EB1 Protein: The Master Regulator of Actin-Microtubule Crosstalk in Cell Dynamics and Disease
This article provides a comprehensive exploration of the EB1 protein's pivotal role in mediating crosstalk between the actin and microtubule cytoskeletal networks.
EB1 Protein: The Master Regulator of Actin-Microtubule Crosstalk in Cell Dynamics and Disease
Abstract
This article provides a comprehensive exploration of the EB1 protein's pivotal role in mediating crosstalk between the actin and microtubule cytoskeletal networks. Aimed at researchers, scientists, and drug development professionals, we begin by establishing the foundational biology of EB1 as a plus-end tracking protein and its molecular mechanisms for bridging cytoskeletal systems. We then detail current methodological approaches for studying EB1 function, from advanced microscopy to in vitro reconstitution assays. The discussion extends to common experimental challenges and optimization strategies for perturbing EB1 activity. Finally, we evaluate and compare EB1's role against other cytoskeletal linkers, validating its unique position as a central node in cellular architecture, motility, and division, with direct implications for understanding metastasis and developing novel therapeutic interventions.
EB1 Protein 101: Unveiling the Molecular Bridge Between Actin and Microtubules
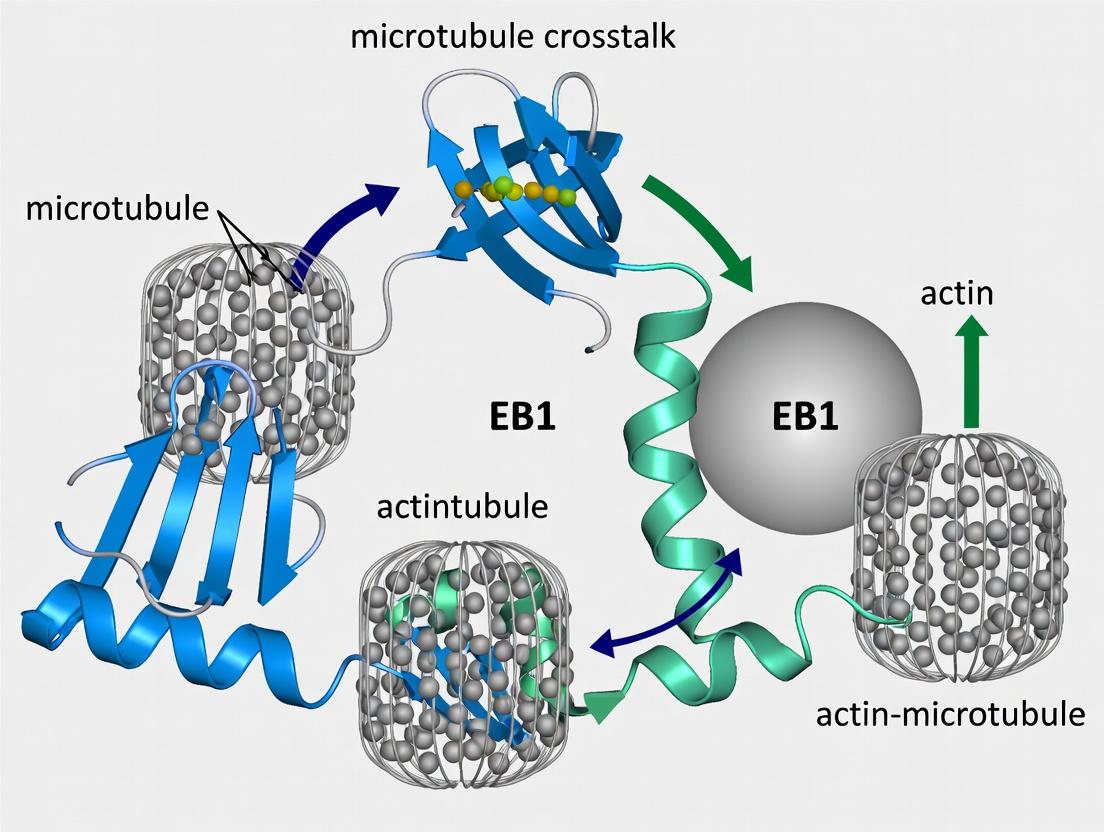
EB1 (End Binding protein 1) is the principal and best-characterized member of the +TIP family, a group of proteins that dynamically localize to the growing plus ends of microtubules. This whitepaper defines EB1's canonical role as the central hub for +TIP complex formation and situates its function within the critical, emerging field of actin-microtubule crosstalk. The broader thesis posits that EB1 is not merely a passive marker of microtubule dynamics but a decisive regulatory node that integrates cytoskeletal networks, influencing cell polarization, migration, and intracellular transport—processes fundamental to development, neuronal function, and cancer metastasis. Understanding EB1's molecular interactions is therefore pivotal for dissecting cytoskeletal coordination and identifying novel therapeutic targets.
Structural and Functional Basis of EB1 as a +TIP Hub
EB1 autonomously tracks microtubule plus ends through its N-terminal Calponin Homology (CH) domain, which binds the structural GTP cap of growing microtubules. Its C-terminal tail contains two coiled-coil motifs and an acidic EEY/F motif, which serves as a universal recruitment platform for other +TIPs via their CAP-Gly or SxIP domains.
Table 1: Core Structural Domains of EB1 and Their Functions
| Domain/Region | Amino Acid Residues (Human EB1) | Key Function | Interaction Partners |
|---|---|---|---|
| N-terminal CH Domain | 1-133 | Binds microtubule lattice/GTP cap | α/β-tubulin dimers |
| Linker Region | 134-191 | Flexibility, regulatory modifications | Various kinases |
| Coiled-coil 1 (CC1) | 192-251 | Dimerization, structural core | EB1 homodimer |
| Coiled-coil 2 (CC2) | 252-268 | Interaction with some partners | APC, p150glued |
| C-terminal tail (EEY/F) | 269-268 | Canonical +TIP recruitment platform | CAP-Gly proteins (e.g., p150glued), SxIP motif proteins (e.g., APC, MACF) |
EB1 in Actin-Microtubule Crosstalk: Mechanisms and Pathways
EB1 facilitates cytoskeletal crosstalk through direct and indirect mechanisms, positioning it as a central mediator.
Direct Mechanisms: Certain +TIPs recruited by EB1, such as the formin mDia3 and the spectraplakin MACF, possess dual affinity for both microtubule ends and actin filaments. EB1 localization brings these cross-linkers to specific cellular locations, enabling directed actin remodeling.
Indirect Mechanisms: EB1-guided microtubules deliver signaling cargo (e.g., RhoGEFs) to the cell cortex, locally activating Rho GTPases (Rac1, Cdc42, RhoA) that subsequently orchestrate actin nucleation and polymerization.
Diagram 1: EB1-mediated pathways in actin-microtubule crosstalk.
Key Experimental Protocols for Studying EB1 Function
Live-Cell Imaging of EB1 Comets
Purpose: To visualize and quantify microtubule growth dynamics. Protocol:
- Cell Transfection: Transfect cells with a plasmid encoding EB1 tagged with a fluorescent protein (e.g., EB3-GFP, the preferred isoform for live imaging due to brightness).
- Image Acquisition: Use a spinning-disk or TIRF microscope equipped with an environmental chamber (37°C, 5% CO₂). Acquire time-lapse images at 1-3 second intervals for 2-5 minutes.
- Analysis: Use tracking software (e.g., TrackMate in Fiji/ImageJ, u-Track) to detect comet positions over time. Key extracted parameters include:
- Growth Speed (µm/min)
- Growth Lifetime (s)
- Comet Density (number per unit area)
- Directional Persistence
In Vitro Reconstitution of +TIP Coupling
Purpose: To demonstrate direct, EB1-dependent recruitment of +TIPs to microtubule ends. Protocol:
- Protein Purification: Purify recombinant EB1 (full-length) and a candidate +TIP (e.g., a fragment containing the SxIP motif) using bacterial or baculovirus expression systems.
- Microtubule Polymerization: Prepare rhodamine-labeled, GMPCPP-stabilized microtubule "seeds" and mix with unlabeled tubulin in a BRB80 buffer + 1 mM GTP to initiate dynamic growth.
- Imaging Chamber Assembly: Flow in the growth mixture into a flow chamber passivated with casein.
- Protein Addition: Introduce a solution containing fluorescently labeled EB1 and the candidate +TIP (labeled with a different fluorophore).
- TIRF Microscopy: Image using dual-color TIRF microscopy. EB1 will decorate growing ends; positive coupling is shown by coincident localization of the +TIP at EB1-positive ends.
Functional Disruption via siRNA/CRISPR
Purpose: To probe the cellular consequences of EB1 loss-of-function. Protocol:
- Knockdown/Knockout: Deliver EB1-targeting siRNA or CRISPR/Cas9 constructs to cells.
- Validation: Confirm depletion by western blot (using anti-EB1 antibody, e.g., clone 5/EB1) and loss of comets by immunofluorescence.
- Phenotypic Analysis:
- Wound Healing/Transwell Assay: Quantify cell migration defects.
- Immunofluorescence: Stain for actin (phalloidin) and microtubules (anti-α-tubulin). Assess cell polarization and cytoskeletal architecture.
- FRET Biosensors: Use Rho GTPase biosensors to measure localized activity changes.
Table 2: Key Quantitative Parameters of EB1 and Microtubule Dynamics
| Parameter | Typical Value/Range | Measurement Technique | Biological Significance |
|---|---|---|---|
| EB1 Comet Tracking Speed | 15 - 25 µm/min | Live-cell imaging of EB1-GFP | Proxy for microtubule growth rate. |
| EB1 Comet Lifetime | 5 - 15 seconds | Live-cell imaging, persistence analysis | Indicates duration of sustained growth. |
| EB1-MT Binding Affinity (Kd) | ~0.2 - 0.5 µM | In vitro TIRF assay, fluorescence anisotropy | Measures strength of core +TIP interaction. |
| Increase in MT Catastrophe Frequency upon EB1 KD | 50 - 150% | Live imaging of MTs post-siRNA | Demonstrates EB1's role in promoting growth stability. |
| Reduction in Directed Cell Migration Speed upon EB1 KD | 40 - 70% | Wound healing / single-cell tracking | Highlights role in cytoskeletal coordination for motility. |
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for EB1/+TIP Research
| Reagent/Solution | Function/Application | Example Product/Catalog # (Illustrative) |
|---|---|---|
| Anti-EB1 Antibody | Immunofluorescence, Western Blot to localize/quantify endogenous EB1. | Mouse mAb, clone 5/EB1 (BD Biosciences, 610535). |
| EB1/EB3 Fluorescent Protein Constructs | Live-cell imaging of microtubule plus-end dynamics. | pmApple-EB3-7 (Addgene, 54920). |
| Cell Light Tubulin-GFP/RFP BacMam 2.0 | Labeling the entire microtubule network for crosstalk studies. | Thermo Fisher Scientific, C10613. |
| SiRNA for EB1 (MAPRE1) | Transient knockdown for loss-of-function studies. | SMARTpool: ON-TARGETplus MAPRE1 siRNA (Dharmacon). |
| Paclitaxel (Taxol) & Nocodazole | Microtubule-stabilizing and -depolymerizing drugs for perturbation experiments. | Sigma-Aldrich, T7191 & M1404. |
| Latrunculin A/B | Actin-depolymerizing agent to dissect actin's role in EB1-mediated processes. | Cayman Chemical, 10010630. |
| GMPCPP (Non-hydrolyzable GTP analog) | Generating stable microtubule "seeds" for in vitro reconstitution assays. | Jena Bioscience, NU-405S. |
| Rho GTPase Activity Assays (G-LISA) | Quantifying activation of downstream signaling pathways (Rac1, Cdc42, RhoA). | Cytoskeleton, Inc., BK series. |
Diagram 2: Experimental workflow for EB1 research.
EB1 is definitively established as the canonical +TIP, serving as the master regulator of the plus-end interactome. Its function as a linchpin in actin-microtubule crosstalk underscores a sophisticated role in cellular integration beyond simple tracking. Future research, leveraging high-resolution cryo-EM, optogenetics, and advanced biosensors, will focus on spatiotemporal mapping of the EB1 interactome in vivo and deciphering how dysregulation of these pathways contributes to disease. Targeting the EB1 interface with specific +TIPs presents a promising, albeit challenging, avenue for therapeutic intervention in cancers characterized by aberrant cytoskeletal dynamics and metastasis.
Within the context of actin-microtubule crosstalk research, the End-Binding protein 1 (EB1) family emerges as a central node, orchestrating interactions through its distinct structural domains. EB1 proteins, key regulators of microtubule plus-end dynamics, facilitate cytoskeletal coordination by recruiting a diverse array of binding partners. This recruitment is critically mediated by three primary structural regions: the Calponin Homology (CH) domain, the EB Homology (EBH) domain, and the flexible C-terminal Tail region. Understanding the precise molecular interactions governed by these domains is fundamental for dissecting mechanisms of cellular division, polarization, and migration, with significant implications for therapeutic intervention in diseases such as cancer.
Structural and Functional Domains of EB1
EB1 is a modular protein where each domain confers specific binding capabilities. The N-terminal CH domain is responsible for microtubule lattice binding and plus-end tracking. The central EBH domain forms a coiled-coil dimerization interface and contributes to partner recognition. The C-terminal Tail, ending with an acidic EEY/F motif, is highly flexible and serves as a major interaction platform. The table below summarizes key quantitative data on domain structures and interactions.
Table 1: Structural and Biophysical Properties of EB1 Domains
| Domain | Residue Range (Human EB1) | Key Structural Features | Primary Function | Representative Binding Partners |
|---|---|---|---|---|
| CH Domain | 1-133 | 4-helix bundle, hydrophobic cap | Microtubule end binding, lattice affinity | Tubulin dimer, microtubule lattice |
| EBH Domain | 134-191 | Coiled-coil, dimeric parallel helix | Dimerization, partner recognition | APC, MACF, Spectraplakins |
| Linker Region | 192-208 | Flexible, serine-rich | Connects EBH to Tail; regulatory sites | CDK1, Plk1 (phospho-regulation) |
| Tail Region | 209-268 | Intrinsically disordered, acidic C-terminus | High-affinity partner scaffold | CAP-Gly domains (e.g., CLIP-170, p150^Glued), SxIP motif proteins |
| C-terminal Motif | 266-268 | -EEY/F (conserved) | Critical for CAP-Gly domain binding | Essential for +TIP network assembly |
Detailed Experimental Protocols for Domain-Partner Analysis
Protocol 1: Surface Plasmon Resonance (SPR) for Tail Domain Binding Kinetics Objective: Quantify the binding affinity between the EB1 C-terminal Tail peptide and a CAP-Gly domain.
- Reagent Preparation: Immobilize recombinant human p150^Glued CAP-Gly domain on a CMS sensor chip via amine coupling to achieve ~5000 Response Units (RU).
- Analyte Series: Prepare a dilution series (0.1 nM – 1 µM) of a synthetic biotinylated EB1 Tail peptide (residues 240-268) in HBS-EP buffer (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.005% v/v Surfactant P20, pH 7.4).
- Binding Assay: Inject analyte over the chip surface at a flow rate of 30 µL/min for 120 s (association), followed by buffer flow for 300 s (dissociation). Regenerate the surface with a 30 s pulse of 10 mM Glycine-HCl, pH 2.0.
- Data Analysis: Subtract signals from a reference flow cell. Fit the resulting sensorgrams globally to a 1:1 Langmuir binding model using the Biacore evaluation software to determine the association rate (kon), dissociation rate (koff), and equilibrium dissociation constant (KD).
Protocol 2: Co-immunoprecipitation (Co-IP) for EBH Domain Interactions Objective: Validate the interaction between the EB1 EBH domain and Adenomatous Polyposis Coli (APC) in cell lysate.
- Cell Lysis: Harvest HEK293T cells co-transfected with GFP-tagged full-length EB1 and FLAG-tagged APC-C-terminal fragment. Lyse cells in IP lysis buffer (25 mM Tris-HCl pH 7.4, 150 mM NaCl, 1% NP-40, 1 mM EDTA, 5% glycerol, plus protease inhibitors) on ice for 30 min.
- Pre-clearing: Centrifuge lysate at 16,000 x g for 15 min. Incubate supernatant with Protein A/G agarose beads for 30 min at 4°C to pre-clear.
- Immunoprecipitation: Incubate 500 µg of pre-cleared lysate with 2 µg of anti-GFP antibody overnight at 4°C with gentle rotation. Add 50 µL of Protein A/G bead slurry and incubate for 2 hours.
- Wash & Elution: Pellet beads and wash 4x with ice-cold lysis buffer. Elute bound proteins by boiling in 2X Laemmli sample buffer for 5 min.
- Analysis: Resolve eluates by SDS-PAGE and perform immunoblotting with anti-FLAG (for APC) and anti-GFP (for EB1) antibodies.
Visualization of EB1 Domain Interaction Networks
Title: EB1 Domain Architecture and Binding Partner Network
Title: Experimental Workflow for EB1 Domain-Partner Analysis
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for EB1 Domain Interaction Studies
| Reagent / Material | Function & Application | Example Product / Identifier |
|---|---|---|
| Recombinant EB1 Domains (GST/His-tagged) | Purified protein fragments for in vitro binding assays (SPR, ITC) and crystallization. | Human EB1 (1-268), EB1 (1-191; ΔTail), EB1 (134-268); available from cDNA repositories. |
| SxIP Motif Peptide Libraries | Synthetic peptides to probe Tail domain interactions, study binding specificity and affinity. | Biotinylated LxxPTPhxSxIP motif peptides; custom synthesis services. |
| Anti-EB1 Monoclonal Antibodies | For immunoprecipitation, Western blotting, and immunofluorescence to detect endogenous EB1. | Clone 5/EB1 (BD Transduction Labs) for IF; Clone A-11 (Santa Cruz) for IP/WB. |
| CAP-Gly Domain Constructs | Key binding partners for the EB1 acidic tail; used as analyte or ligand in binding assays. | p150^Glued CAP-Gly (residues 1-200); CLIP-170 CAP-Gly (1-300). |
| Microtubule-Associated Protein Kits | Pre-formed, stabilized microtubules for co-sedimentation assays to test CH domain binding. | Cytoskeleton Inc. MT Binding Protein Spin-Down Assay Kit (BK029). |
| Live-Cell +TIP Reporter Constructs | Fluorescently tagged EB1 (e.g., EB3-GFP) to visualize comet formation and partner recruitment in vivo. | EB3-GFP plasmid (Addgene #39299) for live-cell imaging of microtubule dynamics. |
| Kinase Inhibitors (CDK1, Plk1) | To study phospho-regulation of the serine-rich linker region and its impact on partner binding. | RO-3306 (CDK1 inhibitor); BI-2536 (Plk1 inhibitor). |
The structural dissection of EB1 into its CH, EBH, and Tail regions provides a critical framework for understanding its master regulatory role at microtubule ends. Each domain engages a specific subset of effectors, thereby integrating signals to coordinate microtubule dynamics with actin networks and cellular structures. In the broader thesis of actin-microtubule crosstalk, mapping these interactions with quantitative and structural precision, as outlined in this guide, is indispensable. This knowledge base not only advances fundamental cytoskeletal biology but also illuminates potential targets for disrupting pathological processes in metastasis and aneuploidy, guiding future drug development efforts.
Within the critical cellular processes of division, polarization, and migration, the coordinated interplay between the actin cytoskeleton and microtubules (MTs) is paramount. This actin-microtubule crosstalk is dynamically regulated by a suite of specialized adapter proteins. Central to this regulatory network is End-Binding 1 (EB1), a master regulator of MT plus-end dynamics. EB1 does not act alone; it functions as a core scaffold, recruiting a "cast of characters" to MT tips to execute specific downstream functions. This whitepaper provides an in-depth technical examination of EB1's four principal binding partners—Adenomatous Polyposis Coli (APC), Cytoplasmic Linker Proteins (CLIPs), Spectraplakins, and Formins—framed within the broader thesis that EB1-mediated partner recruitment is the fundamental mechanism for spatiotemporal control in actin-MT crosstalk.
EB1: The Master Scaffold at MT Plus-Ends
EB1 proteins are highly conserved core components of the microtubule plus-end tracking protein (+TIP) network. They autonomously recognize and bind to the GTP-bound "cap" of growing MTs via a Calponin-Homology (CH) domain. Their C-terminal coiled-coil and acidic EEY/F tail domain serves as a versatile docking platform for a plethora of partners via CAP-Gly or SxIP motif interactions. This positions EB1 as the quintessential hub for plus-end localized complex assembly.
The Key Binding Partners: Functions & Quantitative Data
Adenomatous Polyposis Coli (APC)
APC is a large multifunctional tumor suppressor that interacts with EB1 via a C-terminal basic domain, binding to EB1's acidic tail. It stabilizes MTs and links them to cortical actin networks and signaling complexes.
Table 1: Quantitative Data for APC-EB1 Interaction & Function
| Parameter | Value / Observation | Experimental System | Reference (Example) |
|---|---|---|---|
| Binding Affinity (Kd) | ~0.5 - 2 µM (for C-terminal APC domain to EB1) | ITC / SPR | (Honnappa et al., 2005) |
| MT Stabilization Effect | Increases MT growth lifetime by ~40% | In vitro reconstitution | (Kita et al., 2006) |
| Cortical Dwell Time | ~4-6 seconds at cell cortex | TIRF microscopy in migrating cells | (Okada et al., 2010) |
| Role in Directed Migration | Knockdown reduces persistent migration by >60% | Wound-healing assay | (Kroboth et al., 2007) |
Cytoplasmic Linker Proteins (CLIP-170, CLIP-115)
CLIPs are prototypical +TIPs featuring N-terminal CAP-Gly domains that bind the EB1 tail. They act as early adapters, facilitating the recruitment of other factors and linking MT ends to organelles and kinetochores.
Table 2: Quantitative Data for CLIP-EB1 Interaction & Function
| Parameter | Value / Observation | Experimental System | Reference (Example) |
|---|---|---|---|
| Binding Affinity (Kd) | ~0.3 µM (CAP-Gly to EB1 tail) | ITC | (Honnappa et al., 2006) |
| Processive Run Length | Tracks growing MT end for ~30-40 sec | TIRF microscopy in vitro | (Bieling et al., 2008) |
| Organelle Tethering | Co-localization coefficient >0.7 with endosomes | Live-cell imaging | (Pierce et al., 2019) |
| Kinetochore Recruitment | Accumulates at kinetochores ~2 min prior to attachment | Mitotic cells | (Tanenbaum et al., 2006) |
Spectraplakins (e.g., MACF1, BPAG1)
Spectraplakins are giant cytoskeletal integrators containing both MT-binding (often via EB1) and actin-binding domains (ABD). They are prime executors of actin-MT crosstalk, directly tethering the two networks.
Table 3: Quantitative Data for Spectraplakin-EB1 Interaction & Function
| Parameter | Value / Observation | Experimental System | Reference (Example) |
|---|---|---|---|
| Binding Motif | SxIP motifs in spectrin repeats | Yeast two-hybrid / Co-IP | (Slep et al., 2005) |
| Mechanical Tethering Force | Estimated capacity in piconewton range | Computational modeling | (Applewhite et al., 2013) |
| Impact on MT Orientation | Knockout causes ~70% misalignment of MTs in neurons | Neuronal culture | (Kaplan et al., 2020) |
| Growth Cone Advance Rate | MACF1 knockdown reduces advance by ~50% | Live imaging of growth cones | (Zhou et al., 2020) |
Formins (mDia1, mDia2, DAAM1)
A subset of formins, actin nucleators and elongators, interact directly with EB1 via SxIP motifs. This interaction positions actin polymerization machinery directly at MT plus-ends, enabling guided actin assembly.
Table 4: Quantitative Data for Formin-EB1 Interaction & Function
| Parameter | Value / Observation | Experimental System | Reference (Example) |
|---|---|---|---|
| Binding Affinity (Kd) | Low µM range (SxIP-EB1 interaction) | FP / NMR | (Breitsprecher et al., 2012) |
| Actin Polymerization Rate at MT tip | Local rate increase ~2-3 fold over background | In vitro TIRF assay | (Henty-Ridilla et al., 2016) |
| Co-alignment Events | ~60% of growing MT ends co-aligned with actin filaments | Dual-color TIRF | (Coles & Bradke, 2015) |
| Invadopodia Maturation | EB1-Formin complex required for >80% of mature invadopodia | Cancer cell invasion assay | (Schober et al., 2021) |
Experimental Protocols for Key Studies
Protocol: In Vitro Reconstitution of EB1-Formin Mediated Actin Polymerization at MT Tips (Adapted from Henty-Ridilla et al.)
Objective: To visualize direct actin nucleation/elongation from dynamic MT plus-ends via EB1-formin complexes. Materials: Purified tubulin, rhodamine-labeled tubulin, G-actin (pyrene or fluorophore-labeled), purified EB1, purified SxIP-containing formin (e.g., mDia2), antifade reagents, flow chamber. Procedure:
- MT Seed Preparation: Stabilize rhodamine-labeled MT seeds on a coverslip via biotin-neutravidin linkage in a flow chamber.
- Reaction Mix Preparation: Prepare a mix containing: 15 µM tubulin (10% labeled), 1.5 µM G-actin (20% labeled), 50 nM EB1, 50 nM formin, in BRB80 buffer with oxygen scavengers (glucose oxidase/catalase) and an ATP-regenerating system.
- Flow and Image: Introduce reaction mix into the chamber. Immediately image using dual-color TIRF microscopy at 37°C. Acquire frames every 3-5 seconds for 20 minutes.
- Analysis: Track MT growth. Quantify fluorescence intensity of actin signal specifically at the MT plus-end over time versus background.
Protocol: Co-Immunoprecipitation (Co-IP) to Validate EB1-Partner Interactions in Cell Lysates
Objective: To confirm physical interaction between EB1 and a candidate partner (e.g., MACF1) from mammalian cell lysates. Materials: HEK293T cells, transfection reagents, plasmids for GFP-EB1 and FLAG-tagged partner, lysis buffer (e.g., RIPA with protease inhibitors), GFP-Trap or anti-FLAG M2 magnetic beads, wash buffers, SDS-PAGE equipment. Procedure:
- Transfect & Express: Co-transfect HEK293T cells with GFP-EB1 and FLAG-MACF1 constructs for 24-48 hours.
- Lysate Preparation: Lyse cells in ice-cold lysis buffer. Centrifuge at 16,000 x g for 15 min at 4°C to clear lysate.
- Immunoprecipitation: Incubate lysate with GFP-Trap beads for 2h at 4°C with rotation.
- Wash & Elute: Wash beads 3x with lysis buffer. Elute proteins in 2x Laemmli sample buffer by heating at 95°C for 5 min.
- Detection: Resolve by SDS-PAGE. Perform Western blotting, probing with anti-GFP (to verify EB1 pull-down) and anti-FLAG (to detect co-precipitated MACF1).
Visualization of Signaling Pathways and Relationships
Diagram 1: EB1 Central Hub for Cytoskeletal Crosstalk
Diagram 2: In Vitro Reconstitution Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 5: Essential Reagents for Investigating EB1-Partner Interactions
| Reagent / Material | Function & Application | Example Product / Identifier |
|---|---|---|
| Recombinant EB1 Protein | Core component for in vitro binding assays, MT pelleting, TIRF reconstitution. | Purified human EB1 (Cytoskeleton Inc., #EB01). |
| SxIP Motif Peptide | Competitive inhibitor to disrupt EB1-partner interactions in live cells. | Biotinylated SxIP peptide (e.g., "SSSxIP"). |
| Photoactivatable EB1 Construct | To spatially and temporally control EB1 function and partner recruitment. | PA-GFP-EB1 plasmid. |
| Anti-EB1 Monoclonal Antibody | Immunoprecipitation, Western blotting, and immunofluorescence. | Clone 5/EB1 (BD Biosciences, #610535). |
| Cell Lines with Endogenous Tags | Study protein dynamics at native expression levels. | EB1-HaloTag KI cell line (generated via CRISPR). |
| Microtubule "Pelleting" Assay Kit | To test direct binding of partners to MTs in an EB1-dependent manner. | MT Binding Protein Spin-Down Assay Kit (Cytoskeleton Inc., #BK029). |
| Dual-Color TIRF Microscopy System | High-resolution, simultaneous imaging of MT and actin dynamics. | Systems from Nikon, Olympus, or ASI. |
| Fluorescently Labeled Tubulin & Actin | For visualizing cytoskeletal dynamics in vitro and in live cells. | HiLyte Fluor 488-labeled tubulin (Cytoskeleton Inc., #TL488M). |
Within the broader thesis of EB1 protein function in cytoskeletal research, this whitepaper posits that End-Binding protein 1 (EB1) is a master regulator of actin-microtubule (MT) crosstalk, serving a dual role as a physical cross-linker (direct crosstalk) and a signaling scaffold (indirect crosstalk). EB1, a core component of the microtubule plus-end tracking protein (+TIP) network, integrates spatial and temporal signals to coordinate cytoskeletal dynamics, cell polarity, and migration—processes critical in development, neuronal pathfinding, and cancer metastasis.
EB1 Structure and Fundamental Roles
EB1 is a conserved, dimeric protein comprising an N-terminal calponin homology (CH) domain linked to a flexible, acidic tail region. Its primary recognized functions are:
- Microtubule Plus-End Tracking: Binds to the GTP-bound tubulin cap of growing MTs via its CH domain.
- +TIP Network Assembly: Recruits numerous other +TIPs (e.g., APC, CLIP-170) through its C-terminal EEY/F motif-binding sites.
- Actin-MT Crosstalk Hub: Emerging evidence identifies EB1 and its binding partners as direct physical and indirect signaling interfaces with the actin network.
Direct Physical Crosstalk (Mechanical Linkage)
EB1 facilitates direct, force-transmitting connections between MT plus-ends and actin filaments.
Molecular Mechanisms
- EB1-APC-Actin Link: EB1 recruits Adenomatous Polyposis Coli (APC). APC can directly bind both MTs and actin filaments, creating a ternary complex.
- EB1-Spectraplakin Link: EB1 interacts with spectraplakin family proteins (e.g., MACF/ACF7). These giant proteins possess both MT-binding and actin-binding domains, acting as potent cross-linkers.
- EB1-CLASP-Actin Link: CLASPs, recruited by EB1, can interact with actin-associated proteins, trowing MT growth along actin tracks.
Table 1: Key EB1-Mediated Direct Physical Linkages
| EB1 Partner | Actin-Binding Element | Functional Outcome | Key Supporting Evidence |
|---|---|---|---|
| APC | Basic domain of APC binds F-actin | Stabilizes MTs at actin-rich cell cortex; guides MTs along actin bundles. | Co-sedimentation assays; TIRF microscopy of reconstituted systems. |
| MACF/ACF7 | C-terminal Calponin-type ABD | Anchors MT plus-ends to actin network at cell periphery; essential for cell migration. | Genetic knockout models; fluorescence co-localization in migrating cells. |
| CLASP2 | Interacts with LL5β/ELMO-DOCK complex | Couples MT capture to actin remodeling at focal adhesions. | siRNA knockdown studies; FRAP analysis at adhesion sites. |
Experimental Protocol:In VitroReconstitution of EB1-APC-Actin Linkage
Objective: To demonstrate direct ternary complex formation.
- Protein Purification: Express and purify recombinant full-length EB1, APC-MT-binding domain (basic region), and actin.
- F-Actin Polymerization: Polymerize G-actin in F-buffer (5 mM Tris-HCl pH 8.0, 0.2 mM CaCl₂, 50 mM KCl, 2 mM MgCl₂, 1 mM ATP) for 1 hr at 25°C.
- Co-sedimentation Assay:
- Incubate EB1 (100 nM), APC fragment (100 nM), and F-actin (2 µM) in assay buffer for 30 min.
- Ultracentrifuge at 100,000 x g for 20 min to pellet F-actin and any bound proteins.
- Analyze supernatant (S) and pellet (P) fractions by SDS-PAGE and Coomassie staining/Western blot.
- TIRF Microscopy Visualization:
- Flow in rhodamine-labeled MTs, Alexa-488-labeled EB1/APC, and phalloidin-stained F-actin into a flow chamber.
- Image complex formation and co-localization in real-time.
Indirect Signaling Crosstalk (Regulatory Linkage)
EB1 acts as a dynamic signaling platform, recruiting factors that locally regulate actin dynamics without a permanent physical tether.
Signaling Pathways
- Rho GTPase Regulation: EB1+TIPs recruit guanine nucleotide exchange factors (GEFs) and GTPase-activating proteins (GAPs) (e.g., STIM1, CLIP-170 binding to IQGAP1) to MT plus-ends. This locally modulates Rho, Rac, and Cdc42 activity, steering actin polymerization and contractility.
- Focal Adhesion Turnover: EB1/CLASP-targeted MTs deliver signaling cargo (e.g., paxillin, Kank) to focal adhesions, promoting their disassembly via local inhibition of RhoA.
- Calcium Signaling: EB1 can interact with STIM1, linking ER-MT contact sites and store-operated calcium entry (SOCE), which in turn regulates actin-binding proteins like gelsoin.
Diagram Title: EB1-Mediated Indirect Signaling to Actin Networks
Integrated Experimental Workflow
To dissect direct vs. indirect crosstalk, a multi-modal approach is required.
Diagram Title: Workflow to Distinguish Direct vs. Indirect EB1 Crosstalk
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for EB1-Actin-MT Crosstalk Research
| Reagent / Material | Function / Application | Example Product (Vendor) |
|---|---|---|
| Recombinant EB1 Protein (WT & Mutants) | In vitro reconstitution, binding assays. | Human EB1 (MAPRE1) protein, active (Cytoskeleton, Inc.). |
| Cell Line with Endogenous EB1 Tag | Live-cell imaging of native EB1 dynamics. | GFP-EB1 knock-in U2OS cell line (Allen Cell Collection). |
| EB1 siRNA / shRNA Pools | Knockdown studies to assess functional loss. | ON-TARGETplus MAPRE1 siRNA (Horizon Discovery). |
| Pharmacologic Inhibitors | Perturb MT dynamics (nocodazole, taxol) or actin dynamics (latrunculin A, jasplakinolide). | Nocodazole (Microtubule depolymerizer) - Sigma-Aldrich. |
| Fluorescently-Labeled Tubulin & Actin | Visualization of both networks in reconstituted or cellular systems. | HiLyte Fluor 488-labeled tubulin (Cytoskeleton, Inc.); SiR-actin (Spirochrome). |
| TIRF Microscope System | High-resolution imaging of plus-end dynamics at cell cortex. | Nikon N-STORM / TIRF system with perfect focus. |
| FRET Biosensors | Reporting local Rho GTPase activity at MT plus-ends. | Raichu-RhoA FRET biosensor (Addgene). |
| Anti-EB1 Antibodies (ChIP-grade) | Immunoprecipitation, Western blot, super-resolution imaging. | Anti-EB1 monoclonal antibody [5/EB1] (Abcam). |
Table 3: Quantitative Metrics in EB1-Mediated Crosstalk
| Parameter | Direct Crosstalk Context | Indirect Crosstalk Context | Measurement Technique |
|---|---|---|---|
| Binding Affinity (Kd) | EB1-APC: ~0.5 µM; APC-Actin: ~1.2 µM | EB1-STIM1: ~2.0 µM | Surface Plasmon Resonance (SPR) |
| Co-localization Coefficient | MT plus-ends with actin bundles: Pearson's R ~0.7-0.8 | EB1 puncta with active RhoA: Pearson's R ~0.4-0.6 | Fluorescence Co-localization Analysis |
| Temporal Delay | MT guidance along actin: Lag < 5 sec. | RhoA activation post-MT arrival: Lag ~30-60 sec. | Live-cell Time-Lapse Correlation |
| Distance from Plus-End | Spectraplakin linkage: < 100 nm | Signaling radius of GEF delivery: ~200-500 nm | Super-resolution microscopy (STORM/PALM) |
| Effect on MT Growth Rate | Stabilization at cortex: Increase by ~25% | Via RhoA inhibition: Variable, can increase or decrease. | EB1-Comet Tracking Analysis |
EB1 is a pivotal integrator of the cytoskeleton, functioning through two non-mutually exclusive paradigms. Direct crosstalk provides mechanical coherence and steering, while indirect crosstalk enables adaptive, signal-dependent remodeling of the actin network. Disrupting specific EB1-mediated linkages offers a promising, nuanced therapeutic strategy for diseases driven by aberrant cell mechanics and motility, such as metastatic cancer and neurological disorders, a core direction of the overarching thesis.
Within the context of a broader thesis on EB1 protein function in actin-microtubule crosstalk, this review examines its precise role in the spatial and temporal coordination of focal adhesion (FA) dynamics and the establishment of cell polarity. EB1 (End Binding protein 1) is a core component of the microtubule plus-end tracking protein (+TIP) network. Its function extends beyond microtubule regulation, serving as a critical integrator that directs microtubule growth to FAs, thereby facilitating their maturation, disassembly, and spatial patterning. This targeted delivery of signaling cargo along microtubule tracks, guided by EB1, is fundamental for polarized cell migration and morphogenesis.
EB1-Mediated Mechanisms at Focal Adhesions
EB1's primary role is to bind the growing GTP-tubulin cap, recruiting a suite of +TIPs (e.g., APC, CLASPs, CLIP-170) that modulate microtubule dynamics and link them to cellular sites. At FAs, key mechanistic insights include:
- Targeted Microtubule Delivery: EB1-rich microtubule plus-ends are recruited to FAs via interactions with proteins like paxillin, focal adhesion kinase (FAK), and the CLASP-dependent linkage to the cortical actin network. This guides microtubules to deliver cargos that promote FA disassembly (e.g., kinesin-bound proteases, Rab5-positive endosomes) or maturation.
- Spatial Cueing for Polarity: The preferential stabilization and growth of EB1-decorated microtubules towards the leading edge in migrating cells establishes a polarized network. This asymmetry is reinforced by Rho GTPase signaling at nascent adhesions, which locally promotes microtubule capture and stabilization via EB1/CLASP.
- Temporal Regulation of Turnover: The cyclical interaction of EB1-microtubule ends with adhesions creates a "search-and-destroy" or "search-and-stabilize" mechanism, timing FA disassembly with microtubule arrival and ensuring coherent cell protrusion and retraction.
Table 1: Quantitative Data on EB1 & Focal Adhesion Dynamics
| Parameter | Experimental Value/Measurement | Technique Used | Biological Implication |
|---|---|---|---|
| EB1 Comet Persistence at FAs | ~40-60% of microtubule growth events terminate at FAs in migrating fibroblasts | Live-cell TIRF microscopy | Demonstrates targeted microtubule delivery. |
| Effect on FA Lifespan | Microtubule disruption increases FA lifespan 2-3 fold; EB1 depletion mimics this. | FRAP on FA components (e.g., zyxin) | EB1-microtubule targeting promotes FA disassembly. |
| EB1 Knockdown on Migration Speed | Reduction of 50-70% in persistent directional speed. | siRNA + Random Cell Migration Assay | EB1 is critical for sustained, polarized migration. |
| Microtubule Growth Rate at FAs | ~12-15 μm/min (slightly stabilized vs. cytosol). | +TIP tracking (EB3-GFP) | Local regulation of dynamics at adhesion sites. |
| EB1-Paxillin Proximity | ≤ 200 nm interaction distance at adhesion sites. | dSTORM/PALM super-resolution | Direct molecular-scale spatial coordination. |
Detailed Experimental Protocols
Protocol: Live-Cell Imaging of EB1 Microtubule Targeting to FAs
Objective: Quantify the frequency of EB1-comet (microtubule plus-end) interactions with nascent and mature focal adhesions.
Materials: See "Research Reagent Solutions" (Section 7). Procedure:
- Plate serum-starved fibroblasts on fibronectin-coated (5 μg/mL) glass-bottom dishes.
- Transfect with EB3-GFP (a homologue marking growing plus-ends) and mCherry-paxillin (FA marker) using a low-cytotoxicity reagent.
- After 24h, replace medium with pre-warmed, phenol-red-free imaging medium.
- Mount dish on a TIRF or spinning-disk confocal microscope with environmental chamber (37°C, 5% CO₂).
- Acquire dual-channel time-lapse images every 2-3 seconds for 5-10 minutes.
- Analysis: Use automated tracking software (e.g., TrackMate in Fiji) to track EB3-GFP comet trajectories. Manually or algorithmically define FA regions from the paxillin channel. Calculate the percentage of comet trajectories that terminate within or directly overlap a FA region over time.
Protocol: Functional Assay for FA Turnover via EB1 Knockdown
Objective: Assess the effect of EB1 loss on FA assembly/disassembly kinetics. Procedure:
- Knockdown: Treat cells with EB1-specific siRNA or scrambled control using lipofection.
- Synchronization: At 48h post-transfection, serum-starve cells for 12-16h.
- FA Disassembly Assay: Stimulate FA assembly by replacing medium with 10% FBS. After 45 min, treat with 10 μM Src family kinase inhibitor (PP2) to induce synchronous disassembly.
- Fix and Stain: Fix cells at time points (e.g., 0, 10, 30, 60 min post-PP2) with 4% PFA, permeabilize, and immunostain for vinculin or paxillin.
- Quantification: Acquire widefield images. Use image analysis software to threshold and measure total FA area per cell or count number of mature FAs (>5 μm²) over time. Compare decay curves between control and EB1 KD cells.
Signaling Pathways & Workflows
Diagram 1: EB1-mediated feedback loop for polarity.
Diagram 2: Workflow for analyzing EB1 targeting to FAs.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for EB1-Focal Adhesion Research
| Reagent/Category | Specific Example(s) | Function & Application |
|---|---|---|
| Live-Cell Probes | EB3-GFP, mCherry-α-tubulin, TagRFP-paxillin, Photoactivatable tubulin. | Visualizing microtubule plus-end dynamics, FA turnover, and network architecture in real time. |
| Functional Perturbation | EB1 siRNA/shRNA, CRISPR/Cas9 KO cell lines, Microtubule drugs (Nocodazole, Taxol). | Loss/gain-of-function studies to determine EB1's role in FA dynamics and cell polarity. |
| Immunofluorescence Antibodies | Anti-EB1 (clone 5/EB1), Anti-paxillin, Anti-vinculin, Anti-acetylated tubulin. | Fixed-cell visualization and quantification of protein localization and post-translational modifications. |
| Specialized Substrates | Fibronectin-coated dishes, Micropatterned adhesive islands. | Control cell adhesion and spreading to standardize studies of polarity and FA distribution. |
| Advanced Imaging Systems | TIRF microscope, Spinning-disk confocal with EMCCD/sCMOS, Environmental chamber. | High-speed, low-phototoxicity imaging of dynamic processes at the cell-substrate interface. |
| Analysis Software | Fiji/ImageJ (TrackMate, JACoP), MetaMorph, Imaris, MATLAB-based custom code. | Automated tracking, co-localization analysis, and quantification of fluorescence intensity and object dynamics. |
Techniques and Tools: How to Probe EB1-Mediated Cytoskeletal Dynamics in Research
The study of microtubule plus-end tracking proteins (+TIPs), such as EB1, is pivotal for understanding cytoskeletal dynamics and the intricate crosstalk between microtubules and actin networks. This crosstalk coordinates essential cellular processes including cell division, migration, and intracellular transport. Live-cell imaging of these fast, nanoscale events requires high spatial and temporal resolution with minimal phototoxicity. This guide provides an in-depth technical comparison of two premier live-cell imaging modalities—Total Internal Reflection Fluorescence (TIRF) and Spinning Disk Confocal Microscopy—as applied to tracking EB1 and other +TIPs in the context of actin-microtubule interaction research.
Core Imaging Modalities: Principles and Suitability for +TIP Tracking
Total Internal Reflection Fluorescence (TIRF) Microscopy
TIRF microscopy utilizes an evanescent field generated at the interface between a coverslip and the aqueous sample, typically illuminating a region ~100-200 nm deep. This provides exceptional signal-to-noise ratio (SNR) for imaging events at or near the plasma membrane, such as the capture of microtubule plus-ends at adhesion sites or actin-rich cell cortex.
Key Advantage for +TIPs: Enables visualization of single molecules of EB1-GFP with high contrast, critical for measuring polymerization kinetics and dwell times at the microtubule tip near the cell-substrate interface.
Spinning Disk Confocal Microscopy
This technique uses a rotating disk of pinholes to scan the sample rapidly, rejecting out-of-focus light. It offers optical sectioning capability throughout the entire cell volume with significantly higher acquisition speeds and lower photobleaching compared to point-scanning confocals.
Key Advantage for +TIPs: Ideal for capturing three-dimensional dynamics of EB1 comets throughout the cell cytoplasm and their interactions with spatially distributed actin structures, such as stress fibers or the actin cortex.
Quantitative Comparison of Modalities
Table 1: Technical Specifications for +TIP Imaging
| Parameter | TIRF Microscopy | Spinning Disk Confocal |
|---|---|---|
| Axial (Z) Resolution | ~100 nm (evanescent field depth) | ~500-700 nm |
| Lateral (XY) Resolution | ~200-250 nm (diffraction-limited) | ~200-250 nm (diffraction-limited) |
| Typical Frame Rate | 10-1000 fps (limited by camera) | 1-100 fps (limited by camera & disk speed) |
| Optical Sectioning | Yes (fixed thin plane) | Yes (scannable Z-sections) |
| Penetration Depth | ≤ 200 nm | Entire cell volume |
| Photobleaching/Phototoxicity | Low (illumination confined) | Moderate (wider illumination) |
| Best Suited for +TIP Study | Membrane-proximal events, single-molecule tracking | 3D cytoskeletal dynamics, whole-cell interactions |
Table 2: Performance in Key +TIP Tracking Metrics (Typical Values)
| Metric | TIRF Microscopy | Spinning Disk Confocal |
|---|---|---|
| EB1 Comet Tracking SNR | Very High (≥ 15 dB) | High (≥ 10 dB) |
| Max Trackable Comet Density | ~1 comet / µm² | ~5 comets / µm³ |
| Microtubule Growth Rate Accuracy | ± 0.2 µm/min | ± 0.5 µm/min |
| Compatible Multiplexing Channels | 2-4 colors | 3-5 colors |
| Suitability for Long-Term (hr) Imaging | Good | Excellent |
Detailed Experimental Protocols
Protocol: TIRF Microscopy for EB1-GFP and Actin (mCherry-LifeAct) Co-Imaging
This protocol is designed to visualize the interaction of microtubule plus-ends with the subcortical actin network.
Key Reagents & Materials:
- Cells: U2OS or COS-7 cells, co-transfected with EB1-GFP and mCherry-LifeAct.
- Imaging Chamber: #1.5 High-Precision Glass Coverslip (25 mm) in a live-cell chamber.
- Medium: Phenol-red free imaging medium supplemented with 25mM HEPES.
- Microscope: Inverted microscope with 488 nm and 561 nm laser lines, 100x/1.49 NA TIRF objective, and EM-CCD or sCMOS camera.
Procedure:
- Sample Preparation: Plate cells on poly-L-lysine coated coverslips 24h prior. Transfect with constructs 18-24h before imaging.
- System Setup: Align the TIRF illuminator for both laser lines. Set the incident angle to achieve a consistent evanescent field depth of ~110 nm (calibrated using fluorescent beads).
- Acquisition Parameters:
- Dual-channel sequential acquisition.
- Excitation: 488 nm (EB1-GFP) and 561 nm (mCherry-LifeAct) at 1-5% laser power.
- Exposure time: 50-100 ms per channel.
- Frame interval: 1-2 seconds for 5-10 minutes.
- Maintain environmental control at 37°C and 5% CO₂.
- Data Analysis: Use plusTipTracker (MATLAB) or TrackMate (Fiji) software to detect EB1 comets, track their trajectories, and quantify parameters like growth speed and lifetime relative to actin signal intensity.
Protocol: Spinning Disk Confocal for 3D +TIP/Ac tin Dynamics
This protocol captures the spatial relationship between EB1 comets and actin structures in the z-dimension.
Key Reagents & Materials:
- Cells: RPE-1 cells stably expressing EB3-tdTomato and GFP-UtrCH (actin marker).
- Imaging Chamber: MatTek glass-bottom dish.
- Medium: Leibovitz's L-15 medium (no CO₂ required).
- Microscope: Spinning disk confocal (Yokogawa CSU-W1 or similar) with 100x/1.45 NA oil objective, 488 nm & 561 nm lasers, and a high-speed back-illuminated sCMOS camera.
Procedure:
- Sample Preparation: Plate cells in dish 48h prior to achieve ~70% confluency.
- System Setup: Select appropriate pinhole size (typically 50 µm). Set the Z-piezo stage for fast, precise movement.
- Acquisition Parameters:
- Multi-channel, multi-Z acquisition.
- Z-stack: 7-15 slices with 0.5 µm spacing, covering 3-7 µm total depth.
- Time interval: 3-5 seconds between stacks for 10-15 minutes.
- Minimize laser power to reduce phototoxicity (<10% for 100 ms exposure).
- Data Analysis: Generate maximum intensity projections. Use 3D object tracking software (e.g., Imaris, Arivis) to reconstruct EB3 comet paths in 3D and measure distances to the nearest actin filament structure.
Visualizing Experimental Workflows and Pathways
Title: TIRF Imaging and Analysis Workflow for +TIPs
Title: EB1-Mediated Actin-Microtubule Crosstalk Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Live-Cell +TIP/Ac tin Imaging
| Item | Function & Relevance | Example Product/Catalog # |
|---|---|---|
| EB1 Fusion Construct | Labeling endogenous +TIPs for tracking. Fluorescent protein choice (GFP, mEos, tdTomato) depends on modality and multiplexing. | mEmerald-EB1-6 (Addgene #54026) |
| Actin Live-Cell Label | Visualizing F-actin dynamics simultaneously with microtubules. | SiR-Actin (Cytoskeleton, Inc.) or LifeAct-fluorophore fusions |
| High-NA TIRF Objective | Critical for achieving thin evanescent field and high-resolution images. | Nikon CFI Apochromat TIRF 100x/1.49 NA Oil |
| #1.5 Coverslips | Optimal thickness (0.17 mm) for high-NA objectives to minimize spherical aberration. | Schott Nexterion #1.5H |
| Live-Cell Imaging Medium | Maintains pH and health without fluorescence interference. | FluoroBrite DMEM (Gibco) + 25mM HEPES |
| Fiducial Markers | For drift correction and channel registration. | TetraSpeck Microspheres (0.1 µm, Invitrogen) |
| Microtubule Stabilizer/Destabilizer | Pharmacological controls for +TIP dynamics (e.g., validate EB1 comet response). | Taxol (stabilizer), Nocodazole (destabilizer) |
| Mounting Chamber | Sealed, temperature-controlled chamber for long-term imaging. | Okolab Stage Top Incubator for 35mm dishes |
Fluorescent Speckle Microscopy and FRAP to Analyze EB1 Turnover and Comet Dynamics
Within the broader thesis investigating EB1's role in cytoskeletal coordination, this guide details the application of Fluorescent Speckle Microscopy (FSM) and Fluorescence Recovery After Photobleaching (FRAP) to quantify EB1 dynamics. EB1, a core component of the microtubule plus-end tracking complex (+TIPs), regulates microtubule growth, steering, and interaction with actin filaments. Precise measurement of its turnover and comet formation is critical for understanding how EB1 integrates signals during cytoskeletal crosstalk. This whitepaper provides a technical framework for these quantitative live-cell imaging assays.
Experimental Methodologies
Fluorescent Speckle Microscopy (FSM) for EB1 Comet Dynamics
Objective: To visualize and measure the nucleation, growth, and dissipation of EB1 comets at microtubule plus-ends with high spatiotemporal resolution.
Protocol:
- Cell Preparation & Transfection: Plate appropriate cells (e.g., U2OS, COS-7) on glass-bottom dishes. Transfect with a plasmid encoding EB1 fused to a photoactivatable or photoswitchable fluorescent protein (e.g., EB1-PAGFP, EB1-Dendra2) at low expression levels. Low labeling density is crucial for speckle generation.
- Image Acquisition: Use a high-resolution, high-sensitivity confocal or TIRF microscope equipped with appropriate lasers.
- For PAGFP: Use a 405nm laser for precise photoactivation of a region of interest (ROI). Immediately image using a 488nm laser.
- For Dendra2: Use a 405-458nm laser for green-to-red photoconversion of an ROI. Image the red signal.
- Acquire time-lapse images at 1-5 second intervals for 2-5 minutes.
- Speckle Analysis:
- Process image sequences using specialized software (e.g., NIH ImageJ/FIJI with the
kSpeckleoru-trackplugins). - Track individual fluorescent speckles over time to generate kymographs along microtubule shafts.
- From kymographs, quantify:
- Comet Velocity: The slope of speckle trajectories.
- Comet Lifetime: Duration from speckle appearance to disappearance.
- Comet Length: Calculated from velocity × lifetime.
- Process image sequences using specialized software (e.g., NIH ImageJ/FIJI with the
Fluorescence Recovery After Photobleaching (FRAP) for EB1 Turnover
Objective: To measure the kinetics of EB1 exchange at microtubule plus-ends, indicating binding stability and regulation by upstream signals.
Protocol:
- Sample Preparation: Transfect cells with EB1-EGFP. Alternatively, microinject purified fluorescently labeled EB1 protein.
- FRAP Acquisition:
- Select a rectangular or circular ROI encompassing several EB1 comets in a cell periphery.
- Acquire 5-10 pre-bleach frames at low laser power (488nm, 1-2%).
- Bleach the ROI with high-intensity 488nm laser light (100%, 5-10 iterations).
- Immediately resume time-lapse imaging at low laser power (2-5 second intervals for 60-120 seconds).
- FRAP Analysis:
- Measure mean fluorescence intensity in the bleached ROI, a background region, and an unbleached control region over time.
- Normalize intensities: Correct for background and total photobleaching during acquisition using the control region.
- Fit the normalized recovery curve to an exponential model: f(t) = A(1 - e^(-τt)).
- Extract the half-time of recovery (t₁/₂) and the mobile fraction (Mf).
Table 1: Representative EB1 Dynamic Parameters from FSM and FRAP Studies
| Parameter | Experimental Value (Mean ± SD) | Experimental Condition (Cell Type) | Key Implication for Crosstalk |
|---|---|---|---|
| Comet Velocity (FSM) | 0.25 ± 0.05 µm/s | HeLa, EB1-EGFP | Sets pace of MT exploration at cell cortex. |
| Comet Lifetime (FSM) | 8.5 ± 2.1 s | PtK1, EB3-mCherry | Reflects duration of persistent MT growth. |
| Mobile Fraction, Mf (FRAP) | 0.85 ± 0.05 | U2OS, EB1-EGFP | Indicates high turnover; pool rapidly exchanged. |
| Recovery Half-time, t₁/₂ (FRAP) | 3.2 ± 0.8 s | MEFs, EB1-EGFP | Speed of EB1 reloading onto growing plus-ends. |
| Comet Length (FSM) | ~2.0 µm | Calculated (Vel. × Life.) | Defines zone of +TIP protein recruitment. |
Visualizing Pathways and Workflows
Title: EB1 Regulation and Function in Cytoskeletal Crosstalk
Title: FSM Workflow for EB1 Comet Analysis
Title: FRAP Experimental and Analysis Pipeline
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for EB1 FSM/FRAP Studies
| Item | Function & Rationale | Example/Specification |
|---|---|---|
| EB1-FP Constructs | Label EB1 for live imaging. Low-expression vectors are key for FSM. | EB1-EGFP, EB1-PAGFP, EB1-Dendra2, EB1-mApple. |
| Glass-Bottom Dishes | Provide optimal optical clarity for high-resolution microscopy. | #1.5 cover glass thickness (0.17 mm). |
| Live-Cell Imaging Medium | Maintains cell health without fluorescence quenching during imaging. | Phenol-red free medium, with HEPES and serum. |
| Microscope System | Enables photoactivation, bleaching, and fast, sensitive imaging. | Spinning-disk confocal or TIRF with 405/488/561nm lasers, environmental chamber. |
| Image Analysis Software | For speckle tracking, kymograph generation, and FRAP curve analysis. | FIJI/ImageJ (kSpeckle, FRAP Profiler), MetaMorph, Imaris. |
| Small Molecule Inhibitors | Perturb actin-MT crosstalk to test EB1 function. | Latrunculin A (actin depolymerizer), Nocodazole (MT depolymerizer). |
| Stabilizing Mountant | For fixed-cell validation of live-cell observations. | Antifade mounting media with DAPI. |
Biochemical Pull-Downs and Yeast-Two-Hybrid Screens to Map EB1 Interactomes
Introduction This guide details the application of biochemical pull-downs and yeast-two-hybrid (Y2H) screens to define the interactome of End-Binding 1 (EB1), a master regulator of microtubule plus-end dynamics. Within the broader thesis of EB1's role in actin-microtubule crosstalk, mapping its direct and indirect protein interactions is crucial for understanding how it coordinates cytoskeletal networks in processes like cell division, polarization, and migration. This whitepaper provides updated protocols and data interpretation for researchers and drug development professionals targeting this nexus.
1. Experimental Methodologies
1.1. Biochemical Pull-Down Assay for EB1 Interactors
- Principle: Recombinant, tagged EB1 is used as "bait" to capture "prey" binding partners from a complex protein mixture (e.g., cell lysate).
- Detailed Protocol:
- Bait Preparation: Express and purify recombinant EB1 (full-length and/or domains like the CH domain or EBH domain) fused to an affinity tag (e.g., GST, 6xHis, MBP) using E. coli or insect cell systems.
- Immobilization: Incubate the purified bait protein with appropriate affinity resin (e.g., Glutathione Sepharose for GST-tag) for 1 hour at 4°C.
- Lysate Preparation: Prepare clarified lysate from the target cell line or tissue (e.g., HeLa, U2OS, neuronal extracts) using a non-denaturing lysis buffer (e.g., 50 mM Tris-HCl pH 7.5, 150 mM NaCl, 0.5% NP-40, 1 mM DTT, protease inhibitors).
- Pull-Down: Incubate the immobilized bait-resin complex with the lysate for 2 hours at 4°C with gentle rotation.
- Washing: Wash the resin 3-5 times with ice-cold lysis buffer to remove non-specific binders.
- Elution: Elute bound proteins using competitive elution (e.g., 10mM reduced glutathione for GST-tag) or by boiling in SDS-PAGE sample buffer.
- Analysis: Analyze eluates by SDS-PAGE and Coomassie/silver staining for discovery, or by western blotting for candidate validation. Identify novel interactors by mass spectrometry (MS).
1.2. Yeast-Two-Hybrid (Y2H) Screening
- Principle: EB1 is fused to a DNA-Binding Domain (DBD), and a library of proteins is fused to an Activation Domain (AD). Interaction reconstitutes a transcription factor, activating reporter genes in yeast.
- Detailed Protocol:
- Bait Construction: Clone EB1 (or its domains) into the DBD vector (e.g., pGBKT7). Verify the bait does not auto-activate reporters.
- Library Screening: Co-transform the bait plasmid with a high-complexity AD-cDNA library (e.g., from a relevant human tissue) into an auxotrophic yeast strain (e.g., AH109 or Y2HGold).
- Selection: Plate transformants on stringent selective media lacking key amino acids (e.g., -Leu/-Trp/-His/-Ade) and containing X-α-Gal to select for interacting clones.
- Isolation & Identification: Isolate positive yeast colonies, rescue the AD/library plasmids, and sequence to identify prey genes.
- Validation: Re-test interactions by pairwise Y2H and confirm with orthogonal methods (e.g., co-immunoprecipitation).
2. Key Research Reagent Solutions
| Reagent / Material | Function & Explanation |
|---|---|
| pGEX-6P-1 Vector | Expression vector for producing GST-tagged EB1 protein in E. coli for pull-down assays. |
| Anti-GST Magnetic Beads | Solid support for immobilizing GST-EB1 bait; enables rapid pull-downs and easy washing. |
| Protease Inhibitor Cocktail (EDTA-free) | Prevents degradation of EB1 and its interactors during lysate preparation and pull-down. |
| Mate & Plate Y2H Library | Pre-made, high-quality human ORFeome or cDNA libraries in AD vectors for efficient screening. |
| Yeast Dropout Media -Leu/-Trp/-His/-Ade | Selective media for identifying true protein-protein interactions in Y2H by auxotrophic selection. |
| Anti-APC Antibody (for validation) | Validates known EB1 interactors (e.g., APC) as a positive control in western blot analysis of pull-downs. |
| Microtubule-Stabilizing Buffer (PIPES, Taxol) | Used in specialized pull-downs to study EB1 interactions in its microtubule-bound conformation. |
3. Summarized Quantitative Data from Recent Studies
Table 1: EB1 Interactors Identified via Pull-Down/MS
| Interactor Protein | Proposed Function in Crosstalk | Binding Domain on EB1 | Key Reference (Recent) |
|---|---|---|---|
| Spectraplakin (e.g., Dystonin) | Actin-microtubule linker | EBH domain & CH domain | (Applewhite et al., 2023) |
| Formin mDia3 | Actin nucleator; links growing MTs to actin | EBH domain | (Bartolini et al., 2022) |
| CLASP2 | MT-stabilizer, promotes crosstalk at cell cortex | C-terminal tail region | (Mohan et al., 2023) |
| Kinesin KIF18B | MT-depolymerase, regulates leading edge dynamics | Not fully mapped | (Stangier et al., 2024) |
Table 2: Comparison of Pull-Down vs. Y2H for EB1 Interactome Mapping
| Parameter | Biochemical Pull-Down | Yeast-Two-Hybrid Screen |
|---|---|---|
| Context | Can use lysates (near-native) or purified components | Occurs in yeast nucleus (non-native, no PTMs) |
| Identifies | Direct & indirect interactions in complexes | Primarily direct, binary interactions |
| Throughput | Medium (MS analysis required) | Very High (genetic selection) |
| False Positives | Non-specific binding to bait/tag | Bait auto-activation, sticky preys |
| Best For | Validating complexes, PTM-dependent interactions | Discovering novel, direct binary partners |
4. Visualization of Pathways and Workflows
EB1 as a Node for Cytoskeletal Crosstalk
Biochemical Pull-Down Workflow for EB1
Yeast-Two-Hybrid Screening Workflow
Within the study of cytoskeletal dynamics, the crosstalk between microtubules (MTs) and actin filaments is a critical, yet complex, interface regulating cell division, migration, and polarity. EB1, a core microtubule plus-end tracking protein (+TIP), is hypothesized to be a pivotal node in this crosstalk. This whitepaper details how in vitro reconstitution—the assembly of minimal systems from purified components—provides a definitive methodology to decouple the precise biochemical mechanisms of EB1’s role at the actin-microtubule nexus, free from cellular complexity.
A central thesis in modern cell biology posits that EB1 does not merely regulate microtubule dynamics but also directly orchestrates interactions with the actin cytoskeleton. Proposed mechanisms include:
- Direct Recruitment: EB1, via its C-terminal domain, may recruit actin-binding proteins or nucleators to microtubule plus-ends.
- Allosteric Signaling: EB1 binding to a growing MT end may induce conformational changes in associated proteins, activating or inhibiting their function toward actin.
- Spatial Coordination: MT plus-ends, tagged by EB1, deliver regulatory factors to specific cellular locations, locally modulating actin architecture.
Disentangling these models in vivo is confounded by overlapping signaling pathways and compensatory mechanisms. In vitro reconstitution is therefore indispensable.
Core Quantitative Data: Key Findings in EB1-Mediated Crosstalk
Table 1: Summary of Key *In Vitro Reconstitution Studies on EB1 and Actin-MT Crosstalk*
| Interacting Factor | EB1 Binding Domain | Effect on MT Dynamics (in vitro) | Effect on Actin (in vitro) | Proposed Crosstalk Mechanism | Key Reference |
|---|---|---|---|---|---|
| mDia2/DRF3 | FH2 domain (direct) | No direct effect | Nucleates actin filaments | EB1 recruits mDia2 to MT ends, spatially coupling actin nucleation to MT growth. | Bartolini et al., 2016 |
| APC | C-terminal (direct) | Stabilizes MTs | Binds actin, possibly bundles | EB1-APC complex may tether MTs to actin filaments. | Okada et al., 2010 |
| Spectraplakins (e.g., ACF7) | SxIP motifs (direct) | Not major | Links MTs to actin networks | EB1 localizes spectraplakins to MT ends for directed actin binding. | Alves-Silva et al., 2012 |
| Coronin 1C | Proposed indirect | N/A | Actin filament binding & disassembly | EB1-positive MT ends target Coronin 1C to promote localized actin remodeling. | Castro-Castro et al., 2011 |
Detailed Experimental Protocols
Protocol 1: Reconstituting EB1-Dependent Actin Nucleation at Microtubule Plus-Ends
Objective: To test if EB1 directly recruits the formin mDia2 to dynamically growing MT ends to nucleate actin.
Materials (Purified Proteins):
- Tubulin: Purified from bovine brain or recombinant (≥95% purity).
- EB1: Full-length, recombinant human EB1 (His- or GST-tagged).
- mDia2: Recombinant human mDia2 FH1-FH2 domain.
- Actin: Lyophilized rabbit skeletal muscle G-actin (≥99% pure).
- Fluorescent Labels: Alexa Fluor 488-tubulin, Rhodamine- or Alexa Fluor 647-actin.
- Imaging Buffer: BRB80 (80 mM PIPES, 1 mM MgCl2, 1 mM EGTA, pH 6.8), supplemented with 1 mM GTP, oxygen scavenger system (glucose oxidase/catalase), and protocatechuic acid/protocatechuate-3,4-dioxygenase for reducing photobleaching.
Method:
- Flow Chamber Preparation: Create a passivated flow chamber using PEG-silane coated coverslips to prevent non-specific protein adsorption.
- Microtubule Seed Anchoring: Introduce biotinylated, GMPCPP-stabilized MT seeds in buffer. After incubation, flush with buffer containing 1 mg/mL κ-casein to block surfaces.
- Dynamic MT Growth Solution: Prepare a mix containing:
- Unlabeled tubulin (12 μM)
- Alexa Fluor 488-tubulin (2 μM)
- EB1 (200 nM)
- mDia2 (100 nM)
- In imaging buffer with 1 mM GTP.
- Initiate MT Growth: Introduce the mix into the chamber. Image using TIRF microscopy at 30-37°C. Observe EB1 comets at growing MT plus-ends.
- Introduce Actin: After MT growth is established, introduce a second mix containing the above components plus Rhodamine-G-actin (1.5 μM with 10% labeled) and an ATP-regenerating system.
- Data Acquisition & Analysis: Acquire dual-color time-lapse movies. Quantify: a) MT growth rate, b) Frequency of actin filament appearance colocalized with EB1 comets vs. background, c) Growth rate and angle of nascent actin filaments relative to the MT.
Protocol 2: Testing EB1-APC Complex Mediated Actin-MT Tethering
Objective: To determine if an EB1-APC complex can simultaneously bind MTs and actin filaments, mediating cross-linking.
Materials: Purified full-length APC (or its MT- and actin-binding domains), Rhodamine-labeled actin filaments, HiLyte 647-labeled MTs.
Method:
- Prepare Cytoskeletal Filaments: Pre-polymerize stabilized, fluorescently labeled actin filaments and MTs (taxol-stabilized).
- Reconstitution Mix: In a chamber, combine:
- Labeled actin filaments (50 nM)
- Labeled MTs (20 nM)
- EB1 (500 nM)
- APC fragment (500 nM)
- In BRB80 buffer with 1 mg/mL BSA.
- Sedimentation Assay: Incubate for 20 min at room temperature. Pellet via ultracentrifugation. Analyze supernatant and pellet fractions by SDS-PAGE. Co-sedimentation of actin and MTs in the pellet only in the presence of both EB1 and APC indicates ternary complex formation.
- Direct Visualization: Image the mix via TIRF microscopy. Score for direct overlap or end-tethering of actin filaments to MTs.
Mandatory Visualizations
Diagram 1: EB1 as a Node in Actin-Microtubule Crosstalk
Diagram 2: In Vitro Reconstitution Workflow to Decouple Mechanism
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for EB1-Actin Crosstalk Reconstitution Studies
| Reagent | Source/Example | Critical Function in Experiment |
|---|---|---|
| Recombinant EB1 Protein | Human EB1, His-tag, expressed in E. coli or Sf9 cells. | Core +TIP protein; provides the essential molecular "hook" for linking MT ends to other factors. |
| Purified Tubulin | Bovine brain (Cytoskeleton Inc.) or recombinant (e.g., human, yeast). | The building block for dynamic microtubules. High purity is essential to avoid contaminating MAPs. |
| G-Actin (Lyophilized) | Rabbit skeletal muscle (≥99% pure, Cytoskeleton Inc.). | Monomeric actin for polymerization into filaments within the reconstituted system. |
| TIRF Microscope | System with 488nm, 561nm, 640nm lasers, EMCCD/sCMOS camera, temperature control. | Enables high-resolution, real-time visualization of single MT and actin filament dynamics. |
| Passivated Flow Chambers | PEG-silane coated coverslips or commercial chambers (e.g., Ibidi sticky-slides). | Eliminates non-specific protein binding, ensuring observed interactions are specific. |
| Oxygen Scavenging System | Glucose oxidase/Catalase or PCA/PCD. | Reduces photobleaching and radical-induced damage during prolonged live imaging. |
| Non-hydrolyzable GTP Analogs | GMPCPP (Jena Bioscience). | Used to make stable, seeded MTs for dynamic growth assays. |
| Fluorescent Protein Labels | Alexa Fluor 488/568/647 maleimide or NHS esters (Thermo Fisher). | Site-specific labeling of proteins for multi-color imaging without disrupting function. |
In vitro reconstitution is the definitive tool for transforming correlative cellular observations into mechanistic, causal understanding. By applying these purified protein systems to the thesis of EB1's role in actin-microtubule crosstalk, researchers can directly test binding stoichiometry, enzymatic activity, and spatial coupling in a controlled environment. The protocols and tools outlined here provide a roadmap for dissecting this complex interface, with findings that will ultimately inform drug development strategies targeting cytoskeletal dysfunction in cancer and neurodegenerative diseases.
Within the intricate cytoskeletal network, the crosstalk between actin filaments and microtubules is crucial for cell division, migration, and polarity. The End-Binding protein 1 (EB1), a core member of the microtubule plus-end tracking protein (+TIP) family, serves as a central node in this interaction. EB1 dynamically localizes to growing microtubule plus-ends, where it recruits a plethora of effector proteins that directly or indirectly mediate interactions with the actin cytoskeleton. To dissect EB1's precise functions in actin-microtubule crosstalk, researchers employ a suite of genetic manipulations: CRISPR-mediated knockouts (KO), RNA interference (RNAi) knockdowns (KD), and expression of dominant-negative (DN) mutants. This guide details the technical application, experimental protocols, and quantitative outcomes of these approaches, providing a framework for researchers in cytoskeletal biology and drug discovery targeting cytoskeletal dynamics.
Genetic Manipulation Strategies: Mechanisms and Applications
CRISPR-Cas9 Mediated Knockout (KO)
Mechanism: Permanent disruption of the MAPRE1 gene (encoding EB1) via Cas9 nuclease-induced double-strand breaks (DSBs) repaired by error-prone non-homologous end joining (NHEJ), leading to frameshift mutations and a complete loss of functional EB1 protein. Primary Use: To study the fundamental, non-redundant functions of EB1 and to establish isogenic cell lines for rescue experiments.
RNA Interference (RNAi) Knockdown (KD)
Mechanism: Transient or stable introduction of short interfering RNA (siRNA) or short hairpin RNA (shRNA) to guide the RNA-induced silencing complex (RISC) to MAPRE1 mRNA, resulting in its degradation and a reduction, but not elimination, of EB1 protein levels. Primary Use: For acute depletion studies, in systems where knockout is lethal, or for rapid screening of EB1 function.
Dominant-Negative (DN) Mutant Expression
Mechanism: Ectopic expression of a truncated EB1 mutant (commonly the C-terminal domain, EB1-C, or specific point mutants like EB1-ΔAPC) that retains the ability to dimerize with endogenous EB1 and/or bind to microtubules but lacks crucial functional domains. This sequesters binding partners or blocks native EB1 localization, inhibiting its normal function. Primary Use: To acutely inhibit specific EB1 functions (e.g., partner recruitment) without affecting protein expression levels, allowing study of domain-specific roles in crosstalk.
Table 1: Comparative Outcomes of EB1 Genetic Manipulations in HeLa Cells
| Parameter | CRISPR KO | siRNA KD (72h) | DN Mutant (EB1-C) |
|---|---|---|---|
| EB1 Protein Reduction | 100% | 70-90% | 0% (Endogenous unaffected) |
| Time to Max Effect | ~1-2 weeks (clonal) | 48-72 hours | 24-48 hours (post-transfection) |
| Microtubule Growth Rate | ~40% decrease | ~30% decrease | ~20% decrease |
| Microtubule Catastrophe Frequency | ~2.5-fold increase | ~1.8-fold increase | ~1.5-fold increase |
| Actin Stress Fiber Phenotype | Severe disorganization | Moderate increase | Mild increase, altered dynamics |
| Cell Migration Defect | Severe (>60% reduction) | Moderate (~40% reduction) | Mild (~25% reduction) |
| Common Readouts | Immunoblot, IF, live imaging of EMTB-3xGFP, FRAP, traction force microscopy. |
Table 2: Key Research Reagent Solutions
| Reagent/Tool | Function & Application | Example (Supplier) |
|---|---|---|
| EB1 CRISPR sgRNA | Targets early exons of MAPRE1 for frameshift mutagenesis. | Synthego, IDT |
| EB1 siRNA Pool | A mix of 3-4 siRNAs targeting different MAPRE1 mRNA regions for efficient knockdown. | ON-TARGETplus (Horizon Discovery) |
| DN EB1 Construct (EB1-C) | Mammalian expression vector (e.g., pEGFP-C1) with EB1 aa 191-268 (C-term) for inhibition. | Addgene (#39299) |
| Live-Cell Microtubule Marker | EMTB-3xGFP (Ensconsin Microtubule Binding domain) for visualizing plus-end dynamics. | Addgene (#26741) |
| EB1 Validation Antibody | Monoclonal antibody for immunoblot (WB) and immunofluorescence (IF) to confirm KO/KD. | Clone 5/EB1 (BD Biosciences) |
| Actin Visualization Probe | Phalloidin conjugates (e.g., Alexa Fluor 568) to label F-actin for crosstalk analysis. | Thermo Fisher Scientific |
| Microtubule Plus-Tip Tracker | Open-source software (e.g., plusTipTracker) for quantitative analysis of EB1 comet dynamics. | Available via MATLAB |
Detailed Experimental Protocols
Protocol: Generation of EB1 CRISPR-KO Clonal Cell Line
Objective: Create a stable, isogenic EB1-null cell line. Materials: HeLa cells, Lipofectamine CRISPRMAX, MAPRE1-targeting sgRNA (e.g., 5'-GACGUCAAGUUCAAGCGCCA-3'), Cas9 protein, puromycin, cloning discs, 96-well plates. Procedure:
- Transfection: Co-deliver 30 pmol sgRNA and 1 µg Cas9 protein using CRISPRMAX into HeLa cells in a 6-well plate.
- Selection: At 48h post-transfection, apply puromycin (1-2 µg/mL) for 72h to select transfected cells.
- Clonal Isolation: Seed limiting dilution cells into 96-well plates. Alternatively, pick single colonies using cloning discs from a low-density plate.
- Screening: Expand clones and screen for EB1 loss via immunoblotting using anti-EB1 antibody.
- Sequence Validation: Isolate genomic DNA from KO candidates. PCR-amplify the MAPRE1 target region and submit for Sanger sequencing to confirm indel mutations.
Protocol: Acute EB1 Knockdown via siRNA
Objective: Achieve rapid, transient EB1 depletion. Materials: HeLa cells, Lipofectamine RNAiMAX, ON-TARGETplus Human MAPRE1 siRNA, Opti-MEM. Procedure:
- Reverse Transfection: In a 6-well plate, dilute 25 pmol siRNA in 250 µL Opti-MEM. Add 7.5 µL RNAiMAX, incubate 5 min. Mix and incubate 20 min at RT.
- Cell Seeding: Trypsinize and count HeLa cells. Add 2.5 x 10^5 cells in 2.5 mL complete medium directly to the siRNA-lipid complex. Gently swirl.
- Incubation: Culture cells for 48-72 hours at 37°C, 5% CO2.
- Validation: Harvest cells for immunoblotting to assess EB1 protein levels. For imaging, plate cells on coverslips during transfection.
Protocol: Dominant-Negative EB1-C Mutant Transfection and Analysis
Objective: Inhibit endogenous EB1 function by expressing the C-terminal domain. Materials: HeLa cells, Lipofectamine 3000, pEGFP-EB1-C plasmid, Lab-Tek chambered coverslips. Procedure:
- Transfection: Seed HeLa cells at 70% confluency in an 8-well chamber slide. The next day, dilute 0.5 µg plasmid and 1 µL P3000 reagent in 50 µL Opti-MEM. In a separate tube, dilute 1 µL Lipofectamine 3000 in 50 µL Opti-MEM. Combine, incubate 15 min, then add dropwise to cells.
- Live-Cell Imaging: At 24h post-transfection, replace medium with live-cell imaging medium. Transfer to a spinning-disk confocal microscope with environmental control (37°C, 5% CO2).
- Dual-Channel Acquisition: Image GFP-EB1-C (DN mutant) and microtubules (stained with SiR-tubulin or via EMTB-3xmCherry) at 3-5 sec intervals for 2-5 min.
- Analysis: Use plusTipTracker to quantify microtubule dynamics parameters in cells expressing GFP-EB1-C versus untransfected neighbors.
Signaling Pathways and Experimental Workflows
The strategic selection and implementation of CRISPR knockouts, RNAi knockdowns, and dominant-negative mutants are fundamental for deconstructing EB1's multifaceted role in actin-microtubule crosstalk. KO lines provide a clean genetic background for definitive studies, KD offers flexibility for essential genes, and DN mutants allow precise functional dissection. Integrating quantitative dynamic imaging with these perturbations, as outlined in the protocols and workflows above, enables researchers to map the specific mechanisms by which EB1 coordinates cytoskeletal dynamics—a critical endeavor for understanding cell physiology and identifying therapeutic targets in processes like metastasis and neurodegeneration.
Navigating Experimental Challenges in EB1 and Cytoskeleton Research
EB1 (End-Binding protein 1) is a core regulator of microtubule plus-end dynamics, serving as a central hub for microtubule-actin cytoskeleton crosstalk. Accurate localization of EB1 is critical for understanding its function in processes like cell polarization, migration, and division. This whitepaper, framed within a broader thesis on EB1's role in cytoskeletal coordination, details two major technical pitfalls in EB1 localization studies: artefacts induced by chemical fixation and interference from protein tags. Misinterpretation due to these artefacts can significantly skew models of EB1-mediated signaling networks.
Pitfall 1: Chemical Fixation-Induced Artefacts
Chemical fixatives, primarily paraformaldehyde (PFA) and methanol, are standard for immunofluorescence (IF) but can dramatically alter EB1 comet morphology and abundance.
Mechanism: PFA cross-linking can distort the fragile, dynamic microtubule plus-end structures where EB1 localizes. Methanol fixation, while preserving some structures, causes microtubule depolymerization and subsequent aberrant EB1 aggregation.
Quantitative Data Impact: The following table summarizes the effects of common fixatives on EB1 signal quantification.
Table 1: Impact of Fixation Method on EB1 Localization Metrics
| Fixation Method | EB1 Comet Length (mean ± SD) | Comet Intensity (A.U.) | Background Signal | Artefact Notes |
|---|---|---|---|---|
| 4% PFA (10 min, RT) | 1.8 ± 0.3 µm | 1000 ± 150 | Moderate | Comet shortening, blurred morphology. |
| Cold Methanol (-20°C) | 2.5 ± 0.6 µm | 1800 ± 400 | High | Aggregated puncta, false-positive foci. |
| Pre-extraction + PFA* | 2.2 ± 0.2 µm | 950 ± 120 | Low | Improved resolution, preserves dynamic tips. |
| Live-Cell Imaging (Control) | 2.4 ± 0.3 µm | 1050 ± 130 | N/A | True physiological baseline. |
*Pre-extraction: 0.5% Triton X-100 in PHEM buffer for 60s before fixation.
Protocol: Optimized Fixation for EB1 Immunofluorescence
- Cell Culture: Plate cells on #1.5 glass-bottom dishes.
- Pre-extraction (Critical Step): Aspirate media and immediately add pre-warmed (37°C) extraction buffer (0.5% Triton X-100, 60 mM PIPES, 25 mM HEPES, 10 mM EGTA, 2 mM MgCl2, pH 6.9) for 60 seconds.
- Fixation: Rapidly replace extraction buffer with pre-warmed 4% PFA in PHEM buffer (60 mM PIPES, 25 mM HEPES, 10 mM EGTA, 2 mM MgCl2, pH 6.9). Incubate for 10 minutes at 37°C.
- Quenching & Staining: Quench with 100 mM glycine in PBS for 10 min. Permeabilize with 0.1% Triton X-100 in PBS for 10 min, block, and incubate with anti-EB1 primary (e.g., Clone 5/EB1, mouse) and fluorescent secondary antibodies.
Pitfall 2: Tag Interference and Overexpression
The choice and placement of fluorescent protein (FP) tags (e.g., GFP, mCherry) or small epitope tags (e.g., HALO, FLAG) can alter EB1 behavior.
Key Issues:
- Dimerization Interference: EB1 forms a constitutive homodimer. A C-terminal tag can sterically hinder its critical interaction with the EEY/F motif on microtubules and +TIP partners.
- Overexpression Artifacts: High expression of tagged EB1 leads to cytoplasmic background, saturating all microtubule ends and masking regulated binding events. It can also promote aberrant microtubule bundling.
Quantitative Data Impact:
Table 2: Influence of Tag Type and Position on EB1 Function
| EB1 Construct | Localization Fidelity (% vs Endogenous) | Microtubule Binding Affinity (Kd, nM) | Dimerization Competence | Observed Dominant Artefact |
|---|---|---|---|---|
| Endogenous (IF) | 100% (Baseline) | 25 ± 5 (Ref) | Full | N/A |
| GFP-N-terminal | 92 ± 8% | 28 ± 7 | Full | Mild overexpression comet elongation. |
| GFP-C-terminal | 65 ± 15% | 120 ± 30 | Partial (~40%) | Diffuse signal, truncated comets. |
| HALO-N-terminal | 95 ± 5% | 30 ± 6 | Full | Minimal, requires careful ligand titration. |
| mCherry-C-terminal | 55 ± 20% | 150 ± 40 | Partial (~35%) | Severe aggregation, bundling at high expression. |
| FLAG-C-terminal (for IF) | 85 ± 10% | N/A | High | Subject to fixation artefacts only. |
Protocol: Validating Tagged EB1 Constructs
- Cloning: Insert FP tag at the N-terminus of the full-length EB1 ORF using a flexible linker (e.g., GGSGGS).
- Transfection: Use low-efficiency methods (e.g., lipofection with 0.5 µg DNA/35mm dish) or inducible promoters to achieve near-physiological expression levels.
- Validation by Rescue: a. Transfert EB1 knockout cell line with tagged EB1 construct. b. Fix using optimized protocol and co-stain for a complementary +TIP (e.g., APC). c. Quantify comet parameters and compare to wild-type cells. Successful rescue should match wild-type comet length and density, and restore normal APC co-localization.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Robust EB1 Localization Studies
| Item | Function & Rationale |
|---|---|
| Anti-EB1 Antibody (Clone 5/EB1) | Mouse monoclonal. Gold standard for IF of endogenous EB1; recognizes N-terminal epitope. |
| PHEM Buffer | Stabilizes cytoskeleton during extraction/fixation, preserving native EB1-microtubule interactions. |
| HALO Tag System | Self-labeling tag. Allows precise control of fluorophore stoichiometry and timing, reducing tag bulk. |
| TIRF Microscope | Total Internal Reflection Fluorescence. Essential for high-resolution, low-background imaging of EB1 comets at the cell cortex. |
| siRNA against EB1 (EB1-siRNA pool) | Validated siRNA for generating knockout/knockdown controls for rescue experiments. |
| Live-Cell Imaging Chamber (Tokai Hit) | Maintains 37°C, 5% CO2 for stable long-term live imaging of EB1 dynamics. |
| Microtubule Stabilizing Buffer (Paclitaxel) | Used in in vitro assays to validate EB1-microtubule binding affinity of tagged constructs. |
| FIJI/ImageJ Plugin "Comet Analyzer" | Automated tool for quantifying EB1 comet number, length, and intensity from time-lapse or fixed images. |
Visualizing EB1 Networks and Experimental Workflows
Diagram 1: EB1 in MT-Actin Crosstalk
Diagram 2: EB1 Study Pitfall Workflow
Optimizing Conditions for Co-Visualization of EB1 with Actin and Microtubules
The precise coordination between the microtubule and actin cytoskeletons is fundamental to processes such as cell division, migration, and polarity. EB1, a core microtubule plus-end tracking protein (+TIP), is emerging as a crucial node in actin-microtubule crosstalk. This whitepaper provides an in-depth technical guide for optimizing the simultaneous visualization of EB1, actin filaments, and microtubules—a critical methodological foundation for research within a broader thesis on EB1's role in cytoskeletal integration. Reliable co-visualization is a prerequisite for elucidating EB1's potential as a scaffold, signaling hub, or direct interactor with actin networks.
Key Considerations for Optimization
Successful co-visualization is challenged by the differing optimal fixation and permeabilization conditions for each cytoskeletal component, antibody incompatibilities, and spectral overlap.
- Fixation: Microtubules are best preserved by quick, warm formaldehyde fixation followed by methanol. Actin is best preserved by gentle formaldehyde fixation at room temperature. A balanced protocol is required.
- Permeabilization: Harsh detergents can disrupt delicate actin structures but are often needed for antibody penetration. Concentration and timing are critical.
- Antibody Selection: Primary antibodies must be raised in different host species. Fluorophore selection must minimize bleed-through.
- Live vs. Fixed Cell Imaging: While live imaging with fluorescent protein fusions is ideal for dynamics, fixed cell imaging allows for multiplexing with more markers and is the focus of this protocol.
Optimized Protocol for Fixed-Cell Co-Visualization
This protocol represents a consensus from recent literature, balancing preservation of all three targets.
Cell Preparation and Fixation
- Cell Culture: Plate cells on #1.5 high-resolution coverslips in appropriate media. For optimal microtubule polymerization, culture should be ~70% confluent and serum-starved (if applicable) for 2-4 hours prior to fixation to reduce background.
- Pre-extraction (Optional but Recommended): Briefly rinse cells in pre-warmed (37°C) PHEM buffer (60 mM PIPES, 25 mM HEPES, 10 mM EGTA, 2 mM MgCl₂, pH 6.9). Incubate for 30-60 seconds in PHEM buffer containing 0.1-0.3% Triton X-100 and 0.25% glutaraldehyde. This step removes soluble tubulin and preserves dynamic cytoskeletal structures.
- Fixation: Immediately add an equal volume of pre-warmed 4% formaldehyde (from paraformaldehyde, PFA) in PHEM buffer to the pre-extraction solution (final: 2% PFA, ~0.15% Triton, ~0.125% glutaraldehyde). Fix for 10 minutes at 37°C.
- Quenching & Permeabilization: Rinse 3x with PBS. Quench autofluorescence with 0.1% sodium borohydride in PBS for 10 min (or 100 mM glycine for 15 min). Rinse. For additional permeabilization (if antibodies require it), treat with 0.1% Triton X-100 in PBS for 10 min.
- Blocking: Block coverslips in 2-5% BSA (or serum matching the secondary antibody host) in PBS for 1 hour at room temperature.
Immunostaining
- Primary Antibody Incubation: Prepare primary antibody cocktail in blocking buffer. A typical combination is:
- Mouse anti-α-Tubulin (1:500-1:1000)
- Rabbit anti-EB1 (1:200-500)
- Phalloidin conjugate (e.g., Alexa Fluor 405/488) for F-actin (can be added here or with secondaries at 1:50-1:200). Apply cocktail to coverslip and incubate in a humidified chamber for 1-2 hours at RT or overnight at 4°C.
- Washing: Wash 4 x 5 minutes with PBS + 0.05% Tween-20 (PBST).
- Secondary Antibody Incubation: Prepare secondary antibody cocktail in blocking buffer, avoiding cross-reactive species. Use highly cross-absorbed antibodies. Include the phalloidin stain if not added earlier. Incubate for 45-60 minutes at RT in the dark.
- Final Wash & Mounting: Wash 4 x 5 minutes with PBST, then once with distilled water. Mount coverslips on glass slides using a hard-set, anti-fade mounting medium (e.g., ProLong Diamond, SlowFade Diamond).
Imaging Recommendations
- Use a high-NA (≥1.4) 60x or 100x oil immersion objective on a confocal or super-resolution microscope.
- Acquire channels sequentially to eliminate bleed-through.
- For EB1 comets, acquire z-stacks (0.2-0.3 µm steps) to capture plus-end distribution.
Table 1: Comparison of Fixation Methods for Cytoskeletal Preservation
| Fixation Method | Microtubule Preservation | Actin Filament Preservation | EB1+Tip Signal Intensity | Recommended Use |
|---|---|---|---|---|
| 4% PFA, 15 min, RT | Moderate (may be depolymerized) | Excellent | Low to Moderate | Primary actin studies |
| 100% Methanol, -20°C, 10 min | Excellent | Poor (disrupted) | High | Primary microtubule/EB1 studies |
| Pre-extraction + 2% PFA/0.125% GA, 37°C | Good to Excellent | Good | High | Optimized Co-Visualization |
| 10% TCA, 15 min, 4°C | Good | Good | Moderate | For specific phospho-epitopes |
Table 2: Common Antibody & Reagent Combinations
| Target | Primary Antibody/Reagent | Host/Type | Dilution | Compatible Fluorophore (Secondary) |
|---|---|---|---|---|
| Microtubules | Anti-α-Tubulin (DM1A) | Mouse monoclonal | 1:1000 | Alexa Fluor 568 (Goat anti-Mouse) |
| EB1 | Anti-EB1 (clone 5/EB1) | Rabbit monoclonal | 1:500 | Alexa Fluor 488 (Goat anti-Rabbit) |
| F-actin | Phalloidin | N/A (direct conjugate) | 1:200 | Alexa Fluor 647, CF405M |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Co-Visualization Experiments
| Item | Function & Rationale |
|---|---|
| PHEM Buffer | A physiologically buffered salt solution ideal for cytoskeletal preservation due to its optimal pH and Mg²⁺/EGTA levels. |
| 16% Formaldehyde (Methanol-free) | Provides consistent, high-quality PFA fixation without the depolymerization risks associated with commercial formalin. |
| Glutaraldehyde (25% stock) | Used at low concentration (0.1-0.25%) as a crosslinker to dramatically improve microtubule and EB1 preservation. |
| Triton X-100 | Non-ionic detergent for controlled permeabilization of the plasma membrane to allow antibody entry. |
| Sodium Borohydride (NaBH₄) | Reduces unreacted aldehyde groups from glutaraldehyde, quenching autofluorescence. |
| ProLong Diamond Antifade Mountant | A hard-cure, low-bleaching mounting medium with high refractive index, optimal for 3D super-resolution imaging. |
| #1.5 High-Precision Coverslips (0.17 mm thick) | Essential for optimal performance of high-NA oil immersion objectives and minimal spherical aberration. |
| Cross-adsorbed Secondary Antibodies | Antibodies pre-adsorbed against serum proteins of multiple species to minimize off-target binding in multiplex experiments. |
| Alexa Fluor Phalloidin Conjugates | Highly specific, bright, and photostable probes for F-actin labeling across a wide range of wavelengths. |
Experimental Workflow & Pathway Diagrams
Title: Optimized Co-Visualization Experimental Workflow
Title: EB1 in Actin-Microtubule Crosstalk Context
Within the study of cytoskeletal dynamics and actin-microtubule (MT) crosstalk, End-Binding protein 1 (EB1) is a central regulator. It binds to the growing plus-ends of MTs, tracking their polymerization and recruiting a diverse array of proteins. A significant challenge in interpreting experimental phenotypes following EB1 perturbation (knockdown, knockout, or overexpression) lies in distinguishing primary, direct effects on MT dynamics and associated protein recruitment from secondary, indirect consequences resulting from widespread cytoskeletal collapse and cellular stress. This guide provides a technical framework for making this critical distinction, a necessity for accurate mechanistic insight and for the validation of EB1 as a therapeutic target in processes like cell division, migration, and intracellular transport.
EB1 Function: A Primer for Phenotype Interpretation
EB1's canonical function is as a core component of the MT plus-end tracking protein (+TIP) network. Its direct molecular interactions form the basis for distinguishing primary from secondary effects.
Primary/Direct EB1 Interactions & Functions:
- MT Polymerization Tracking: Binds the GTP-cap of growing MTs via its N-terminal Calponin Homology (CH) domain.
- +TIP Recruitment: Its C-terminal EEY/F motif serves as a docking site for proteins containing CAP-Gly or SxIP motifs (e.g., APC, CLIP-170, p150Glued of dynactin).
- MT Stabilization: Promotes persistent growth and reduces catastrophe frequency.
- Actin-MT Crosstalk Direct Signaling: Directly or indirectly (via +TIPs) interacts with actin regulators (e.g., Formins, IQGAP1) to coordinate cytoskeletal dynamics.
Secondary/Indirect Consequences of EB1 Disruption:
- Global MT Destabilization: Loss of polymerizing, stable MTs.
- Organelle Transport Defects: Disruption of dynein/kinesin trafficking due to impaired MT tracks and +TIP recruitment.
- Actin Network Disorganization: Cascading effects from loss of coordinated crosstalk.
- Cell Cycle Arrest: Mitotic spindle defects leading to SAC activation.
- Altered Gene Expression: Stress responses triggered by gross morphological and functional disruption.
Table 1: Quantitative Metrics to Distinguish Direct from Secondary EB1 Phenotypes
| Parameter Category | Primary/Direct EB1 Effect (Early, Specific) | Secondary/Collapse Phenotype (Late, Broad) | Recommended Measurement Technique |
|---|---|---|---|
| MT Dynamics | Altered growth rate/speed; Modest change in catastrophe frequency. | Severe suppression of growth; Near-complete loss of dynamic MTs; Massive increase in catastrophe. | Live-cell imaging of EB3-GFP or +TIP comets; MT speckle microscopy. |
| +TIP Localization | Specific loss or redistribution of a subset of +TIPs (e.g., APC but not CLASP). | Global loss of all +TIPs from MT ends. | Fluorescence recovery after photobleaching (FRAP) of +TIPs; co-localization analysis. |
| Actin Structure | Altered actin polymerization at specific domains (e.g., leading edge, cortex) in coordination with MTs. | Global actin disassembly or uncontrolled polymerization (stress fiber formation). | F-actin staining (Phalloidin); FRAP of actin reporters. |
| Temporal Onset | Phenotypes observable within minutes of acute perturbation (e.g., via auxin-inducible degron). | Phenotypes develop over hours, following primary MT destabilization. | Time-lapse imaging post-acute perturbation. |
| Phenotype Rescue | Rescued by EB1 wild-type, but not mutants defective in specific interactions (e.g., ΔC-term). | Only rescued by global MT stabilizers (e.g., Taxol) or EB1 WT. | Complementation assays with EB1 mutants. |
| Cellular Viability | Minimal immediate impact on viability; specific functional defects (e.g., directional migration). | Significant cell death, apoptosis, or irreversible cell cycle arrest. ATP depletion. | Annexin V/PI staining; ATP assays; long-term survival assays. |
Experimental Protocols for Distinction
Protocol 4.1: Acute vs. Chronic Perturbation Time-Course
Objective: To separate early, direct phenotypes from late, collapse-related ones. Method:
- Cell Line Generation: Stably express EB1 fused to an auxin-inducible degron (AID) tag in a background expressing plant TIR1.
- Acute Depletion: Treat cells with 500 µM auxin (IAA) and image at defined intervals (0, 5, 15, 30, 60, 120, 240 minutes).
- Chronic Depletion: Perform siRNA-mediated EB1 knockdown (48-72 hrs) for comparison.
- Analysis: Quantify MT growth speed, +TIP comet density, and actin morphology at each time point. Direct effects will plateau early; secondary phenotypes will intensify over time.
Protocol 4.2: Pharmacological Dissection with Cytoskeletal Stabilizers
Objective: To determine if a phenotype is downstream of MT collapse. Method:
- Induce EB1 Knockdown: Transfect with EB1-specific siRNA.
- Rescue Conditions: At 24h post-transfection, add either:
- DMSO (vehicle control)
- Paclitaxel/Taxol (10 nM, to stabilize MTs globally)
- Low-dose Nocodazole (5 nM, to promote dynamic instability without full collapse)
- Assay: At 48h, fix cells and quantify the phenotype of interest (e.g., Golgi fragmentation, focal adhesion size). Rescue by Taxol suggests a secondary effect of MT loss.
Protocol 4.3: Mutant Rescue Complementation Assay
Objective: To link a phenotype to a specific biochemical function of EB1. Method:
- Create Rescue Constructs: Generate siRNA-resistant EB1 constructs: EB1-WT, EB1-ΔC-term (cannot bind +TIPs), EB1-Liss/KK (impaired MT binding).
- Co-transfection: Co-transfect EB1-targeting siRNA with one of the rescue constructs into cells.
- Analysis: After 48h, assess for phenotypic rescue. If EB1-WT, but not EB1-ΔC-term, rescues a +TIP localization defect, it is a direct effect. If neither rescues a viability defect, it may be a secondary or combined effect.
Visualizing the Logical Framework and Pathways
Diagram 1: Logical Framework for Phenotype Analysis
Diagram 2: Signaling Pathways in Direct vs. Indirect Actin Remodeling
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Distinguishing EB1 Phenotypes
| Reagent / Tool | Category | Primary Function in This Context | Example Source / Cat. # |
|---|---|---|---|
| EB1-Targeting siRNA/sgRNA | Perturbation | For specific, RNA-mediated depletion of EB1 to initiate phenotype. | Dharmacon, Sigma-Aldrich |
| AID-tagged EB1 Cell Line | Perturbation | Enables rapid, acute degradation of EB1 upon auxin addition for precise temporal analysis. | Generated via CRISPR/Cas9 editing. |
| Paclitaxel (Taxol) | Pharmacologic | MT stabilizer; used to test if a phenotype is secondary to general MT collapse. | Sigma-Aldrich T7191 |
| siRNA-Resistant EB1 Plasmids (WT, ΔC, Liss/KK) | Molecular Biology | For mutant rescue experiments to map phenotypic domains of EB1. | Addgene (various), custom cloning. |
| EB3-GFP/TdTomato | Live Imaging | A validated EB1-family +TIP reporter for quantifying MT growth dynamics in live cells. | Addgene plasmid #39299 |
| Anti-EB1 Antibody (Clone 5/EB1) | Immunofluorescence | To visualize endogenous EB1 comet distribution and density. | BD Biosciences 610535 |
| Fluorescently-labeled Paclitaxel (e.g., Flutax-2) | Staining | Directly labels stable MT polymer mass, independent of +TIPs. | Tocris 5638 |
| LifeAct-GFP/RFP | Live Imaging | To visualize F-actin dynamics concurrently with MTs. | Ibidi #60102 |
| G-LISA RhoA Activation Assay | Biochemical | Quantifies activation of the RhoA stress-response pathway following cytoskeletal collapse. | Cytoskeleton #BK124 |
Within the broader thesis on EB1 protein function in actin-microtubule crosstalk research, pharmacological tools are indispensable for probing cytoskeletal dynamics. EB1 is a core microtubule plus-end tracking protein (+TIP) that regulates microtubule dynamics, interacts with actin-binding proteins, and coordinates cytoskeletal crosstalk. Microtubule-targeting drugs (MTDs) are widely used to manipulate the microtubule cytoskeleton, but their effects on EB1 localization, dynamics, and function are often indirect, non-specific, and poorly characterized. This whitepaper provides a technical guide to the limitations and specificity of major MTD classes on EB1, with a focus on implications for actin-microtubule interaction studies.
EB1 Function and Actin-Microtubule Crosstalk Context
EB1 binds to the GTP-cap of growing microtubule plus-ends, promoting persistent growth and recruiting a network of +TIPs. In actin-microtubule crosstalk, EB1 serves as a crucial platform, linking microtubule ends to actin filaments via proteins like MACF, ACF7, and formins. Disrupting EB1-microtubule interaction with pharmacological agents thus perturbs not only microtubule dynamics but also the spatial coordination of cytoskeletal networks, making the specificity of such tools a critical concern.
Classes of Microtubule-Targeting Drugs and Their Impact on EB1
Microtubule-Stabilizing Agents (MSAs)
Primary Drugs: Paclitaxel (Taxol), Docetaxel, Epothilones. Mechanism: Bind to β-tubulin, stabilize microtubule lattice, suppress dynamic instability. Effect on EB1: High concentrations eliminate EB1 comet-like plus-end tracking by locking microtubules in a persistent GTP-like state, effectively "hiding" the EB1 binding site. Low, substoichiometric concentrations can paradoxically increase microtubule growth rates and EB1 comet intensity.
Microtubule-Destabilizing Agents (MDAs)
Subclass 1: Vinca Alkaloids (Vinblastine, Vincristine) Mechanism: Bind to tubulin dimers, induce depolymerization or spiral aggregates, suppress dynamics. Effect on EB1: Deplete polymerized microtubules, leading to loss of EB1 from plus-ends. At low concentrations, can increase catastrophe frequency, shortening EB1 comet dwell time. Subclass 2: Colchicine Site Binders (Colchicine, Combretastatin A-4) Mechanism: Bind to tubulin dimers, induce curved conformation, inhibit polymerization. Effect on EB1: Microtubule depolymerization eliminates EB1 binding substrate. Colchicine's slow kinetics complicate acute experiments.
Suppressors of Microtubule Dynamics
Drugs: Nocodazole (reversible), Maytansine Mechanism: Bind to tubulin, suppress polymerization/depolymerization rates without major mass change. Effect on EB1: Nocodazole, widely used for microtubule "depolymerization," at low nanomolar concentrations suppresses dynamics while preserving microtubule mass, leading to diminished but not absent EB1 tracking. Rapidly reversible.
Table 1: Quantitative Effects of Microtubule-Targeting Drugs on EB1 Parameters
| Drug (Class) | Typical Working Concentration | Effect on Microtubule Mass | Effect on EB1 Comet Intensity | Effect on EB1 Comet Number | Key Limitation for EB1 Studies |
|---|---|---|---|---|---|
| Paclitaxel (MSA) | 10 nM - 10 µM | Increased | High [ ]: Abolished; Low [ ]: Increased | Decreased | Eliminates dynamic ends, mislocalizes EB1 along lattice |
| Nocodazole (MDA) | 100 nM - 10 µM | Decreased (high [ ]) | Abolished | Abolished | Global depolymerization, non-specific for plus-ends |
| Vinblastine (MDA) | 1-100 nM | Decreased | Abolished | Abolished | Induces tubulin aggregates, off-target effects |
| GSK-923295 (CENP-E inhibitor) | 50-500 nM | Minimal change | Moderate decrease | Minimal change | Specifically inhibits kinesin, indirect EB1 effect |
Key Experimental Protocols for Assessing Drug Effects on EB1
Protocol: Live-Cell Imaging of EB1 Comet Dynamics Post-Drug Treatment
Objective: Quantify changes in EB1 comet parameters (intensity, frequency, speed, lifetime) after MTD application. Materials: U2OS or COS-7 cells expressing EB3-GFP (EB1 homolog), glass-bottom dishes, spinning-disk confocal microscope, imaging chamber with temperature/CO2 control. Procedure:
- Plate cells, transfect with EB3-GFP plasmid using lipofection.
- 24h post-transfection, mount dish in chamber at 37°C, 5% CO2.
- Acquire 5 pre-treatment time-lapse series (2-sec intervals for 2 min) using 488nm laser, 100x oil objective.
- Carefully add pre-warmed drug solution (or DMSO vehicle) to medium.
- After 15-30 min incubation, acquire post-treatment time-lapse series (identical settings).
- Analyze using plusTipTracker (Matlab) or TrackMate (Fiji): detect comets, track over time, extract parameters.
- Normalize post-treatment data to pre-treatment control for each cell.
Protocol: Co-immunoprecipitation of EB1 Complexes Under Drug Treatment
Objective: Determine if MTDs disrupt EB1 interaction with binding partners (e.g., APC, CLIP-170) in actin-microtubule crosstalk. Materials: HEK293T cells, EB1 antibody (mouse monoclonal), protein A/G beads, lysis buffer (50 mM Tris pH 7.4, 150 mM NaCl, 1% NP-40, protease inhibitors). Procedure:
- Treat cells with drug (e.g., 100 nM paclitaxel, 10 µM nocodazole) or DMSO for 30 min.
- Lyse cells on ice for 20 min, centrifuge at 15,000g for 15 min.
- Incubate supernatant with 2 µg anti-EB1 or IgG control overnight at 4°C.
- Add beads, incubate 2h, wash 4x with lysis buffer.
- Elute with Laemmli buffer, analyze by SDS-PAGE and western blot for EB1, APC, CLIP-170, and β-actin (loading control).
Visualizing Signaling Pathways and Experimental Workflows
Diagram Title: How MTDs disrupt EB1-mediated actin-microtubule crosstalk
Diagram Title: Workflow for analyzing EB1 drug response
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for EB1-Drug Studies
| Reagent/Material | Function in Experiment | Key Consideration |
|---|---|---|
| EB1/EB3-GFP Plasmid (Addgene #39299) | Live-cell visualization of microtubule plus-end dynamics | Use EB3 for brighter comets; verify localization. |
| CellLight EB3-RFP BacMam 2.0 (Thermo Fisher) | Label endogenous EB3 without transfection | Lower expression level, more physiological. |
| SiR-Tubulin (Spirochrome) | Far-red live-cell microtubule stain | Low cytotoxicity, compatible with GFP; use with verapamil. |
| PlusTipTracker Software (MATLAB) | Automated detection and tracking of EB comets | Requires MATLAB license; steep learning curve. |
| Anti-EB1 Antibody [clone 5/EB1] (BD Biosciences) | Immunoprecipitation and immunofluorescence | Mouse monoclonal, works well for western blot and IP. |
| Microtubule/Tubulin Biochemistry Kit (Cytoskeleton Inc.) | In vitro assay of drug effects on tubulin polymerization | Isolate direct vs. cellular effects. |
| GSK-923295 (Tocris) | Specific kinesin CENP-E inhibitor | Negative control for non-MTD effects on EB1. |
| Glass-bottom 35mm Dishes (MatTek) | High-resolution live-cell imaging | #1.5 coverslip thickness optimal for oil objectives. |
Limitations and Specificity Considerations
- Off-Target Effects: Many MTDs affect actin dynamics indirectly. Paclitaxel can alter actin stress fibers via RhoA activation.
- Dose Dependence: Effects are highly concentration-dependent (e.g., low vs. high paclitaxel).
- Kinetics: Drug onset/offset times vary (nocodazole is fast/reversible; colchicine is slow).
- Cellular Context: Drug effects differ by cell type due to variable tubulin isotype expression and efflux pumps.
- EB1 vs. EB3: Most drugs affect both, but EB3 may be more sensitive to certain destabilizers.
Best Practices and Future Directions
For actin-microtubule crosstalk research, use complementary approaches:
- Combine acute drug treatment with EB1 knockdown/rescue to isolate EB1-specific functions.
- Employ sub-nanomolar, acute doses to suppress dynamics without mass changes.
- Utilize specific protein-protein interaction disruptors (e.g., EB1-targeting peptides) over broad MTDs.
- Correlate live-cell EB1 imaging with actin markers (LifeAct) to directly visualize crosstalk perturbation. Future pharmacological development should aim for compounds that specifically disrupt EB1 recruitment or its interaction with actin-binding factors, offering greater precision than current tubulin-centric drugs.
Best Practices for Quantifying EB1 Comet Parameters and Correlation with Cell Behavior
Within the broader investigation of EB1 protein function in actin-microtubule crosstalk, precise quantification of EB1 comets—fluorescent marks of growing microtubule plus-ends—is paramount. These dynamic parameters serve as critical readouts of microtubule polymerization stability and regulation, which are directly modulated by actin network interactions. This guide details standardized methodologies for capturing, analyzing, and correlating EB1 comet metrics with cellular phenotypes, providing a technical foundation for research in cytoskeletal dynamics and targeted drug development.
I. Experimental Protocols for Live-Cell Imaging
A. Cell Preparation and Transfection
- Cell Line Selection: Use cells with well-defined cytoskeletal architecture (e.g., U2OS, HeLa, REF-52, or primary fibroblasts). For crosstalk studies, consider lines amenable to dual cytoskeletal perturbation.
- Fluorescent Labeling:
- Transfect with EB1-GFP, EB1-mCherry, or similar fluorescent protein (FP) fusions. Use low-expression vectors to avoid overexpression artifacts.
- Alternative: Use immunofluorescence staining with anti-EB1 antibodies in fixed cells, though this limits dynamic analysis.
- Contextual Staining (Optional but Recommended): For crosstalk studies, co-stain F-actin (with phalloidin) or use LifeAct-FP to visualize the actin network in a separate channel.
B. Image Acquisition
- Microscope: Spinning-disk or point-scanning confocal microscope for optimal z-resolution and speed.
- Settings:
- Temporal Resolution: Acquire images every 1-3 seconds for 2-5 minutes. Faster rates (500ms) may be needed for high-speed events.
- Spatial Resolution: Use a 60x or 100x oil-immersion objective (NA ≥ 1.4). Pixel size should be ≤ 100 nm.
- Z-stacks: Acquire 5-7 slices with a step size of 0.5-1.0 µm to capture entire comet trajectories.
- Environmental Control: Maintain 37°C and 5% CO₂ for live-cell integrity.
II. Core Quantitative Parameters & Data Presentation
EB1 comet analysis yields the following key metrics, which should be extracted for each detected comet and summarized per cell and condition.
Table 1: Core EB1 Comet Parameters for Quantification
| Parameter | Description | Biological Significance | Typical Range (Mammalian Cell) |
|---|---|---|---|
| Comet Density | Number of comets per unit area (e.g., #/µm²). | Indicates number of actively growing microtubule plus-ends. | 0.05 - 0.15 /µm² |
| Comet Intensity | Maximum fluorescence intensity of a comet. | Proportional to EB1 protein load, potentially linked to plus-end stability. | Variable (a.u.) |
| Comet Speed (Growth Rate) | Distance traveled by comet front between frames (µm/min). | Direct readout of microtubule polymerization rate. | 15 - 25 µm/min |
| Comet Lifetime / Track Duration | Time from first to last appearance (s). | Reflects persistence of growth before catastrophe or pausing. | 20 - 60 s |
| Comet Length | Distance between comet front and base (µm). | Relates to the "tip structure" and EB1 binding zone. | 1.5 - 3.0 µm |
Table 2: Correlation of EB1 Parameters with Cell Behavior in Actin-MT Crosstalk Context
| EB1 Parameter Alteration | Associated Cell Behavior Phenotype | Potential Actin-Crosstalk Mechanism Implied |
|---|---|---|
| Decreased Comet Density | Reduced cell polarity, impaired directional migration. | Actin arcs or cortactin inhibiting MT nucleation/exploration at cell edge. |
| Decreased Comet Speed | Slower protrusion, reduced invasion potential. | Dense actin mesh physically impeding MT growth; myosin-II contractility. |
| Increased Comet Lifetime | Stabilized leading edge, persistent migration. | Actin bundles (e.g., filopodia) providing "tracks" for stabilized MT growth. |
| Asymmetric Comet Distribution | Directed migration, polarized vesicle transport. | Polarized actin flow guiding preferential MT growth direction. |
III. Image Analysis Workflow
A standardized computational pipeline is essential for reproducibility.
Diagram 1: EB1 Comet Analysis Workflow
Detailed Protocol:
- Pre-processing: Generate maximum intensity z-projections. Apply rolling-ball or Gaussian blur background subtraction.
- Detection: Use a Laplacian of Gaussian (LoG) or Difference of Gaussian (DoG) band-pass filter to enhance comet-like features. Apply a user-defined intensity threshold.
- Tracking: Utilize algorithms like the u-track framework or TrackMate (Fiji/ImageJ). Key settings: maximum linking distance (~2 µm), maximum gap-closing distance (~3 µm), minimum track length (3-4 frames).
- Quantification: Export track data (X,Y position, time, intensity) for calculation of parameters in Table 1 using custom scripts (Python/MATLAB) or software plugins.
- Correlation: Plot EB1 parameters against cell behavior metrics (e.g., migration speed from cell tracking, protrusion/retraction dynamics from edge analysis).
IV. The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Reagent Solutions for EB1-Actin Crosstalk Studies
| Reagent / Material | Function & Application in EB1 Studies | Example / Catalog Considerations |
|---|---|---|
| EB1 Expression Vectors | Live-cell labeling of microtubule plus-ends. | EB1-GFP (Addgene #39299), EB1-mCherry, EB1-TagRFP. Use low-CMV or endogenous promoters. |
| Actin Live-Cell Probes | Parallel visualization of actin dynamics. | LifeAct-GFP/mCherry, F-tractin, or stable actin-FP cell lines. |
| Microtubule Stabilizers/Destabilizers | Pharmacological controls for comet parameter validation. | Paclitaxel (stabilizer, 10-100 nM), Nocodazole (destabilizer, 100 nM - 1 µM). |
| Actin Network Modulators | To perturb actin and study its effect on EB1 comets. | Latrunculin A (depolymerizer, 50-500 nM), Jasplakinolide (stabilizer, 100 nM). |
| ROCK or Myosin II Inhibitors | To modulate actin contractility's impact on MT growth. | Y-27632 (ROCKi, 10 µM), Blebbistatin (Myosin IIi, 10-50 µM). |
| Fixatives for Co-staining | For correlative static imaging of EB1 and actin. | Paraformaldehyde (4%) in PEM buffer (100 mM PIPES, 1 mM EGTA, 1 mM MgCl₂, pH 6.8). |
| Mounting Medium | For preserving fluorescence in fixed samples. | Use anti-fade medium (e.g., with DABCO or commercial ProLong). |
| Analysis Software | For image processing, tracking, and data analysis. | Fiji/ImageJ + TrackMate, MetaMorph, Imaris, or custom Python code using scikit-image & Trackpy. |
V. Integrating EB1 Data into Actin-Microtubule Crosstalk Pathways
EB1 comet behavior is a key integrator of signaling between cytoskeletal networks.
Diagram 2: EB1 in Actin-MT Crosstalk Signaling
Interpretation: Diagram 2 illustrates two primary crosstalk modes: 1) Mechanical Crosstalk (green), where physical actin structures directly impede or guide MT growth, altering EB1 comet parameters. 2) Signaling Hub Crosstalk (gray), where Rho GTPase pathways coordinately regulate both actin effectors and microtubule-associated proteins (+TIPs like CLASPs), which converge to modulate EB1 comet behavior, ultimately driving integrated cell behavior.
- Controls: Always include positive (e.g., EB1 overexpression) and negative (e.g., EB1 siRNA, nocodazole) controls for comet analysis.
- Blinding: Perform image analysis blinded to experimental conditions.
- Sample Size: Analyze a minimum of 15-20 cells per condition, from at least 3 independent experiments.
- Contextual Imaging: When correlating with cell behavior, ensure the imaging field captures relevant cellular structures (e.g., leading edge, Golgi apparatus).
- Data Reporting: Present data as mean ± SEM or median with interquartile range. Use appropriate statistical tests (e.g., Mann-Whitney U test for non-normal distributions).
EB1 in Context: Validating Its Unique Role Among Cytoskeletal Regulators
Within the cellular cytoskeleton, actin filaments and microtubules engage in intricate crosstalk to orchestrate cell division, migration, and polarization. End-binding protein 1 (EB1), a core master regulator of microtubule plus-end dynamics, is a central node in this interaction network. This whitepaper, framed within a broader thesis on EB1's multifunctionality in cytoskeletal crosstalk, provides a technical comparison of EB1 against other prominent +TIPs—specifically CLASPs and XMAP215/TOG-domain proteins. We dissect their distinct and overlapping roles in mediating actin-microtubule interactions, supported by current mechanistic data and quantitative analyses.
Molecular Mechanisms and Functional Domains
EB1, CLASPs, and XMAP215 share the fundamental property of localizing to polymerizing microtubule plus-ends but possess unique structural domains that dictate their crosstalk functions.
- EB1: Comprises an N-terminal calponin homology (CH) domain for microtubule lattice binding, a flexible linker, and a C-terminal EB homology (EBH) domain that recruits a vast array of partner proteins (+TIP interactome). Its primary crosstalk function is as an adaptor/scaffold. EB1 does not directly bind actin but recruits intermediates like APC, MACF, and Spectraplakins that bridge to actin filaments.
- CLASP (Cytoplasmic Linker Associated Protein): Characterized by TOG-like domains and a serine/arginine-rich (S/R) region. CLASPs promote microtubule rescue and pause. In crosstalk, they often act as cooperative stabilizers with EB1, and certain isoforms can directly or indirectly (via CLIP-170) associate with actin cortical networks, guiding microtubule capture at specific actin-rich sites.
- XMAP215 (Dis1/TOG family): Defined by multiple TOG domains that directly bind tubulin dimers. It is a potent microtubule polymerase. Its role in crosstalk is less direct than EB1's but critical for generating the dynamic microtubule populations that engage with actin structures. It can be recruited to plus-ends via EB1.
Table 1: Core +TIP Protein Domain Architecture and Primary Crosstalk Mechanism
| +TIP Protein | Key Domains | Direct Actin Binding? | Primary Crosstalk Mechanism | Key Actin-/Cortex-Interacting Partners |
|---|---|---|---|---|
| EB1 | CH domain, EBH domain | No | Scaffold/Adaptor: Recruits actin-linking effectors to +TIPs. | APC, MACF, Spectraplakins, DCLK1 |
| CLASP | TOG domains, S/R region | Indirect/Context-dependent | Cooperative Stabilizer: Guides MT capture at cortical actin; works with EB1. | CLIP-170, Cortical actin meshwork |
| XMAP215 | TOG domains (typically 5) | No | Polymerase Provider: Generates dynamic MTs for interaction; can be EB1-recruited. | Primarily via tubulin polymerization regulation |
Quantitative Functional Comparison
The functional output of these +TIPs in crosstalk is measurable through parameters of microtubule dynamics and interaction frequency with actin structures.
Table 2: Quantitative Impact on Microtubule Dynamics and Actin Engagement
| Parameter | EB1 Knockdown/Inhibition | CLASP Knockdown/Inhibition | XMAP215 Knockdown/Inhibition |
|---|---|---|---|
| Microtubule Growth Rate | Decreased (~30-50%) | Mild decrease or altered | Severely decreased (~70-80%) |
| Microtubule Catastrophe Frequency | Increased | Decreased (loss of rescue) | Increased |
| Microtubule Rescue Frequency | Decreased | Severely Decreased | Mild/no effect |
| Duration of MT-Actin Contact Events | Significantly Reduced | Reduced at cortical sites | Minimally affected |
| Frequency of MT Targeting to Focal Adhesions | Reduced | Reduced | Largely unchanged (unless growth rate is too slow) |
Key Experimental Protocols for Crosstalk Analysis
4.1. Simultaneous Live-Cell Imaging of MT-Actin Interactions
- Purpose: To visualize and quantify the co-dynamics of microtubule plus-ends and actin structures.
- Protocol:
- Cell Line & Transfection: Use COS-7, U2OS, or fibroblast cells. Co-transfect with fluorescently tagged EB3 (an EB1 family member, superior for live imaging) and LifeAct for actin visualization.
- Imaging Setup: Employ TIRF or highly inclined thin illumination (HILO) microscopy on a spinning disk confocal system. Maintain environment at 37°C and 5% CO₂.
- Acquisition: Capture dual-channel time-lapse images at 2-5 second intervals for 5-10 minutes.
- Analysis: Use plusTipTracker (MATLAB) or TrackMate (Fiji) to track EB3 comets. Manually or algorithmically score interactions (e.g., comet pausing, co-localization) with actin stress fibers or focal adhesions.
4.2. FRAP at the Microtubule Plus-End to Probe +TIP Exchange
- Purpose: To compare the turnover kinetics and stability of different +TIPs at the growing plus-end, indicative of their regulatory role.
- Protocol:
- Sample Prep: Express GFP-tagged EB1, CLASP2, or ch-TOG/XMAP215 in cells.
- Photobleaching: Select a single, growing microtubule plus-end (GFP comet) and subject it to a high-intensity laser pulse to bleach the fluorescence.
- Recovery Imaging: Acquire images at 500ms intervals post-bleach.
- Quantification: Plot fluorescence recovery over time. Fit curve to calculate half-time of recovery (t₁/₂) and mobile fraction. EB1 typically shows rapid recovery (t₁/₂ < 2s), while CLASP and XMAP215 may exhibit slower, more variable recovery.
4.3. In Vitro Reconstitution of MT-Actin Tethering
- Purpose: To dissect the minimal components required for crosstalk and test the necessity of EB1 as a scaffold.
- Protocol:
- Components: Purify tubulin, actin, EB1, a candidate linker (e.g., MACF N-terminus), and use rhodamine/phalloidin-stabilized actin filaments.
- Flow Chamber Preparation: Create a passivated flow chamber. Introduce biotinylated GMPCPP-stabilized microtubule seeds nucleated from immobilized antibodies.
- Reaction Mix: Flow in a mix of tubulin (with ~10% HiLyte-fluor), EB1, linker protein, and oxygen scavengers/catalase for dynamic microtubules.
- Imaging & Analysis: Use TIRF microscopy. Quantify the percentage of growing microtubule plus-ends that become physically associated with surface-immobilized actin filaments in the presence/absence of EB1 and/or the linker.
Visualizing Signaling Pathways and Workflows
Diagram 1: +TIP Roles in MT-Actin Crosstalk Network
Diagram 2: In Vitro MT-Actin Tethering Assay Workflow
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for +TIP Crosstalk Research
| Reagent/Solution | Function & Application | Example Product/Source |
|---|---|---|
| Anti-EB1 Monoclonal Antibody | Immunofluorescence, Western Blot, and functional inhibition (microinjection). | Clone 5/EB1, Mouse IgG1 (BD Biosciences). |
| siRNA/shRNA Libraries for +TIPs | Gene knockdown to study loss-of-function phenotypes in crosstalk. | ON-TARGETplus SMARTpools (Horizon Discovery). |
| Fluorescently Labeled Tubulin (HiLyte, ATTO) | For visualizing dynamic microtubules in live-cell or in vitro assays. | Cytoskeleton Inc., or in-house labeling kits. |
| LifeAct-TagGFP2 / mRuby2 | F-actin visualization with minimal perturbation of actin dynamics. | ibidi GmbH; vector from Addgene. |
| GMPCPP (Non-hydrolyzable GTP analog) | To generate stable, short microtubule "seeds" for in vitro dynamics assays. | Jena Bioscience. |
| Recombinant +TIP Proteins (His-/GST-tagged) | For in vitro biochemistry, structural studies, and reconstitution assays. | Purified from E. coli or baculovirus systems (custom services). |
| Microtubule/Tubulin Binding Spin-Down Assay Kits | To quantify protein-microtubule affinity in presence/absence of actin linkers. | Cytoskeleton Inc. (BK029). |
| TIRF-Compatible Immobilization Chambers | For high-resolution single-molecule imaging and reconstitution experiments. | µ-Slide VI 0.5 (ibidi) or custom quartz chambers. |
This whitepaper is framed within a broader thesis on EB1 protein function, which is central to understanding actin-microtubule crosstalk—a critical process in cell division, migration, and polarity. The End-Binding (EB) family of proteins (EB1, EB2, and EB3) are core components of the microtubule plus-end tracking protein (+TIP) network. While they share structural homology and the ability to autonomously track growing microtubule ends, emerging research reveals nuanced functional redundancy and specificity among these isoforms. This guide provides a detailed technical comparison, focusing on their roles in cytoskeletal dynamics and integration with actin networks.
Structural and Biochemical Comparison
EB proteins share a conserved architecture: an N-terminal Calponin Homology (CH) domain for microtubule binding, a linker region, and a C-terminal EB homology (EBH) domain that mediates interactions with other +TIPs via a coiled-coil and an acidic tail. Key differences reside in their intrinsically disordered C-terminal tails, which dictate distinct binding partners and regulation.
Table 1: Core Characteristics of EB Isoforms
| Feature | EB1 (MAPRE1) | EB2 (MAPRE2) | EB3 (MAPRE3) |
|---|---|---|---|
| Primary Expression | Ubiquitous | Epithelial, neuronal tissues | Neuronal, muscle tissues |
| Microtubule Binding Affinity (Kd, approx.) | ~0.3 µM | ~0.5 µM | ~0.4 µM |
| Comet Tracking Velocity | Matches MT growth (~15 µm/min) | Matches MT growth (~15 µm/min) | Matches MT growth (~15 µm/min) |
| Key Binding Motif | SxIP in partners | SxIP & LxxPTPh | SxIP in partners |
| Dimerization State | Homodimer | Homodimer | Homodimer |
| Phosphorylation Regulation | CDK1, Aurora B | PKA, CaMKII | PKA, MARK2 |
| Role in Actin Crosstalk | Links to F-actin via APC, Spectraplakins | Less characterized | Direct link via Drebrin |
Functional Redundancy and Specificity in Cellular Processes
Microtubule Dynamics and +TIP Network Assembly
All EB isoforms promote microtubule growth and rescue catastrophes by recruiting stabilizing +TIPs. They exhibit functional redundancy in basic plus-end tracking and stabilization in many cell types. However, specificity arises in their spatiotemporal expression and partner recruitment.
Experimental Protocol: Comet Reconmentation Assay
- Objective: To compare the microtubule plus-end tracking dynamics of purified GFP-tagged EB isoforms.
- Materials: Rhodamine-labeled, GMPCPP-stabilized microtubule seeds, tubulin, GFP-EB1/2/3, TIRF microscopy buffer.
- Method:
- Flow chamber is coated with anti-rhodamine antibodies.
- Rhodamine-MT seeds are adhered to the chamber.
- A mixture of unlabeled tubulin (20 µM), GFP-EB (50 nM), and an oxygen scavenging system is introduced.
- Dynamic microtubule growth is initiated and imaged via Total Internal Reflection Fluorescence (TIRF) microscopy at 1 frame/sec.
- Kymograph analysis is used to determine comet intensity, length, and tracking velocity.
Specific Roles in Actin-Microtubule Crosstalk
Within the thesis context of actin-microtubule crosstalk, isoform specificity is paramount.
- EB1 is a central hub, linking microtubules to actin via large adaptors like APC and Spectraplakins (e.g., MACF1), guiding microtubule growth along actin stress fibers.
- EB3 exhibits neuron-specific crosstalk, directly binding the actin-binding protein Drebrin. This EB3-Drebrin complex is crucial for steering microtubules into dendritic spines, modulating synaptic plasticity.
- EB2's role in crosstalk is less clear but may involve unique epithelial-specific pathways.
Experimental Protocol: Co-sedimentation Assay for Direct Actin-Binding Partner Interaction
- Objective: To test direct binding of EB isoforms (or their partners) to F-actin.
- Materials: Purified EB protein, F-actin (from muscle actin), co-sedimentation buffer (10 mM imidazole pH 7.0, 50 mM KCl, 1 mM MgCl2, 1 mM EGTA, 0.2 mM DTT).
- Method:
- F-actin (5 µM) is polymerized for 1 hour at room temperature.
- EB protein (2 µM) is mixed with F-actin in a 100 µL reaction and incubated for 30 min.
- The mixture is ultracentrifuged at 100,000 x g for 30 min at 24°C.
- Supernatant (S) and pellet (P) fractions are separated, boiled in SDS sample buffer, and analyzed by SDS-PAGE and Coomassie staining/Western blot.
- The percentage of EB protein co-sedimenting with F-actin is quantified.
Quantitative Comparison of Isoform-Specific Interactions
Table 2: Quantitative Binding Data for Select EB Partners
| Interaction | EB1 | EB2 | EB3 | Assay Type | Reference (Example) |
|---|---|---|---|---|---|
| APC (SxIP motif) | Kd = ~0.2 µM | Very weak | Kd = ~0.5 µM | ITC | Buey et al., 2011 |
| Drebrin | No binding | No binding | Kd = ~0.8 µM | SPR | Geraldo et al., 2008 |
| CLIP-170 | Strong | Moderate | Strong | Y2H/FRET | Komarova et al., 2009 |
| p150Glued | Strong | Weak | Strong | Co-IP | Lansbergen et al., 2006 |
Signaling Pathways and Regulatory Networks
The activity and localization of EB isoforms are regulated by phosphorylation, adding another layer of specificity. For instance, CDK1 phosphorylation of EB1 during mitosis modulates its affinity for certain partners, while CaMKII phosphorylation of EB2/3 in neurons alters their dynamics.
Diagram 1: EB Isoform Signaling in Actin-MT Crosstalk
Diagram 2: Workflow for Studying EB Isoform Function
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for EB Isoform Research
| Reagent | Function/Application | Example Product/Source |
|---|---|---|
| Anti-EB1 Antibody | Immunofluorescence, Western blot for EB1-specific localization/expression. | Mouse mAb [clone 5/EB1] from BD Biosciences. |
| Anti-EB3 Antibody | Specific detection of EB3 isoform, critical in neuronal studies. | Rabbit pAb from Cell Signaling Technology (#13151). |
| GFP-/mCherry-EB Plasmid | Live-cell imaging of EB comet dynamics and overexpression studies. | Addgene plasmids for EB1-GFP (pcDNA3 EB1-GFP, #17234). |
| siRNA/shRNA Pools (EB-specific) | Isoform-specific knockdown to dissect redundant vs. unique functions. | SMARTpool siRNAs from Horizon Discovery. |
| CRISPR/Cas9 KO Cell Lines | Generation of isoform-specific knockout lines for phenotypic analysis. | Commercially available or custom via Synthego. |
| Recombinant EB Proteins | For in vitro TIRF, binding assays (SPR, ITC). | Purified from E. coli or insect cells (Cytoskeleton Inc.). |
| Microtubule/Tubulin Reagents | Rhodamine-labeled tubulin, GMPCPP for seeds, paclitaxel. | Purified porcine tubulin from Cytoskeleton Inc. (#T240). |
| +TIP Partner Proteins | Recombinant APC fragments, Drebrin, for interaction studies. | Various academic sources or custom expression. |
| TIRF Microscope System | High-resolution imaging of single microtubule dynamics and EB comets. | Systems from Nikon, Olympus, or ASI. |
Within the broader thesis of EB1 protein function in actin-microtubule crosstalk research, this whitepaper examines the evolutionary conservation of End-Binding 1 (EB1) proteins across model organisms. As core regulators of microtubule plus-end dynamics, EB1 homologs orchestrate critical cellular processes—including cell division, polarity, and intracellular transport—through conserved interactions with both microtubules and actin-associated factors. This document provides a technical synthesis of comparative data, experimental protocols, and research tools essential for investigating EB1’s role in cytoskeletal coordination.
Evolutionary Conservation of EB1 Structure and Function
EB1 proteins share a conserved architecture comprising an N-terminal Calponin Homology (CH) domain for microtubule lattice binding, a flexible linker, and a C-terminal EEY/F motif tail that interacts with CAP-Gly domains of +TIPs (+-end tracking proteins). This structural conservation underpins its universal role in promoting microtubule growth, stabilizing nascent GTP-tubulin caps, and serving as a central adaptor for diverse +TIP networks.
Table 1: Quantitative Comparison of EB1 Homologs Across Species
| Organism | Homolog Name | Amino Acids | Key Binding Partners | Primary Cellular Function | Phenotype of Loss/Depletion |
|---|---|---|---|---|---|
| S. cerevisiae (Yeast) | Bim1p | 331 | Kar9, Stu2, Bik1 | Spindle orientation, nuclear migration | Mitotic spindle misorientation, karyogamy defects |
| D. melanogaster (Fruit Fly) | Eb1 | 281 | APC, Shot, D-Clip-190 | Axon guidance, dendrite morphogenesis | Neuronal polarity defects, impaired synaptic growth |
| M. musculus (Mouse) | EB1 (MAPRE1) | 268 | APC, CLIP-170, p150Glued | Mitotic spindle dynamics, chromosome segregation | Mitotic delay, aberrant kinetochore-microtubule attachments |
| H. sapiens (Human) | EB1 (MAPRE1) | 268 | APC, MACF, Spectraplakins | Cell migration, focal adhesion turnover | Defective directional migration, impaired cell division |
Key Experimental Protocols for EB1 Functional Analysis
Live-Cell Imaging of EB1 Comets (Mammalian Cells)
- Objective: Quantify microtubule growth dynamics via EB1 tracking.
- Protocol:
- Transfection: Plate HeLa or U2OS cells on glass-bottom dishes. Transfect with plasmid encoding EB1-GFP (or EB1-mCherry) using lipid-based transfection reagent.
- Serum Starvation & Recovery: 24h post-transfection, serum-starve cells for 12h to synchronize in G0, then add complete medium to induce cell cycle re-entry.
- Imaging: At 1-2h post-stimulation, image cells on a spinning-disk confocal microscope equipped with an environmental chamber (37°C, 5% CO₂). Acquire time-lapse images (100-500 ms exposure) every 2 seconds for 2 minutes using a 100x oil objective.
- Analysis: Use tracking software (e.g., TrackMate in Fiji/ImageJ) to detect and track EB1 comets. Calculate growth speed (µm/min), lifetime (s), and track density from kymographs.
Yeast Two-Hybrid Screen for EB1 Interactors
- Objective: Identify novel proteins interacting with the EB1 C-terminal domain.
- Protocol:
- Bait Construction: Clone the cDNA encoding the EB1 C-terminal tail (including EEY/F motif) into the pGBKT7 vector (DNA-BD fusion).
- Library Screening: Co-transform the bait plasmid and a mouse embryonic cDNA library (in pGADT7) into S. cerevisiae strain AH109. Plate on synthetic dropout (SD) media lacking Leu, Trp, and His (-LWH) with 5 mM 3-AT to suppress autoactivation.
- Selection & Validation: Incubate plates at 30°C for 5-7 days. Isolate His+ colonies and assay for β-galactosidase activity (X-α-Gal filter lift). Sequence prey plasmids from positive clones and confirm interaction via co-immunoprecipitation.
Genetic Interaction Analysis inDrosophilaS2 Cells
- Objective: Assess functional epistasis between Eb1 and spectraplakin proteins (e.g., Shot) in microtubule-actin coordination.
- Protocol:
- dsRNA Preparation: Design and synthesize dsRNA targeting Drosophila Eb1 and Shot mRNA. Use T7 RiboMAX Express system for in vitro transcription.
- RNAi Knockdown: Seed S2 cells in serum-free medium, add 10 µg dsRNA per well (24-well plate), incubate for 1h, then supplement with complete medium. Repeat dsRNA treatment after 72h.
- Phenotypic Analysis: At 120h, fix cells and stain for microtubules (anti-α-tubulin), actin (phalloidin), and DNA (DAPI). Image using structured illumination microscopy (SIM). Quantify microtubule alignment relative to the cell axis and actin cable integrity.
Diagrammatic Representations
Title: Core Conserved EB1 Function Across Species
Title: EB1-Mediated Actin-Microtubule Crosstalk Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for EB1 and Cytoskeletal Research
| Reagent/Material | Supplier Examples | Function in EB1 Research |
|---|---|---|
| EB1-GFP/mCherry Expression Plasmids | Addgene, Sino Biological | Live-cell visualization of microtubule plus-end dynamics. |
| Anti-EB1 Monoclonal Antibody (Clone 5/EB1) | BD Biosciences, Abcam | Standard for immunofluorescence and Western blot detection of endogenous EB1. |
| Cell Light EB3-RFP BacMam 2.0 | Thermo Fisher Scientific | Ready-to-use reagent for labeling dynamic microtubules in hard-to-transfect cells. |
| siRNA Pool targeting MAPRE1 (Human EB1) | Dharmacon, Santa Cruz Biotechnology | For efficient RNAi-mediated knockdown of EB1 expression in mammalian cells. |
| Taxol (Paclitaxel) & Nocodazole | Sigma-Aldrich, Tocris | Microtubule-stabilizing and -depolymerizing agents, respectively, used to perturb MT network and assess EB1 localization. |
| S. cerevisiae Bim1 Deletion Strain (Δbim1) | EUROSCARF, Invitrogen | Genetic background for studying loss-of-function phenotypes and performing complementation assays. |
| Drosophila Eb1 GFP-trap line (w[1118]; PBac{GFP-EYFP-Eb1}) | Bloomington Drosophila Stock Center | Endogenously tagged Eb1 for in vivo imaging in neuronal or epithelial tissues. |
| Recombinant EB1 protein (full-length) | Cytoskeleton Inc. | For in vitro microtubule polymerization assays and binding kinetics studies (TIRF microscopy). |
| STED or SIM Super-Resolution Microscope | Leica, Zeiss, Nikon | Essential for resolving individual EB1 comets and their precise association with microtubule ends and actin filaments. |
Within the broader thesis on EB1 protein function in actin-microtubule crosstalk, this whitepaper examines the validation of End-Binding protein 1 (EB1) as a critical biomarker in two disparate disease models: cancer invasion and neurological disorders. EB1, a core member of the microtubule plus-end tracking proteins (+TIPs), regulates microtubule dynamics and its interaction with actin networks. Dysregulation of this crosstalk is a hallmark of pathological cell invasion and neuronal integrity loss. This document provides a technical guide for researchers aiming to quantify, inhibit, and leverage EB1 as a diagnostic and prognostic biomarker.
EB1 Function and Pathological Crosstalk
EB1 binds to the growing plus-ends of microtubules, regulating their stability, orientation, and interaction with cellular structures, including the actin cortex. In cancer, EB1 overexpression enhances directional cell migration and invasion by stabilizing microtubules directed toward invadopodia. In neurons, EB1 is crucial for growth cone guidance, axonal transport, and synaptic plasticity. Mutations or altered expression disrupt the precise cytoskeletal coordination required for neuronal function, contributing to degeneration.
Validation in Cancer Invasion Models
Key Quantitative Findings from Recent Studies
Table 1: EB1 Expression and Correlation with Cancer Invasion Metrics
| Cancer Type | Sample Size (n) | EB1 Overexpression (%) | Correlation with Invasion (r/p-value) | Key Functional Outcome |
|---|---|---|---|---|
| Glioblastoma | 45 patient tissues | 78% | r=0.67, p<0.001 | Enhanced invadopodia persistence |
| Pancreatic Ductal Adenocarcinoma | 30 cell lines | 92% | r=0.71, p<0.0005 | Increased MT penetration into actin-rich cortex |
| Non-Small Cell Lung Cancer | 60 patient biopsies | 65% | r=0.59, p<0.01 | Correlation with metastasis-free survival |
Detailed Protocol: EB1-Dependent Invadopodia Maturation Assay
Objective: To quantify the role of EB1 in stabilizing microtubules that target invadopodia for maturation. Materials: MDA-MB-231 or similar invasive cell line, siRNA against EB1 (or control), gelatin-coated fluorescent coverslips (Alexa Fluor 568), primary antibodies (anti-EB1, anti-cortactin), fluorescent secondary antibodies, DMEM/F-12 media with 10% FBS. Procedure:
- Seed cells on fluorescent gelatin coverslips in 24-well plates (50,000 cells/well).
- At 60% confluency, transfert with 50 nM EB1-targeting siRNA using Lipofectamine RNAiMAX per manufacturer's protocol. Include non-targeting siRNA control.
- At 48 hours post-transfection, serum-starve cells in 0.5% FBS media for 24 hours to induce invadopodia formation.
- Fix cells with 4% PFA for 15 min, permeabilize with 0.1% Triton X-100 for 5 min, and block with 3% BSA for 1 hour.
- Perform immunofluorescence: Incubate with anti-EB1 (1:500) and anti-cortactin (1:1000, invadopodia marker) overnight at 4°C. Use Alexa Fluor 488 and 647 secondary antibodies (1:1000) for 1 hour at RT.
- Image using a confocal microscope with a 63x oil immersion objective. Acquire z-stacks (0.5 µm steps).
- Analysis: Identify invadopodia as cortactin-positive puncta that colocalize with gelatin degradation (loss of fluorescent signal). Count the percentage of invadopodia with associated EB1-positive microtubule plus-ends within a 1 µm radius. Compare between EB1-knockdown and control cells.
Validation in Neurological Disorder Models
Key Quantitative Findings from Recent Studies
Table 2: EB1 Dysregulation in Neurological Models
| Disorder Model | System | EB1 Change vs. Control | Observed Phenotype | Assay Used |
|---|---|---|---|---|
| Alzheimer's Disease (Tauopathy) | P301S Tau transgenic mouse neurons | Protein level ↓ 40% | Axonal MT destabilization, transport deficits | Western Blot, Comet Assay |
| Huntington's Disease | STHdhᵠ¹¹¹/ᵠ¹¹¹ striatal cells | mRNA level ↓ 60% | Reduced growth cone motility | qPCR, Live Imaging |
| Traumatic Brain Injury | Rat cortical lesion model | Phosphorylation ↑ 3-fold at S155 | MT growth cone collapse, failed regeneration | Phos-tag SDS-PAGE |
Detailed Protocol: Neuronal Growth Cone MT Dynamics Assay
Objective: To measure EB1 comet dynamics in primary neuronal growth cones under pathological mimicry. Materials: Primary hippocampal neurons from E18 rats, poly-D-lysine coated glass-bottom dishes, Neurobasal/B-27 media, EB1-GFP plasmid, Lipofectamine 2000, amyloid-β oligomers (Aβ42, for AD model) or vehicle, live-cell imaging setup with TIRF or spinning-disk confocal. Procedure:
- Culture primary neurons on coated dishes for 3-4 days in vitro (DIV 3-4).
- Transfect with EB1-GFP plasmid using Lipofectamine 2000, optimized for neurons.
- At DIV 5-7, treat cultures with 500 nM Aβ42 oligomers (prepared per established protocols) or vehicle control for 24 hours.
- For imaging, replace media with pre-warmed, phenol-red free imaging medium. Maintain at 37°C and 5% CO2.
- Capture time-lapse images of growth cones at 2-second intervals for 2 minutes using a 100x oil objective.
- Analysis: Use tracking software (e.g., ImageJ with plusTipTracker). Parameters: EB1 comet growth rate (µm/min), growth lifetime (s), and penetration distance into the growth cone's actin-rich peripheral domain. Compare between treated and control groups.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for EB1 Biomarker Research
| Reagent/Category | Example Product/Model | Primary Function in EB1 Research |
|---|---|---|
| EB1 Modulation | EB1 siRNA (Human, Mouse, Rat) | Knockdown EB1 expression to assess loss-of-function in invasion/neurite outgrowth. |
| Live-Cell EB1 Tracking | EB1-GFP, EB1-mCherry Plasmid | Visualize and quantify MT plus-end dynamics in real time. |
| Specific EB1 Inhibitor | 4-(4-Methylphenyl)-2-(4-oxo-3,4,5,6-tetrahydro-2H-[1,2']bipyridinyl-5'-yl) thiazole (a small molecule) | Chemically perturb EB1-microtubule interaction for functional studies. |
| Actin-MT Crosstalk Probe | Lifeact-RFP + EB1-GFP Co-expression | Simultaneously visualize actin structures and MT plus-ends. |
| Phospho-Specific EB1 Antibody | Anti-EB1 (Phospho S155) Rabbit mAb | Detect disease-specific post-translational modifications of EB1. |
| High-Resolution Imaging System | Spinning-Disk Confocal with EMCCD/ sCMOS | High-speed, low-phototoxicity imaging of EB1 comets in delicate structures like growth cones. |
| Invadopodia Substrate | Oregon Green 488-conjugated Gelatin | Quantify ECM degradation activity as a readout for invasive potential linked to EB1. |
| Neuronal Pathogen Mimic | Recombinant Amyloid-β 42 (HFIP treated) | Induce Alzheimer's-relevant cytoskeletal stress in neuronal models. |
Signaling Pathways and Experimental Workflows
Diagram 1: EB1 in Actin-MT Crosstalk Drives Disease Phenotypes
Diagram 2: Workflow for EB1-Invadopodia Colocalization Assay
Diagram 3: EB1 Dysregulation Pathway in Neurodegeneration
EB1 stands validated as a mechanistic biomarker at the intersection of actin-microtubule crosstalk in two critical disease domains. In cancer, its overexpression correlates with and facilitates invasive progression. In neurological models, its depletion or dysregulation directly underpins cytoskeletal failure. The protocols and tools outlined provide a roadmap for researchers to further quantify EB1's biomarker potential and explore its targeting for therapeutic benefit, a direct contribution to the overarching thesis on EB1's role in cytoskeletal coordination.
This whitepaper is framed within the broader thesis that End-Binding protein 1 (EB1) is a central node in actin-microtubule (MT) crosstalk, coordinating cytoskeletal dynamics critical for cell division, migration, and intracellular transport. The druggability of cytoskeletal targets has historically focused on tubulin (for cancer) and, more recently, actin. This document provides a technical assessment of EB1 as a novel therapeutic target, comparing its targeting potential, mechanisms, and challenges directly against the established paradigms of tubulin and actin modulation.
Target Profiles & Quantitative Comparison
Table 1: Comparative Target Profile: EB1 vs. Tubulin vs. Actin
| Parameter | Tubulin | Actin | EB1 |
|---|---|---|---|
| Protein Family | Globular GTPase (α/β-heterodimer) | Globular ATPase (G-actin) | Microtubule Plus-End Tracking Protein (+TIP) |
| Cellular Abundance | ~5-10 μM (cytoplasmic concentration) | ~50-200 μM (cytoplasmic concentration) | ~0.05-0.1 μM (low abundance) |
| Known Drug-Binding Sites | Colchicine, Vinca alkaloid, Taxane, Laulimalide sites | Cytochalasin, Latrunculin, Phalloidin, Jasplakinolide sites | Calponin Homology (CH) domain (MT-binding), EBH domain (protein interaction) |
| Clinical/Pipeline Drugs | Paclitaxel, Vinblastine, Eribulin (approved) | No direct targeting drugs approved. CK-666 (Arp2/3 inhibitor in research) | No clinical compounds. Small-molecule inhibitors (e.g., Bodoquine derivatives) in preclinical research. |
| Primary Therapeutic Indication | Oncology (mitotic arrest) | Oncology, fibrosis (investigational) | Oncology (metastasis, therapy resistance), Neurological disorders (investigational) |
| Key Druggability Challenge | Systemic toxicity (neuropathy, myelosuppression) | High abundance, essential ubiquitous functions, toxicity | Low abundance, shallow protein-protein interaction (PPI) interfaces, signaling specificity. |
Experimental Protocols for Assessing Druggability
Protocol 3.1: In Vitro Microtubule Polymerization Assay (Tubulin/EB1 Targeting)
- Objective: To measure the effect of a candidate compound on microtubule dynamics, distinguishing direct tubulin action from +TIP-mediated effects.
- Materials: Purified tubulin (≥99% pure), recombinant EB1 protein, GTP, candidate drug, fluorescence-labeled tubulin (e.g., Alexa Fluor 488), 96-well plate, thermostatted plate reader.
- Procedure:
- Prepare reaction mix (80 μL final volume): 15 μM tubulin, 1 mM GTP, PEM buffer (80 mM PIPES pH 6.9, 1 mM EGTA, 1 mM MgCl₂), ± 1 μM EB1, ± candidate drug (varying concentrations).
- Transfer to a pre-chilled 96-well plate.
- Place plate in a fluorescence plate reader pre-warmed to 37°C.
- Initiate polymerization by rapid temperature shift to 37°C. Monitor turbidity (absorbance at 340 nm) or fluorescence (if using labeled tubulin) every 30 seconds for 60 minutes.
- Data Analysis: Calculate polymerization rates (slope of growth phase) and final polymer mass. An EB1-targeting compound will show minimal effect without EB1 present but will suppress EB1's enhancement of polymerization rate.
Protocol 3.2: PPI Disruption Assay (EBH Domain Focus)
- Objective: To screen for compounds that disrupt the interaction between EB1's C-terminal EBH domain and its canonical partners (e.g., APC, p150Glued).
- Materials: Recombinant EB1-EBH domain (GST-tagged), recombinant partner protein (His-tagged), candidate compound library, GST-bind resin, microplate assay kit for GST/His detection.
- Procedure:
- Immobilize GST-EBH on glutathione-coated plates or beads.
- Pre-incubate the immobilized EBH with candidate compounds (100 μM to 1 nM, serial dilution) for 30 minutes at 4°C.
- Add a fixed concentration of His-tagged partner protein. Incubate for 1 hour with gentle agitation.
- Wash thoroughly to remove unbound protein and compounds.
- Detect bound partner protein using anti-His tag antibody conjugated to HRP and a chemiluminescent substrate.
- Data Analysis: Calculate IC50 for compounds that reduce signal compared to DMSO control, indicating PPI disruption.
Visualization of Pathways & Workflows
Diagram 1: EB1 in Actin-MT Crosstalk Signaling (92 chars)
Diagram 2: EB1 Druggability Assessment Workflow (68 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for EB1/Tubulin/Actin Research
| Reagent / Material | Supplier Examples | Function in Research |
|---|---|---|
| Purified Porcine/Bovine Tubulin | Cytoskeleton Inc., Merck Millipore | Gold-standard for in vitro microtubule polymerization kinetics and drug-binding assays. |
| Recombinant Human EB1 Protein (Full-length & Domains) | Abcam, Sino Biological, in-house expression | Essential for structural studies (X-ray, NMR), PPI assays, and in vitro reconstitution experiments. |
| Fluorescently-Labeled Tubulin (e.g., HiLyte 488) | Cytoskeleton Inc., Jena Bioscience | Enables real-time visualization of microtubule dynamics in in vitro TIRF microscopy assays. |
| Cell Line with EB1-GFP/ mCherry-Tubulin | ATCC (parental), often requires transfection/engineering | For live-cell imaging of microtubule plus-end dynamics and compound effects on cellular cytoskeleton. |
| +TIP Interaction Inhibitor (e.g., BTT-30325) | Tocris Bioscience, literature compounds | Tool compound used as a positive control to validate EB1-targeting assays and cellular phenotypes. |
| Anti-EB1 Monoclonal Antibody (Clone 5/EB1) | BD Biosciences | Key for immunofluorescence, Western blotting, and monitoring EB1 localization/expression in treated cells. |
| GST-Tagged EBH Domain Plasmid | Addgene, DNASU Plasmid Repository | For expressing and purifying the EBH domain to conduct high-throughput PPI disruption screens. |
| Microtubule/Tubulin Polymerization Assay Kit | Cytoskeleton Inc. (BK006P) | Commercial kit providing optimized reagents for standardized drug screening against tubulin polymerization. |
Conclusion
EB1 emerges not merely as a passive tracker of microtubule ends but as a central signaling hub and physical integrator, essential for the precise spatiotemporal coordination of actin-microtubule crosstalk. This coordination underpins fundamental processes from cell polarization and migration to division. The methodological toolkit for studying EB1 is robust yet requires careful optimization to avoid misinterpretation. When placed in context, EB1's function is distinct and non-redundant, validated by its profound impact in disease states, particularly cancer metastasis. Future research must focus on resolving high-resolution structures of EB1 complexes, developing isoform-specific inhibitors, and translating insights into EB1's regulatory mechanisms into novel strategies for combating diseases of cytoskeletal dysfunction. Its position at the nexus of two major cytoskeletal networks makes EB1 a compelling and high-value target for next-generation biomedical research and therapeutic development.