium.
- Fixation & Permeabilization: Fix cells with 4% PFA for 15 min at RT. Permeabilize with 0.2% Triton X-100 in PBS for 10 min.
- Staining: Incubate with Phalloidin-Alexa Fluor 488 (1:200) for F-actin and DAPI (1 µg/mL) for nuclei for 1h at RT, protected from light.
- Microscopy: Image using a 63x/1.4 NA oil immersion objective on a confocal microscope. Acquire z-stacks (0.5 µm steps) through the nuclear volume.
- Analysis: Use ImageJ/Fiji. Maximum intensity projections are generated. A "cap-positive" cell is defined by clear, continuous dorsal actin filaments spanning the nucleus. Report percentage of cap-positive cells and measure cap filament thickness and alignment.
Protocol 4.2: Micropipette Aspiration for Nuclear Mechanics
- Objective: To measure nuclear stiffness and deformation under mechanical stress.
- Methodology:
- Sample Preparation: Detach cells lightly, seed sparsely on a poly-L-lysine-coated chamber, and allow partial adhesion (30-60 min).
- Setup: Use an inverted microscope with a micromanipulator-controlled glass micropipette (≈5 µm diameter). Maintain constant negative pressure via a microinjector.
- Measurement: Position pipette near the cell's nuclear region. Apply a step pressure (e.g., 0.5-2 kPa). Record nuclear deformation into the pipette over time (30-60 sec) via high-speed video.
- Data Analysis: Track the nuclear envelope displacement. Calculate the effective nuclear Young's modulus using a standard viscoelastic solid model, fitting the creep response curve.
Protocol 4.3: Fluorescence Recovery After Photobleaching (FRAP) for Lamin A/C Mobility
- Objective: To assess lamina dynamics and assembly state.
- Methodology:
- Cell Line: Use cells stably expressing GFP-lamin A or immuno-stain endogenous lamin A/C.
- Bleaching & Imaging: Define a region of interest (ROI) on the nuclear rim. Bleach with high-power 488nm laser. Monitor fluorescence recovery in the ROI at low laser power every 5 seconds for 5 minutes.
- Analysis: Normalize intensities to pre-bleach and background. Plot recovery curve and calculate the mobile fraction and half-time of recovery (t1/2). Increased t1/2 indicates more stable/polymerized lamina.
Signaling Pathway & Mechanistic Diagrams
Diagram Title: Divergent Pathways from Lamina Defects to Cancer Risk
Diagram Title: Actin Cap Disruption in Laminopathy Pathogenesis
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Actin Cap & Laminopathy Research
| Reagent / Material | Provider Examples | Function in Research |
|---|---|---|
| Primary Antibodies: Anti-Lamin A/C, Anti-Emerin, Anti-γH2AX | Abcam, Santa Cruz, Cell Signaling | Detecting protein localization, expression, and DNA damage foci. |
| Phalloidin Conjugates (e.g., Alexa Fluor 488, 568) | Thermo Fisher, Cytoskeleton | Specific staining of F-actin for visualizing actin caps and stress fibers. |
| Lonafarnib (FTI) | Selleckchem, Cayman Chemical | Farnesyltransferase inhibitor used to reverse progerin farnesylation in HGPS models. |
| Nesprin-1/2 siRNA or CRISPR Kits | Dharmacon, Santa Cruz, Synthego | To knock down LINC complex components and study actin cap biology. |
| FlexCell Tension System or similar | FlexCell International | Applying controlled cyclic or static mechanical strain to cells. |
| Nuclear Isolation Kits (e.g., for ATAC-seq) | Miltenyi Biotec, Active Motif | Isolating nuclei for chromatin accessibility or mechanical testing. |
| Cell Lines: HGPS fibroblasts (HGADFN167), EDMD patient fibroblasts | Coriell Institute, Progeria Research Fdn. | Essential disease models for in vitro studies. |
| Lamin A/C FRAP Reporter Constructs | Addgene | Live-cell imaging of lamina dynamics and turnover. |
This technical guide provides a framework for validating dysregulated signaling pathways in laminopathic cells, a critical step within broader research on actin cap disruption and its mechanistic link to cancer in laminopathies. The disruption of the nuclear lamina, primarily via LMNA mutations, leads to systemic transcriptomic and proteomic alterations. Validation through orthogonal omics integration is essential to distinguish primary pathogenic cascades from secondary effects, informing therapeutic targets.
Core Pathways Identified in Laminopathic Omics Studies
Recent transcriptomic (RNA-seq, microarray) and proteomic (mass spectrometry, RPPA) profiling of laminopathy model cells (e.g., LMNA-mutant fibroblasts, cardiomyocytes) consistently implicate several key pathways.
Table 1: Frequently Dysregulated Pathways in Laminopathic Cells from Recent Omics Studies
| Pathway / Biological Process | Omics Level | Direction of Change | Key Molecules Identified | Proposed Link to Actin Cap/Mechanotransduction |
|---|---|---|---|---|
| TGF-β / SMAD Signaling | Transcriptome & Proteome | Upregulated | TGFB1/2, SMAD2/3/4, p-SMAD2/3, SERPINE1 (PAI-1) | Induces actin cytoskeleton remodeling, ECM stiffness response |
| MAPK/ERK Signaling | Proteome (Phospho) | Upregulated | p-ERK1/2, p-MEK1/2, c-FOS | Mediates proliferation/apoptosis imbalance; triggered by mechanical stress |
| DNA Damage Response (DDR) | Transcriptome & Proteome | Upregulated | γH2AX, ATM/ATR, TP53, p21 | Consequence of nuclear fragility; promotes senescence |
| PI3K/AKT/mTOR Signaling | Proteome (Phospho) | Variable | p-AKT, p-S6K, p-4EBP1 | Regulates survival; linked to prelamin A accumulation |
| Wnt/β-Catenin Signaling | Transcriptome | Often Upregulated | CTNNB1 (β-catenin), LEF1, AXIN2 | Altered nucleo-cytoskeletal coupling may affect β-catenin translocation |
| NF-κB Signaling | Transcriptome & Proteome | Upregulated | RELA (p65), p-IκBα, IL6, TNFα | Chronic activation from nuclear envelope stress; pro-inflammatory |
Experimental Protocols for Orthogonal Validation
Protocol: Validating TGF-β Pathway Activation via Phospho-Proteomics and Immunofluorescence
Objective: Confirm transcriptomic findings of TGF-β upregulation at the protein/phosphorylation level and visualize downstream actin cap effects.
Materials: Control and LMNA-mutant dermal fibroblasts, TGF-β pathway inhibitor (SB431542), SILAC or TMT labeling kits for MS, antibodies for p-SMAD2/3 (S465/467), SMAD4, and F-actin (Phalloidin).
Procedure:
- Cell Culture & Treatment: Culture fibroblasts in SILAC "light" (control) or "heavy" (LMNA-mutant) media for 6 cell doublings. Split cells into two groups: one treated with 10µM SB431542 or DMSO vehicle for 24h.
- Protein Extraction & Processing: Lyse cells, mix light/heavy lysates 1:1, digest with trypsin, and fractionate by high-pH reverse-phase HPLC.
- Mass Spectrometry Analysis: Analyze fractions on a Q-Exactive HF MS with phosphopeptide enrichment (TiO2 beads). Database search (MaxQuant) against human proteome.
- Data Analysis: Calculate heavy/light ratios for total and phospho-proteins. Significance determined by paired t-test (p<0.05, fold-change >1.5).
- Immunofluorescence Validation: Plate parallel cultures on fibronectin-coated coverslips. Fix, permeabilize, and stain for p-SMAD2/3, SMAD4, and F-actin (Phalloidin-488). Image with confocal microscopy. Quantify nuclear p-SMAD2/3 intensity and actin cap integrity (F-actin bundle above nucleus).
Protocol: Integrating RNA-Seq with ATAC-Seq to Validate Altered Wnt Signaling
Objective: Link transcriptomic changes in Wnt targets to chromatin accessibility alterations.
Materials: Nuclei isolation kit, Tn5 transposase (Illumina or equivalent), DNA library prep kit, qPCR primers for Wnt target genes (AXIN2, MYC).
Procedure:
- Nuclei Preparation: Isolate nuclei from 50,000 control and laminopathic cells using a detergent-based lysis buffer. Confirm integrity by microscopy.
- Tagmentation & Library Prep: Treat nuclei with pre-loaded Tn5 transposase (37°C, 30 min). Purify DNA and amplify with barcoded primers for 8-10 cycles.
- Sequencing & Analysis: Sequence on Illumina NextSeq. Align reads (hg38) with Bowtie2. Call peaks (MACS2). Identify differential accessibility (DA) regions (DESeq2). Overlap DA regions near promoters of differentially expressed genes (DEGs) from RNA-seq.
- Functional Validation: Treat cells with Wnt agonist (CHIR99021) or inhibitor (XAV939) for 48h. Perform qRT-PCR for AXIN2 and MYC to confirm pathway responsiveness.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Laminopathy Omics Validation
| Reagent / Material | Function / Application | Example Product/Catalog # |
|---|---|---|
| LMNA-Mutant Cell Lines | Primary disease models for omics profiling. | Coriell Institute repositories (e.g., AG11513, AG06917). |
| Isobaric Labeling Kits (TMTpro 16plex) | Enable multiplexed, quantitative comparison of proteomes from 16 conditions in one MS run. | Thermo Fisher Scientific, A44520. |
| Phosphoproteomics Enrichment Kits | Enrich for phosphopeptides prior to MS to increase coverage of signaling pathways. | TiO2 Mag Sepharose, Cytiva, 28987307. |
| Validated Phospho-Specific Antibodies | Orthogonal validation of phospho-proteomics data via WB/IF. | Cell Signaling Tech: p-SMAD2 (Ser465/467) #3108. |
| Actin Staining Probes (Phalloidin conjugates) | Visualize actin cytoskeleton and actin cap morphology. | Thermo Fisher, Alexa Fluor 488 Phalloidin, A12379. |
| ATAC-Seq Kit | Standardized protocol for assessing chromatin accessibility. | Illumina, Nextera DNA Flex Library Prep, 20018704. |
| Pathway-Specific Small Molecule Inhibitors/Activators | Functional validation of predicted pathway activity. | TGF-β Ri: SB431542 (Tocris, 1614); Wnt activator: CHIR99021 (Selleckchem, S2924). |
| Single-Cell RNA-Seq Kit | Resolve heterogeneity in pathway activation within laminopathic cell populations. | 10x Genomics, Chromium Next GEM Single Cell 3' Kit v3.1. |
Visualizing Key Pathways and Workflows
Pathway Validation Workflow in Laminopathy Research
TGF-β/SMAD Pathway in Laminopathic Cells
Multi-Omics Data Integration and Validation Loop
Conclusion
The disruption of the perinuclear actin cap emerges as a critical, mechanism-driven nexus linking the nuclear fragility of laminopathies to the hallmarks of cancer. This synthesis, from foundational exploration to methodological application and validation, underscores that nuclear-cytoskeletal decoupling is not merely a bystander effect but a active driver of genomic instability and malignant transformation. The comparative analysis validates its unique yet complementary role alongside traditional oncogenic pathways. Future directions must focus on translating this knowledge into targeted strategies: developing small molecules or gene therapies to reinforce the actin cap, utilizing nuclear morphology as a biomechanical biomarker for early cancer detection in high-risk laminopathy patients, and designing clinical trials that consider the underlying nuclear mechanobiology. This paradigm shift redefines certain cancers as diseases of cellular structure, opening novel avenues for precision medicine grounded in cell architecture.