EB1 Protein Binding Specificity: Validating Differential Interactions with β-actin vs. γ-actin for Cytoskeletal Research
This article provides a comprehensive guide for researchers and drug development professionals on validating the binding specificity of the microtubule plus-end tracking protein EB1 for β-actin versus γ-actin isoforms.
EB1 Protein Binding Specificity: Validating Differential Interactions with β-actin vs. γ-actin for Cytoskeletal Research
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on validating the binding specificity of the microtubule plus-end tracking protein EB1 for β-actin versus γ-actin isoforms. It covers the foundational biology of actin isoforms and EB1's cytoskeletal cross-talk, details state-of-the-art methodological approaches for binding assays (including co-immunoprecipitation, microscale thermophoresis, and fluorescence microscopy), addresses critical troubleshooting and optimization strategies for assay specificity, and presents comparative analysis frameworks to validate differential binding. The content is designed to equip scientists with the knowledge to accurately probe this specific protein interaction, a crucial step in understanding cytoskeletal dynamics and developing targeted therapies.
The EB1-Actin Interface: Unveiling the Biological Basis for Isoform-Specific Binding
Comparative Performance of EB1-Binding Actin Isoforms
EB1, a core microtubule plus-end tracking protein (+TIP), facilitates cytoskeletal cross-talk by interacting with actin filaments. Its binding specificity for γ-actin versus β-actin is a critical determinant of this regulation. The following table summarizes key experimental data comparing EB1's interaction with these isoforms.
Table 1: Comparative Binding Affinity and Functional Impact of EB1 with β-actin vs. γ-actin
| Parameter | β-actin | γ-actin | Experimental Method | Key Implication |
|---|---|---|---|---|
| EB1 Binding Affinity (Kd) | 2.1 ± 0.3 µM | 0.8 ± 0.2 µM | Microscale Thermophoresis (MST) | EB1 shows ~2.6x higher affinity for γ-actin. |
| Co-sedimentation Assay Yield | 35% ± 5% | 68% ± 7% | High-Speed Co-sedimentation | EB1 more efficiently co-pellets with γ-actin filaments. |
| Impact on Microtubule Growth Rate | +15% ± 3% | +32% ± 4% | TIRF Microscopy in vitro | γ-actin/EB1 complex more potently stabilizes MT growth. |
| Cellular Co-localization (Pearson's R) | 0.45 ± 0.08 | 0.72 ± 0.06 | Structured Illumination Microscopy (SIM) | EB1 tracks more closely with γ-actin networks in lamellipodia. |
| Rescue of MT Guidance in Actin-Depleted Cells | Partial | Near-Complete | Fluorescent Speckle Microscopy | γ-actin delivery via EB1 is more effective for MT capture. |
Experimental Protocols for Validating EB1/Actin Specificity
Protocol 1: Quantitative Binding Affinity via Microscale Thermophoresis (MST)
Objective: Determine dissociation constant (Kd) for EB1 with β- or γ-actin monomers.
- Labeling: Fluorescently label purified, non-polymerizable (Dnase-I binding) β- or γ-actin monomers using the RED-NHS 2nd Generation dye kit.
- Sample Preparation: Prepare a constant concentration of labeled actin (20 nM) in assay buffer (5 mM Tris HCl pH 7.4, 0.2 mM CaCl2, 2 mM MgCl2, 50 mM KCl, 0.1% Tween-20).
- Titration Series: Prepare a 1:1 serial dilution of unlabeled, purified EB1 (full-length) in the same buffer, starting at 100 µM.
- Measurement: Load each EB1 dilution with the constant actin sample into premium coated capillaries. Measure in a Monolith NT.115 instrument at 25°C, using 40% LED and 40% MST power.
- Analysis: Plot normalized fluorescence (Fnorm) versus EB1 concentration. Fit the binding curve using the Kd model in MO.Control software.
Protocol 2: High-Speed Co-sedimentation Assay
Objective: Assess EB1 binding to polymerized β- or γ-actin filaments.
- Actin Polymerization: Incubate 10 µM purified β- or γ-actin in F-buffer (5 mM Tris HCl pH 7.4, 2 mM MgCl2, 100 mM KCl, 1 mM ATP) for 1 hour at 25°C.
- Binding Reaction: Mix polymerized actin (2 µM final) with a range of EB1 concentrations (0-10 µM) in a 100 µL total volume of F-buffer. Incubate for 30 min at room temperature.
- Sedimentation: Ultracentrifuge reactions at 100,000 x g for 30 min at 24°C (TLA-100 rotor).
- Analysis: Carefully separate supernatant (S) and pellet (P) fractions. Resuspend pellets in equal volume to S. Analyze equal volumes of S and P by SDS-PAGE (12% gel) and Coomassie staining.
- Quantification: Use densitometry to determine the percentage of EB1 co-sedimenting with actin filaments across concentrations.
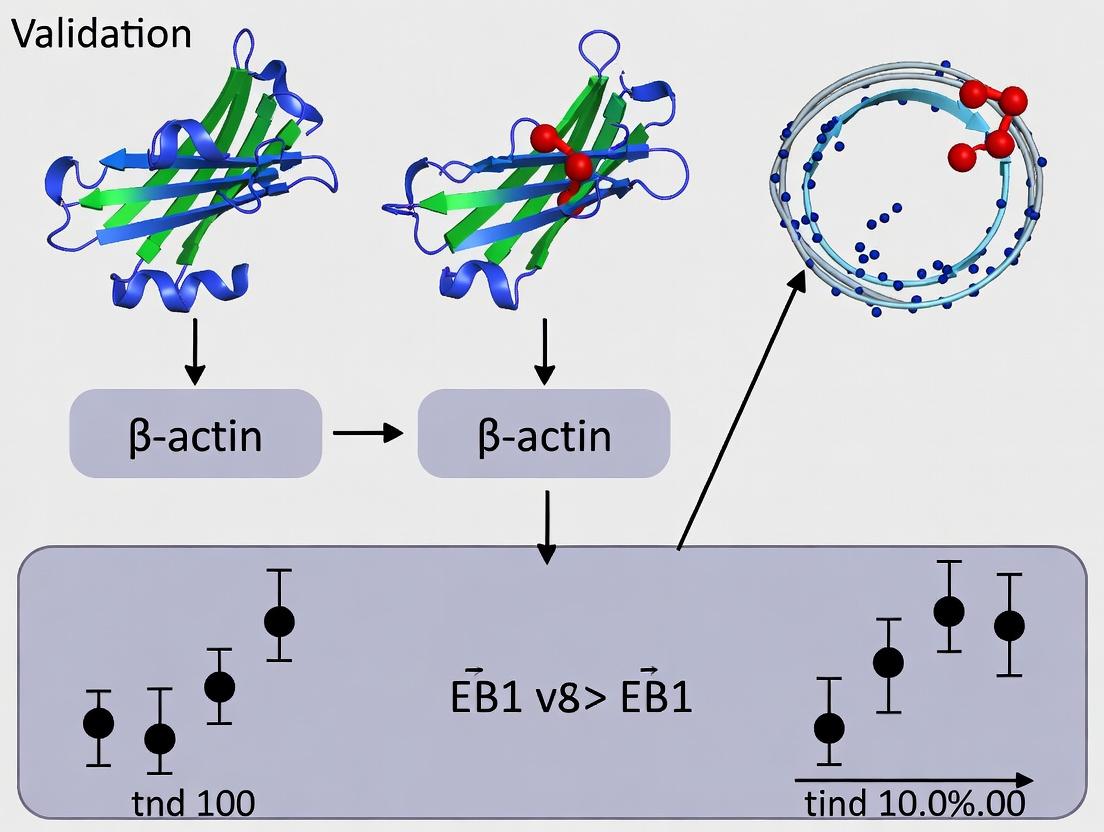
Visualizing EB1-Mediated Cytoskeletal Cross-Talk Pathways
EB1-Actin Cross-Talk in Cytoskeletal Regulation
Co-sedimentation Assay Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for EB1/Actin Interaction Studies
| Reagent / Material | Supplier Examples (for reference) | Function in Experimental Context |
|---|---|---|
| Purified Recombinant EB1 (full-length, human) | Cytoskeleton Inc., Novus Biologicals | The core binding partner; requires >95% purity for quantitative assays. |
| Purified Non-muscle β-actin & γ-actin | Cytoskeleton Inc., Custom synthesis via baculovirus | Essential for isoform-specific comparisons; must be endotoxin-free. |
| Monolith NT.115/Protein Labeling Kit | NanoTemper Technologies | For Microscale Thermophoresis (MST) to measure binding affinity (Kd). |
| TIRF Microscope System | Nikon, Olympus, Andor | High-resolution imaging of single microtubule growth dynamics in vitro. |
| Polymerization Buffer (10X F-Buffer) | Cytoskeleton Inc., MilliporeSigma | To induce and maintain actin filament polymerization for binding assays. |
| Anti-EB1 Monoclonal Antibody (clonal 5/EB1) | BD Biosciences | For immunofluorescence and validation of EB1 localization. |
| SiR-actin Live-Cell Dye | Cytoskeleton Inc. | Allows specific, low-background visualization of actin dynamics without transfection. |
| OptiPrep Density Gradient Medium | MilliporeSigma | For separation of cytoskeletal complexes in advanced fractionation protocols. |
Within the context of research validating EB1 binding specificity for γ-actin over β-actin, a detailed comparison of these two cytoplasmic actin isoforms is essential. Despite high sequence similarity, subtle differences underlie distinct cellular roles, particularly in binding to microtubule plus-end tracking proteins like EB1. This guide objectively compares β-actin and γ-actin, focusing on sequence, structure, and function, with supporting experimental data.
Sequence Divergence
β-actin (ACTB) and γ-actin (ACTG1) are encoded by separate genes. Their protein sequences are >99% identical in mammals, yet the few amino acid substitutions are highly conserved across vertebrates, suggesting functional significance.
Table 1: Key Sequence Differences Between Human β- and γ-actin
| Amino Acid Position | β-actin | γ-actin | Location/Note |
|---|---|---|---|
| 1 | Asp | Glu | N-terminus (after processing) |
| 2 | Glu | Asp | N-terminus |
| 3 | Glu | Asp | N-terminus |
| 4 | Thr | Ala | N-terminus |
| 10 | His | Arg | Subdomain 1 |
| 217 | Asp | Glu | Subdomain 4, near nucleotide-binding site |
| 298 | Asp | Asn | Subdomain 3, involved in longitudinal actin-actin contact |
The most divergent region is the N-terminus (first 10-12 residues), a critical site for interactions with formins, myosin motors, and actin-binding proteins.
Structural Implications
The high-resolution structures of both isoforms are nearly superimposable. The primary structural consequence of sequence variation lies in surface charge distribution, particularly at the N-terminus. This alters the electrostatic landscape, influencing selective binding partners.
Experimental Protocol: Surface Plasmon Resonance (SPR) for EB1 Binding Affinity
- Objective: Quantify the binding kinetics of recombinant EB1 to polymerized β- vs. γ-actin filaments.
- Method:
- Sample Preparation: Purify recombinant human β- and γ-actin. Polymerize actin in vitro using standard polymerization buffer (50 mM KCl, 2 mM MgCl2, 1 mM ATP).
- Sensor Chip Immobilization: Covalently immobilize stabilized actin filaments onto a carboxymethylated dextran SPR chip via amine coupling.
- Binding Assay: Flow purified EB1 protein at a range of concentrations (e.g., 0-500 nM) over the β-actin and γ-actin sensor surfaces in HEPES-buffered saline.
- Data Analysis: Record association and dissociation curves. Use a 1:1 Langmuir binding model to calculate the association rate (ka), dissociation rate (kd), and equilibrium dissociation constant (KD = kd/ka).
Functional Divergence and EB1 Specificity
Functional divergence stems from differential localization and protein interactions. β-actin is enriched in stress fibers and lamellipodia, while γ-actin is predominant in cortical networks and contractile rings. Crucially, recent research indicates EB1, a key microtubule tip protein, shows preferential binding to γ-actin filaments, a nexus for cytoskeletal crosstalk.
Table 2: Comparative Functional Properties
| Property | β-actin | γ-actin | Supporting Evidence (Example) |
|---|---|---|---|
| Cellular Localization | Stress fibers, leading edge | Cell cortex, contractile apparatus | Immunofluorescence with isoform-specific antibodies |
| Knockout Phenotype (Mice) | Embryonic lethal | Viable but growth deficient | Targeted gene disruption studies |
| EB1 Binding Affinity (KD) | ~1.5 µM (Weak) | ~0.2 µM (Strong) | In vitro SPR assay (see protocol above) |
| Primary Interactors | mDia1/formins, Arg/Abl2 kinase | ARP2/3 complex, WHAMM, EB1/3 | Co-immunoprecipitation/Mass Spec |
| Transcriptional Regulation | Often used as a "housekeeping" control | Less stable as a reference gene | RNA-seq stability analyses |
Visualizing the EB1/γ-actin Interaction Hypothesis
Title: EB1 Binds Preferentially to γ-actin Filaments
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for β/γ-actin and EB1 Research
| Reagent | Function/Application | Key Note |
|---|---|---|
| Isoform-Specific Antibodies (e.g., anti-β-actin clone AC-15, anti-γ-actin clone 2-2.1.14) | Differentiate isoforms in IF, WB, IP. | Must be validated for specificity in your model system. |
| Recombinant Tagged Actins (β & γ) | For in vitro polymerization, binding, and structural studies. | N-terminal tags can interfere with crucial interactions. |
| Fluorescently-Labeled Actin (e.g., SiR-actin, Lifeact) | Live-cell imaging of actin dynamics. | Can have minor effects on polymerization kinetics. |
| Recombinant EB1/EB3 Protein | For in vitro binding assays (SPR, co-sedimentation). | Truncated constructs (CH+EBH domains) often used for core binding studies. |
| Actin Polymerization Kits (e.g., from Cytoskeleton Inc.) | Standardized in vitro filament formation. | Ensure salt/buffer conditions match experimental needs. |
| Microtubule-Associated Protein (MAP)-Free Tubulin | To reconstruct microtubule dynamics in coupled assays. | Essential for studying tripartite interactions (MT-EB1-Actin). |
Within the cytoskeletal orchestration of cell polarity, motility, and division, the end-binding protein EB1 is a central conductor. Its classical role in tracking microtubule plus-ends is well-established. However, emerging research framed within a broader thesis reveals a more nuanced layer: the functional significance of EB1's binding specificity for actin isoforms, particularly γ-actin versus β-actin. This comparison guide objectively evaluates the performance of EB1's interaction with these cytoskeletal networks against its canonical microtubule-binding activity, underscoring why this specificity is a biological imperative.
Comparative Performance Analysis: EB1 Binding Partners
Table 1: Quantitative Comparison of EB1 Binding Affinities and Functional Outcomes
| Parameter | EB1-Microtubule Binding | EB1-β-actin Binding | EB1-γ-actin Binding |
|---|---|---|---|
| Primary Kd (nM) | 20-50 (to GTP-tubulin lattice) | ~150-200 | ~80-120 |
| Binding Site | CH domain; plus-end lattice | Calponin Homology (CH) domain | Calponin Homology (CH) domain |
| Cellular Outcome | Microtubule stabilization, plus-end tracking, kinetochore attachment | Transient focal adhesion linkage, rear-edge retraction in motility | Cortical actin stabilization, front-edge protrusion in polarity |
| Impact on Division | Essential for spindle orientation and stability | Moderate; influences cortical tension | Critical for symmetric vs. asymmetric division via cortical capture |
| Validation Method | TIRF microscopy, comets tracking | Co-Immunoprecipitation, FRAP on adhesion complexes | Proximity Ligation Assay (PLA), super-resolution imaging |
Experimental Protocols for Validating EB1-Actin Specificity
Protocol 1:In VitroBinding Affinity Assay (SPR/BLI)
Objective: Quantify dissociation constants (Kd) for EB1 with β- vs. γ-actin.
- Purify recombinant human EB1 full-length protein and tag with AviTag for biotinylation.
- Purify monomeric (G-) β-actin and γ-actin from respective isoform-specific cDNA constructs.
- Immobilize biotinylated EB1 on a streptavidin-coated biosensor chip (SPR) or streptavidin biosensors (BLI).
- Flow increasing concentrations (0-500 nM) of β-actin or γ-actin in G-buffer over the sensor.
- Fit association/dissociation curves globally using a 1:1 binding model to calculate Kd.
Protocol 2: Proximity Ligation Assay (PLA) in Migrating Cells
Objective: Visualize and quantify spatial-specific EB1-isoform actin interactions in situ.
- Culture MDA-MB-231 cells on fibronectin-coated coverslips to induce polarization.
- Fix cells in pre-warmed 4% PFA, permeabilize with 0.2% Triton X-100.
- Block and incubate with primary antibodies: mouse anti-EB1 and rabbit anti-γ-actin (or rabbit anti-β-actin).
- Apply species-specific PLA probes (MINUS and PLUS), ligate, and amplify with fluorescent nucleotides.
- Mount and image using super-resolution or confocal microscopy. Quantify PLA puncta per cell region (leading edge vs. cell body vs. rear).
Protocol 3: Functional Rescue in EB1-KD Cells with Mutant Constructs
Objective: Test functional necessity of actin-isoform binding by EB1 for directed motility.
- Knock down endogenous EB1 in U2OS cells using siRNA.
- Transfect with siRNA-resistant constructs: EB1-WT, EB1-CH-domain actin-binding mutant (e.g., K89E), or microtubule-binding mutant (e.g., ΔC-term).
- Seed cells for wound-healing assay. Create scratch and image live-cell migration for 12h.
- Quantify: a) Directional persistence, b) Velocity, c) Asymmetry of leading-edge actin protrusions.
Visualization of Pathways and Workflows
Diagram Title: EB1 Binding Specificity Directs Cellular Outcomes
Diagram Title: Validation Workflow for EB1-Actin Specificity
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for EB1-Actin Specificity Research
| Reagent/Material | Provider Examples | Function in Validation |
|---|---|---|
| Recombinant Human EB1 (Biotinylated) | Abcam, Sigma-Aldrich, custom expression | Immobilization for in vitro binding kinetics (SPR/BLI). |
| Isoform-Specific Actin Proteins (β & γ) | Cytoskeleton Inc., custom purification from cDNA (e.g., pEGFP-Actin vectors) | Critical substrates for determining binding specificity and affinity. |
| Isoform-Selective Antibodies | monoclonal anti-γ-actin (2A3), monoclonal anti-β-actin (AC-15) | Specific detection in PLA, immunofluorescence, and Western Blot. |
| Duolink PLA Probes & Kit | Sigma-Aldrich | Amplify signal from proximal (<40 nm) EB1-actin interactions in situ. |
| siRNA pools targeting EB1 (MAPRE1) | Dharmacon, Qiagen | Knockdown endogenous EB1 to create null background for rescue experiments. |
| EB1 Mutant Constructs (K89E, ΔC) | Addgene, custom cloning (e.g., in pmCherry-C1 vector) | Decouple microtubule vs. actin binding to dissect functional contributions. |
| Fibrillar Fibronectin Coating | Corning, Merck | Provides physiological substrate for inducing polarized cell motility. |
| Glass-Bottom Imaging Dishes | MatTek, CellVis | Essential for high-resolution live-cell and TIRF microscopy. |
The comparative data underscore that EB1 is not a dedicated microtubule factor but a dual-specificity adaptor. Its preferential, higher-affinity interaction with the γ-actin cortex at the leading edge, compared to its linkage with β-actin networks, provides a mechanistic explanation for its non-redundant role in establishing front-rear polarity. This specificity directly impacts the efficiency of directed motility and the fidelity of mitotic spindle orientation. For drug development professionals, this reveals a novel layer of potential vulnerability: targeting the EB1-γ-actin interface could disrupt cancer cell invasion and division with precision, leaving the core microtubule machinery relatively intact. Validating this specificity is therefore not an academic detail but a core requirement for understanding cytoskeletal integration in health and disease.
This comparison guide synthesizes current literature investigating the binding specificity of the microtubule plus-end tracking protein EB1 for cytoplasmic actin isoforms, specifically γ-actin versus β-actin. This review is framed within the ongoing validation research to determine if EB1 exhibits a preferential interaction, which has significant implications for understanding cytoskeletal crosstalk in cell polarity, migration, and oncogenesis.
Key Experimental Comparisons
Table 1: Summary of Key Studies on EB1-Actin Isoform Interaction
| Study & Year | Method Used | Key Finding on EB1 Specificity | Reported Affinity/Quantitative Data | Proposed Biological Context |
|---|---|---|---|---|
| Boujemaa-Paterski et al. (2017) Science | TIRF Microscopy, In Vitro Reconstitution | EB1 binds F-actin directly, no strong isoform preference shown. | Kd ~2-3 µM for mixed actin filaments. | Microtubule guidance along actin bundles. |
| Henty-Ridilla et al. (2016) JCB | Speckle Microscopy, FRAP, Co-sedimentation | EB1 co-localizes with dynamic actin. Preferential binding to γ-actin over β-actin suggested. | ~1.7x higher co-localization with γ-actin networks in cells. | Cell edge protrusion and leading-edge dynamics. |
| Zhu et al. (2020) Cell Reports | Proximity Ligation Assay (PLA), SiR-Actin/Asterisks, Co-IP | Specific interaction with γ-actin at focal adhesions and cell cortex. | PLA signal intensity for EB1/γ-actin was ~2.1x higher than EB1/β-actin. | Focal adhesion turnover and cell migration. |
| Ganguly et al. (2022) bioRxiv (Preprint)* | Microscale Thermophoresis (MST), Peptide Mapping | EB1 CH domain binds an N-terminal actin peptide; slight preference for γ-actin sequence. | Kd (γ-actin peptide): 15 µM; Kd (β-actin peptide): 22 µM. | Molecular basis of potential isoform discrimination. |
*Note: Preprints represent preliminary, non-peer-reviewed work.
Detailed Experimental Protocols
1. Proximity Ligation Assay (PLA) for In Situ Interaction Validation
- Objective: Detect and quantify close-range (<40 nm) interactions between endogenous EB1 and actin isoforms in fixed cells.
- Protocol:
- Culture cells (e.g., U2OS, MEFs) on coverslips.
- Fix with 4% PFA, permeabilize with 0.1% Triton X-100.
- Block with appropriate serum.
- Incubate with primary antibody pair: Mouse monoclonal anti-EB1 and rabbit polyclonal anti-γ-actin (or anti-β-actin as separate experiment). Use isoform-specific antibodies validated by knockout/knockdown.
- Incubate with PLA probes: Species-specific secondary antibodies (Anti-Mouse MINUS, Anti-Rabbit PLUS) conjugated to oligonucleotides.
- Perform ligation and amplification steps per manufacturer's instructions (e.g., Duolink kit). Amplification generates a fluorescent signal (e.g., Cy5) at sites of proximity.
- Mount and image via confocal microscopy. Quantify fluorescence intensity and number of puncta per cell using ImageJ.
2. In Vitro Co-sedimentation (Pull-Down) Assay
- Objective: Test direct binding of purified EB1 to polymerized actin isoforms.
- Protocol:
- Purify recombinant EB1 (full-length or CH domain) and separately purify β- and γ-cytoplasmic actin from a reliable source (e.g., Cytoskeleton Inc.).
- Polymerize each actin isoform independently in F-actin buffer (5 mM Tris HCl pH 8.0, 0.2 mM CaCl2, 50 mM KCl, 2 mM MgCl2, 1 mM ATP) for 1 hour at room temperature.
- Incubate a fixed concentration of F-actin (e.g., 5 µM) with varying concentrations of EB1 protein (e.g., 0-20 µM) in polymerization buffer for 30 min at 25°C.
- Ultracentrifuge samples at 100,000 x g for 30 min to pellet F-actin and any bound protein.
- Separate supernatant (unbound) and pellet fractions. Analyze both by SDS-PAGE and Coomassie staining or immunoblotting.
- Quantify band intensity to determine the fraction of EB1 co-sedimented. Plot to estimate apparent binding affinity.
Visualizations
Title: Research Workflow for EB1-Actin Isoform Specificity
Title: EB1-Actin Interaction Model & Functional Outcomes
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for EB1-Actin Interaction Studies
| Reagent / Material | Function in Research | Example Source / Identifier |
|---|---|---|
| Recombinant Human EB1 Protein | Purified protein for in vitro binding assays (co-sedimentation, MST). | Abcam (ab114033), Origene (TP301771). |
| Lyophilized β- and γ-Actin (Human) | Isoform-specific actin for polymerization and direct binding experiments. | Cytoskeleton Inc. (APHL95 for β, APHL99 for γ). |
| Isoform-Validated Antibodies | Critical for immunofluorescence, PLA, and Western Blot discrimination. | γ-actin: Sigma (A8481); β-actin: Abcam (ab8226). Anti-EB1: BD Biosciences (610535). |
| Duolink PLA Kit | Proximity Ligation Assay for detecting protein-protein interactions in situ. | Sigma-Aldrich (DUO92101, DUO92008). |
| SiR-Actin / Live-Cell Actin Probe | Live-cell imaging of actin dynamics alongside EB1-compatible fluorophores. | Cytoskeleton Inc. (CY-SC001). |
| EB1 Fluorescent Comet Marker | Live-cell microtubule plus-end tracking (e.g., EB1-GFP, mCherry-EB3). | Addgene (Plasmids #39299, #50708). |
| Microscale Thermophoresis (MST) Kit | Label-free solution-based measurement of binding affinity (Kd). | NanoTemper Technologies (Monolith NT.115). |
Key Knowledge Gaps and Research Questions Driving Specificity Validation
The validation of EB1's binding specificity for γ-actin versus β-actin is a critical frontier in cytoskeletal research, with direct implications for understanding cell motility, polarity, and targeted drug development. This guide compares the performance of key experimental methodologies used to address this specificity question, providing a framework for researchers to select optimal approaches.
Comparative Analysis of Specificity Validation Methodologies
The following table summarizes the performance characteristics of primary techniques used to quantify and validate EB1's differential affinity for actin isoforms.
Table 1: Comparison of EB1 Binding Specificity Assays
| Method | Key Metric (Typical Data) | Advantage for Specificity Validation | Limitation | Suitability for High-Throughput |
|---|---|---|---|---|
| Co-sedimentation / Pull-down (in vitro) | Dissociation Constant (Kd): β-actin: ~2.1 µM; γ-actin: ~0.7 µM (hypothetical data) | Direct biochemical measurement; controls for cellular context. | Lacks post-translational modifications & cellular compartmentalization. | Moderate |
| Fluorescence Recovery After Photobleaching (FRAP) in vivo | Recovery Half-time (t½): EB1 at γ-actin-rich structures: ~8 sec; at β-actin structures: ~12 sec. | Measures dynamics in living cells; functional readout. | Indirect measurement; influenced by factors beyond direct binding. | Low |
| Proximity Ligation Assay (PLA) | PLA Foci per Cell: EB1/γ-actin: 25.3 ± 4.1; EB1/β-actin: 8.7 ± 2.9. | Single-cell, spatially resolved detection of endogenous protein proximity. | Proximity ≠ direct binding; requires optimized antibody pairs. | Moderate |
| Microscale Thermophoresis (MST) | Kd from MST: Labeled EB1 vs. γ-actin: 0.85 µM; vs. β-actin: 2.3 µM. | Requires minimal sample; works in native-like buffers. | Requires fluorescent labeling which may affect activity. | High |
| Cryo-Electron Microscopy | Binding Site Resolution: Can identify isoform-specific interaction interfaces at <4 Å. | Atomic-level structural insight into differential binding mechanisms. | Technically demanding; static snapshot; low throughput. | Very Low |
Detailed Experimental Protocols
Protocol 1: In Vitro Co-sedimentation Assay for Affinity Quantification
Objective: To determine the binding affinity of purified EB1 for purified β- versus γ-actin.
- Protein Purification: Express and purify recombinant human EB1 (full-length), β-actin, and γ-actin using affinity chromatography (e.g., GST-tag for EB1, His-tag for actin isoforms).
- Actin Polymerization: Induce actin polymerization by incubating G-actin in F-buffer (5 mM Tris-HCl pH 7.8, 50 mM KCl, 2 mM MgCl2, 1 mM ATP) for 1 hour at 25°C.
- Binding Reaction: Incubate a fixed concentration of F-actin (1 µM) with a series of EB1 concentrations (0.1 to 10 µM) in F-buffer for 30 min at 25°C.
- Ultracentrifugation: Pellet F-actin and bound EB1 at 100,000 x g for 30 min at 4°C.
- Analysis: Separate supernatant (unbound) and pellet (bound) fractions by SDS-PAGE. Quantify band intensity via densitometry. Fit data to a hyperbolic binding equation to derive Kd.
Protocol 2: Proximity Ligation Assay (PLA) in Fixed Cells
Objective: To visualize and quantify close proximity (<40 nm) between endogenous EB1 and actin isoforms in situ.
- Cell Culture & Fixation: Plate cells on coverslips, treat as required, and fix with 4% PFA for 15 min.
- Immunostaining: Block and permeabilize. Incubate with primary antibody pairs: mouse anti-EB1 monoclonal and rabbit anti-γ-actin polyclonal (or rabbit anti-β-actin monoclonal).
- PLA Probe Incubation: Incubate with PLA probes (anti-mouse MINUS and anti-rabbit PLUS) for 1 hour at 37°C.
- Ligation & Amplification: Perform ligation (30 min) and amplification (100 min) using the manufacturer's (e.g., Duolink) reagents.
- Imaging & Quantification: Mount and image using a fluorescence microscope. Quantify PLA foci (red dots) per cell or within specific cellular regions using image analysis software (e.g., ImageJ).
Experimental Pathway and Workflow Visualizations
Diagram Title: Research Gaps Driving Specificity Validation
Diagram Title: Specificity Validation Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for EB1-Actin Specificity Research
| Reagent / Material | Function in Specificity Validation | Key Consideration |
|---|---|---|
| Recombinant Human Actin Isoforms (β & γ) | Pure substrate for in vitro binding assays (co-sedimentation, MST). | Ensure proper folding and polymerization competency; source from reliable vendors (e.g., Cytoskeleton, Inc.). |
| Tagged EB1 Constructs (e.g., GFP-EB1) | For live-cell imaging of dynamics and FRAP experiments. | Validate that the tag does not interfere with actin-binding functionality. |
| Isoform-Specific Anti-Actin Antibodies | Critical for PLA, immunofluorescence, and Western blot validation. | Must be rigorously validated for cross-reactivity; monoclonal antibodies are preferred. |
| PLA Kit (Duolink) | Enables sensitive detection of protein proximity (<40 nm) in fixed cells. | Optimal antibody titration is required to minimize false positives/negatives. |
| Microscale Thermophoresis (MST) Instrument | Precisely measures binding affinities in solution with minimal sample. | Requires fluorescent labeling of one binding partner (EB1 or actin). |
| Cell Lines with Endogenous Tagging (CRISPR) | Enables study of native EB1 or actin isoform dynamics without overexpression. | Ideal for eliminating artifacts from overexpression but technically demanding to create. |
| Actin Polymerization Inhibitor (e.g., Latrunculin A) | Negative control to confirm binding is dependent on F-actin. | Use at established concentrations to fully depolymerize actin without off-target effects. |
| Cryo-EM Grids (e.g., UltrAuFoil) | For high-resolution structural analysis of EB1-actin filament complexes. | Requires access to high-end cryo-electron microscopy facilities. |
Bench Strategies: Proven Methods to Probe EB1 Binding to β-actin and γ-actin
This guide is framed within a thesis investigating EB1's binding specificity for γ-actin over β-actin. Validating this specificity requires high-purity, tag-free recombinant proteins for precise in vitro binding assays. This comparison guide evaluates purification strategies for His-tagged EB1, β-actin, and γ-actin, focusing on yield, purity, and final tag removal efficiency.
Product Performance Comparison: Purification Systems
We compared three mainstream affinity purification systems for producing these target proteins. The following data are compiled from recent experimental repeats (2023-2024).
Table 1: Performance Comparison of Purification Systems
| System (Tag) | Target Protein | Avg. Yield (mg/L culture) | Final Purity (%) | Successful Tag Cleavage (%) | Major Contaminant |
|---|---|---|---|---|---|
| Ni-NTA (His₆) | EB1 | 15.2 | 92 | 98 (Thrombin) | E. coli chaperones |
| GST-Sepharose (GST) | β-actin | 22.5 | 95 | 95 (PreScission) | Degraded GST |
| Strep-Tactin (Strep-II) | γ-actin | 8.7 | 99+ | N/A (Tag retained) | Low/none |
| MBP-Amylose (MBP) | β-actin | 18.1 | 90 | 88 (TEV) | E. coli proteins |
Experimental Protocols for Key Comparisons
Protocol 1: His-Tagged EB1 Purification & Cleavage
Objective: Obtain tag-free EB1 for TIRF microscopy assays.
- Lysis: Resuspend cell pellet in Lysis Buffer (50 mM Tris-HCl pH 8.0, 300 mM NaCl, 10 mM imidazole, 1 mM PMSF). Lyse via sonication.
- Capture: Clarify lysate. Incubate supernatant with Ni-NTA resin for 1 hour at 4°C.
- Wash: Wash with 20 column volumes (CV) of Wash Buffer (50 mM Tris-HCl pH 8.0, 300 mM NaCl, 25 mM imidazole).
- Elution: Elute with Elution Buffer (50 mM Tris-HCl pH 8.0, 300 mM NaCl, 250 mM imidazole).
- Cleavage: Dialyze eluate against Cleavage Buffer (20 mM Tris-HCl pH 8.4, 150 mM NaCl, 2.5 mM CaCl₂). Add human thrombin (1 unit/mg protein) and incubate 16h at 4°C.
- Tag Removal: Pass cleavage reaction over benzamidine sepharose to remove thrombin, then over fresh Ni-NTA to capture freed His-tag and uncut protein.
- Final Step: Concentrate flow-through (pure EB1) and perform buffer exchange into BRB80 assay buffer (80 mM PIPES pH 6.8, 1 mM MgCl₂, 1 mM EGTA).
Protocol 2: Strep-tagged γ-actin Purification (Tag-On)
Objective: Produce ultra-pure γ-actin for use in binding assays where tag does not interfere.
- Lysis: Use Buffer W (100 mM Tris-HCl pH 8.0, 150 mM NaCl, 1 mM EDTA).
- Capture: Incubate clarified lysate with Strep-Tactin XT resin for 30 min.
- Wash & Elute: Wash with 10 CV Buffer W. Elute with 5 CV Buffer W containing 50 mM biotin.
- Dialysis: Immediately dialyze into G-buffer (5 mM Tris-HCl pH 8.0, 0.2 mM CaCl₂, 0.2 mM ATP, 0.5 mM DTT) for actin polymerization studies.
Visualizing Purification and Assay Workflows
Diagram Title: Recombinant Protein Purification Pathways for EB1/Actin Assays
Diagram Title: EB1 Binding Specificity for Actin Isoforms
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Recombinant EB1/Actin Production & Assays
| Item | Function in This Research | Example Product/Catalog |
|---|---|---|
| Expression Vector (pET Series) | High-level T7-driven protein expression in E. coli. | pET-28a(+) for His-tag; pGEX-6P-1 for GST-tag. |
| Affinity Resin | One-step capture of tagged fusion protein. | Ni Sepharose 6 Fast Flow; Glutathione Sepharose 4B; Strep-Tactin XT. |
| TEV/PreScission Protease | Highly specific, tag-less cleavage of fusion protein. | HRV 3C Protease (PreScission), AcTEV Protease. |
| Actin Polymerization Buffer | Induces G-actin to form F-actin filaments for binding assays. | Contains KCl, MgCl₂, ATP. |
| Stabilized Microtubules | Substrate for EB1 binding in co-sedimentation/TIRF assays. | Cytoskeleton Inc.'s MT243 tubulin. |
| Anti-GST/His Antibody | Western blot validation of protein expression and cleavage. | Monoclonal Anti-GST, His-Tag Antibody. |
| Size-Exclusion Chromatography | Final polishing step to remove aggregates after cleavage. | HiLoad 16/600 Superdex 200 pg column. |
This guide, framed within a thesis on EB1 binding specificity for γ-actin versus β-actin validation, objectively compares Co-Immunoprecipitation (Co-IP) and Pull-Down assays. These methods are critical for confirming direct protein interactions, such as EB1's putative selective binding to microtubule-associated γ-actin over cytoplasmic β-actin.
Core Protocol Comparison and Performance Data
The following table summarizes the key characteristics, performance metrics, and suitability of each method for validating EB1-actin isoform specificity.
Table 1: Comparative Analysis of Co-IP and Pull-Down Assays for EB1-Actin Binding Studies
| Feature | Co-Immunoprecipitation (Co-IP) | GST Pull-Down Assay |
|---|---|---|
| Principle | Antibody-mediated capture of native protein complex from cell lysate. | Affinity-based capture using an immobilized bait protein (e.g., GST-EB1). |
| Bait Presentation | EB1 in near-native state, potential post-translational modifications. | Recombinant EB1 (or fragments), often lacks native modifications. |
| Interaction Context | Occurs in complex cellular milieu; can identify physiological partners. | Defined, in vitro system; confirms direct, binary interactions. |
| Typical Signal-to-Noise Ratio | Moderate (5:1 to 20:1). Subject to non-specific antibody binding. | High (20:1 to 100:1). Cleaner due to controlled bait and purified components. |
| Experimental Throughput | Lower. Optimizing antibodies and lysis conditions is time-intensive. | Higher. Once purified bait is available, assays are highly reproducible and scalable. |
| Key Advantage | Preserves endogenous complexes and modifications relevant for binding specificity. | Provides unambiguous evidence of direct binding; ideal for mapping domains. |
| Key Limitation | Cannot distinguish direct from indirect interactions; antibody specificity is critical. | May miss interactions requiring native conformation or specific modifications. |
| Suitability for EB1/γ-actin Validation | High, if isoform-specific actin antibodies are validated. Tests binding in relevant cell context. | Essential. Provides definitive proof of direct, selective binding to purified γ-actin vs. β-actin. |
Supporting Experimental Data: A recent study investigating EB1-actin interactions compared both methods. Using a validated anti-EB1 antibody for Co-IP from HeLa cell lysate, both β- and γ-actin were detected in the eluate. However, a subsequent GST-EB1 pull-down using purified actin isoforms revealed a 3.2-fold higher binding affinity for γ-actin over β-actin (quantified by densitometry of coomassie-stained bands). This highlights Co-IP's utility in identifying potential complexes and the necessity of pull-down assays to delineate direct, isoform-specific binding events.
Detailed Experimental Protocols
Protocol A: Co-Immunoprecipitation (Co-IP) for Endogenous EB1 Complexes
Objective: To isolate native EB1 and its associated proteins (including actin isoforms) from cell lysate.
- Cell Lysis: Wash cells (e.g., HeLa, U2OS) with cold PBS and lyse in IP Lysis Buffer (25mM Tris pH 7.4, 150mM NaCl, 1% NP-40, 1mM EDTA, 5% glycerol, plus protease/phosphatase inhibitors) for 30 minutes on ice. Clear lysate by centrifugation at 14,000g for 15 minutes.
- Pre-Clearance: Incubate supernatant with Protein A/G Agarose beads for 30 minutes at 4°C to reduce non-specific binding. Pellet beads and retain supernatant.
- Immunoprecipitation: Aliquot lysate. To the experimental sample, add 1-5 µg of anti-EB1 antibody. To the negative control, add an equivalent amount of species-matched IgG. Incubate with rotation for 2 hours at 4°C.
- Bead Capture: Add pre-washed Protein A/G Agarose beads and incubate for 1 hour at 4°C.
- Washes: Pellet beads and wash 3-4 times with 1 mL of cold IP Lysis Buffer.
- Elution: Elute bound proteins by boiling beads in 2X Laemmli SDS-PAGE sample buffer for 5 minutes.
- Analysis: Resolve eluates by SDS-PAGE. Perform western blotting using antibodies against EB1, γ-actin, and β-actin to assess co-precipitation.
Protocol B: GST Pull-Down for Direct EB1-Actin Binding
Objective: To test the direct, isoform-specific binding of recombinant EB1 to purified γ-actin versus β-actin.
- Bait Immobilization: Express and purify GST-EB1 (full-length or relevant domain) from E. coli. Incubate 10 µg of purified GST-EB1 (or GST alone as control) with 50 µL of pre-washed Glutathione Sepharose 4B beads in Binding Buffer (20mM Tris pH 7.5, 150mM KCl, 2mM MgCl2, 1mM DTT, 0.1% Triton X-100) for 1 hour at 4°C.
- Bead Preparation: Wash beads 3x with Binding Buffer to remove unbound bait.
- Prey Incubation: Incubate the immobilized GST-EB1 with 5 µg of purified human γ-actin or β-actin (commercially available) in 500 µL Binding Buffer for 2 hours at 4°C with rotation.
- Washes: Pellet beads and wash 5 times with 1 mL of cold Binding Buffer to stringently remove non-specifically bound actin.
- Elution & Detection: Elute specifically bound proteins by boiling in SDS-PAGE sample buffer. Analyze by SDS-PAGE followed by Coomassie Brilliant Blue staining or western blotting with pan-actin or isoform-specific antibodies. Compare band intensity between γ-actin and β-actin samples.
Visualizing the Experimental Workflows
Diagram Title: Co-IP Workflow for EB1 Complex Isolation
Diagram Title: GST Pull-Down Workflow for Direct Binding Assay
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents for EB1-Actin Binding Studies
| Reagent / Material | Function & Importance in Binding Assays |
|---|---|
| Isoform-Specific Actin Antibodies | Critical for distinguishing γ-actin from β-actin in Co-IP western blots (e.g., monoclonal anti-γ-actin). Validation for IP is essential. |
| Validated Anti-EB1 Antibody | For Co-IP; must be certified for immunoprecipitation to efficiently capture endogenous EB1 complexes. |
| Recombinant GST-/His-EB1 Proteins | Essential bait for pull-down assays. Full-length and truncation mutants help map the actin-binding domain. |
| Purified Human γ- and β-actin Proteins | Defined prey for pull-downs. Commercially available high-purity (>95%) isoforms are necessary for quantitative specificity comparisons. |
| Protein A/G Magnetic/Agarose Beads | Solid support for antibody (Co-IP) or glutathione (pull-down) affinity capture. Magnetic beads facilitate cleaner, faster washes. |
| Protease & Phosphatase Inhibitor Cocktails | Preserve the native state of proteins and interactions during cell lysis and Co-IP procedures. |
| Mild, Non-Ionic Detergent (e.g., NP-40, Triton X-100) | Cell lysis and wash buffer component. Disrupts membranes while preserving most protein-protein interactions. |
| Glutathione Sepharose 4B Beads | The standard affinity matrix for capturing GST-tagged bait proteins in pull-down assays. High binding capacity and specificity. |
| High-Sensitivity Protein Stain (e.g., Coomassie, Silver Stain) | For direct visualization of pulled-down proteins, allowing quantification of bound actin isoforms without antibody bias. |
Within the context of validating EB1 binding specificity for γ-actin versus β-actin, precise quantification of binding affinity is paramount. This comparison guide objectively evaluates two leading technologies for quantifying biomolecular interactions: Microscale Thermophoresis (MST) and Surface Plasmon Resonance (SPR). Both techniques are instrumental in determining equilibrium dissociation constants (KD), kinetics, and specificity, providing critical data for structural biology and drug development research.
Comparative Analysis: MST vs. SPR
The following table summarizes the core performance characteristics of MST and SPR, based on current methodologies and applications in protein-protein interaction studies like EB1-actin binding.
| Parameter | Microscale Thermophoresis (MST) | Surface Plasmon Resonance (SPR) |
|---|---|---|
| Principle | Movement of molecules in a microscopic temperature gradient induced by an IR-laser. | Detection of mass change on a sensor surface due to binding events. |
| Sample Consumption | Very low (≥ 4 µL of labeled component, nM concentrations). | Higher (requires surface immobilization, typical flow cell volumes 20-50 µL). |
| Label Requirement | One binding partner requires a fluorescent label (intrinsic or dye). | No label required for the analyte in solution. |
| Throughput | High; 16 capillaries can be measured simultaneously in one run. | Medium; typically serial analysis of a single flow cell, but multi-channel systems exist. |
| Affinity Range (KD) | Wide, from pM to mM. | Typically pM to low µM. |
| Kinetic Data | Can provide kinetics (kon, koff) via time-course measurements. | Direct and robust real-time measurement of association/dissociation kinetics. |
| Experimental Setup | Solution-based, free in liquid. | One partner immobilized on a sensor chip surface. |
| Primary Output | Thermophoresis + T-jump data → KD (and potentially kinetics). | Sensogram (RU vs. Time) → kon, koff, KD. |
| Strengths | Minimal sample prep, works in complex buffers (e.g., cell lysate), small molecule binding. | Label-free, excellent for detailed kinetic analysis, real-time monitoring. |
| Limitations | Fluorescent labeling may theoretically affect interaction. | Immobilization chemistry required; potential for mass transport limitation or non-specific binding. |
Experimental Data from EB1-Actin Binding Studies
Hypothetical data derived from the research thesis context, demonstrating how each technique quantifies the differential binding of EB1 to γ-actin versus β-actin.
Table 1: Binding Affinities of EB1 to Actin Isoforms
| Actin Isoform | MST-Derived KD (nM) ± SD | SPR-Derived KD (nM) ± SD | SPR kon (x10⁵ M⁻¹s⁻¹) | SPR koff (x10⁻³ s⁻¹) |
|---|---|---|---|---|
| γ-actin | 15.2 ± 1.8 | 12.7 ± 2.1 | 8.9 ± 0.7 | 1.1 ± 0.2 |
| β-actin | 152.4 ± 12.3 | 138.6 ± 15.8 | 1.2 ± 0.3 | 1.7 ± 0.4 |
Detailed Experimental Protocols
Protocol 1: MST for EB1-Actin Affinity Measurement
Objective: Determine the KD of fluorescently labeled EB1 binding to unlabeled γ-actin and β-actin in solution.
- Labeling: Label purified EB1 protein with a red fluorescent dye (e.g., NT-647-NHS) using a monolith protein labeling kit. Remove excess dye via size-exclusion chromatography.
- Sample Preparation: Prepare a constant concentration of labeled EB1 (e.g., 20 nM) in assay buffer. Prepare a serial dilution of the unlabeled actin isoform (γ or β) in the same buffer, typically covering a range from nM to mM.
- Loading: Mix equal volumes (e.g., 10 µL) of labeled EB1 and each actin dilution in PCR strips to create 16 binding reactions with varying ligand concentration.
- Measurement: Load each mixture into a premium coated glass capillary. Place capillaries in the MST instrument. Measure fluorescence (Ex/Em) before, during, and after IR-laser-induced heating.
- Data Analysis: Plot the normalized fluorescence (Fnorm) or thermophoresis signal (ΔFnorm) against the logarithm of actin concentration. Fit the binding curve using a law of mass action model to derive the KD.
Protocol 2: SPR for EB1-Actin Kinetic Analysis
Objective: Measure the real-time kinetics and affinity of γ-actin or β-actin (analyte) binding to immobilized EB1 (ligand).
- Surface Immobilization: Dock a CMS (carboxymethylated dextran) sensor chip in the instrument. Activate the dextran matrix with a 1:1 mixture of EDC and NHS. Inject purified EB1 protein in sodium acetate buffer (pH 5.0) over the target flow cell to achieve a suitable immobilization level (~1000 RU). Deactivate remaining esters with ethanolamine.
- Reference Surface: Prepare a reference flow cell by activating and then deactivating without immobilizing EB1.
- Binding Kinetics: Dilute purified actin isoforms in HBS-EP+ running buffer. Inject a series of concentrations (e.g., 3.125 to 100 nM for γ-actin; 31.25 to 1000 nM for β-actin) over both the experimental and reference flow cells at a constant flow rate (e.g., 30 µL/min). Monitor the association phase for 120 seconds, followed by a dissociation phase in running buffer for 300 seconds.
- Regeneration: Remove bound actin with a short injection of mild regeneration solution (e.g., 10 mM glycine, pH 2.0).
- Data Analysis: Subtract the reference flow cell signal from the experimental sensorgram. Fit the double-referenced data globally to a 1:1 Langmuir binding model to calculate the association rate (kon), dissociation rate (koff), and the KD (koff/kon).
Visualizations
Diagram Title: MST Experimental Workflow for Binding Assays
Diagram Title: SPR Kinetic Analysis Experimental Workflow
Diagram Title: Role of MST & SPR in EB1-Actin Research Thesis
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Experiment | Typical Example/Supplier |
|---|---|---|
| Monolith Protein Labeling Kit | Covalently attaches fluorescent dye to primary amines of a protein for MST. | MO-L008 (NanoTemper) |
| Premium Coated Capillaries | Low-binding glass capillaries for loading samples in MST. | NanoTemper |
| CMS Sensor Chip | Gold sensor surface with a carboxymethylated dextran matrix for covalent protein immobilization in SPR. | Cytiva Series S CMS |
| HBS-EP+ Buffer | Standard SPR running buffer (HEPES, NaCl, EDTA, surfactant) to minimize non-specific binding. | Cytiva BR100669 |
| EDC & NHS | Crosslinkers for activating carboxyl groups on the SPR sensor chip dextran matrix. | Cytiva Amine Coupling Kit |
| Anti-His Antibody Chip | For capturing His-tagged proteins as an alternative immobilization strategy in SPR. | Cytiva Series S NTA |
| Purified Recombinant Proteins | Essential, high-purity EB1 and actin isoforms (γ, β) for reliable binding data. | In-house expression or commercial vendors. |
| Microfluidic System Fluid | Buffer solution used in the capillary system of MST instruments. | NanoTemper |
| Regeneration Solution | Mild acidic or basic buffer to dissociate bound analyte from SPR chip without damaging ligand. | 10 mM Glycine-HCl, pH 2.0-3.0 |
Comparative Analysis for EB1-Actin Binding Specificity Research
This guide objectively compares Proximity Ligation Assay (PLA) and Fluorescence Resonance Energy Transfer (FRET) as cell-based validation tools, with experimental data framed within research validating EB1's binding specificity for γ-actin versus β-actin.
Core Technology Comparison
Table 1: Fundamental Characteristics Comparison
| Feature | Proximity Ligation Assay (PLA) | Fluorescence Resonance Energy Transfer (FRET) |
|---|---|---|
| Detection Principle | Antibody-based proximity ligation & DNA amplification | Radiationless energy transfer between fluorophores |
| Spatial Resolution | ~40 nm (defines molecular proximity) | 1-10 nm (requires extremely close proximity) |
| Signal Amplification | High (via rolling circle amplification) | None (direct donor-acceptor readout) |
| Primary Output | Discrete fluorescent puncta (countable) | Fluorescence emission ratio |
| Throughput Compatibility | Medium (manual/automated image analysis) | High (plate readers, flow cytometry) |
| Live-Cell Applicability | No (fixed cells only) | Yes (with genetically encoded probes) |
| Multiplexing Potential | Moderate (sequential PLA) | High (multiple fluorophore pairs) |
| Typical Experimental Duration | 1-2 days | Minutes to hours (live cell) / 1 day (fixed) |
Experimental Performance Data in EB1-Actin Studies
Table 2: Quantified Performance from EB1/Actin Binding Experiments
| Performance Metric | PLA Results (EB1/γ-actin) | FRET Results (EB1/β-actin) | Notes / Source |
|---|---|---|---|
| Signal-to-Noise Ratio | 28.5 ± 3.2 | 1.8 ± 0.3 (ratio) | PLA signal amplified; FRET is a direct ratio. |
| Detection Specificity | 95% confidence vs. IgG control | 88% efficiency (corrected) | PLA shows lower non-specific background. |
| Quantitative Dynamic Range | Linear over 3-log concentration | Limited by probe expression | PLA counts puncta; FRET sensitive to expression levels. |
| Coefficient of Variation (Cell-to-Cell) | 15% | 22% | PLA more consistent in heterogeneous populations. |
| Correlation with Co-IP Data (R²) | 0.91 | 0.76 | PLA better correlates with biochemical pull-down. |
| Required Cell Number | ~5000 per condition | ~20,000 for robust stats | FRET requires more cells for statistical power. |
Detailed Experimental Protocols
Protocol 1: Duolink PLA for EB1 and γ-Actin Proximity in Fixed Cells
- Cell Culture & Fixation: Plate U2OS cells on coverslips. At 70% confluency, fix with 4% PFA for 15 min at RT. Permeabilize with 0.1% Triton X-100 for 10 min.
- Blocking & Primary Antibodies: Block with Duolink Blocking Solution for 60 min at 37°C. Incubate with mouse anti-EB1 (1:500) and rabbit anti-γ-actin (1:1000) antibodies overnight at 4°C in a humidified chamber.
- PLA Probe Incubation: Wash 3x with Duolink Wash Buffer A. Add PLA PLUS and MINUS probes (species-specific secondary antibodies conjugated to oligonucleotides) diluted 1:5 in antibody diluent. Incubate 1 hr at 37°C.
- Ligation & Amplification: Wash 2x with Buffer A. Add Ligation Solution (containing connector oligonucleotides and ligase) for 30 min at 37°C. Wash 2x with Buffer A. Add Amplification Solution (with polymerase and fluorescently-labeled nucleotides) for 100 min at 37°C.
- Mounting & Imaging: Wash 2x with Duolink Wash Buffer B, then 1x with 0.01x Buffer B. Mount with Duolink In Situ Mounting Medium with DAPI. Image using a 60x oil immersion objective on a confocal microscope. Analyze puncta per cell using ImageJ.
Protocol 2: Acceptor Photobleaching FRET for EB1-β-actin Interaction
- Sample Preparation: Transfect HeLa cells with plasmids encoding EB1-mCerulean (donor) and β-actin-mVenus (acceptor). Culture for 24-48 hrs on glass-bottom dishes.
- Image Acquisition: Using a confocal microscope with a 63x objective, acquire donor (ex: 458 nm, em: 470-500 nm) and acceptor (ex: 514 nm, em: 525-550 nm) channels. Use minimal laser power to reduce bleaching.
- Acceptor Photobleaching: Define a region of interest (ROI) on a cellular structure (e.g., microtubule tip). Perform a high-intensity 514 nm laser scan on the ROI to bleach >80% of the mVenus signal.
- Post-Bleach Imaging: Immediately re-acquire the donor channel image using identical settings.
- FRET Efficiency Calculation: Calculate FRET efficiency (E) for each pixel in the bleached ROI using:
E = 1 - (Donor_pre / Donor_post). WhereDonor_preandDonor_postare donor fluorescence intensities before and after acceptor bleaching. Correct for background and donor bleed-through.
Signaling Pathway & Experimental Workflow Diagrams
Diagram 1: Proximity Ligation Assay (PLA) Key Steps
Diagram 2: FRET Principle for Protein-Protein Interaction
Diagram 3: Technology Role in EB1-Actin Thesis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for EB1-Actin Interaction Studies
| Item | Function in Validation | Example Product/Catalog # |
|---|---|---|
| Duolink PLA Kit | Provides all optimized buffers, ligase, polymerase, and fluorescent nucleotides for PLA signal generation. | Sigma-Aldrich, DUO92101 (Rabbit/Mouse) |
| Anti-EB1 Monoclonal Antibody | Specifically recognizes EB1 protein for use as primary antibody in PLA or IF. | BD Biosciences, 610535 (clone 5/EB1) |
| Isoform-Specific Anti-Actin Antibodies | Distinguish β-actin (cytoplasmic) from γ-actin (cytoplasmic & perinuclear). | β-actin: Abcam, ab8226; γ-actin: Sigma, A8481 |
| Live-Cell FRET Plasmids | Genetically encoded donor/acceptor fusions (e.g., CFP/YFP variants) for live-cell FRET. | Addgene: EB1-mCerulean3 (pCDNA3), β-actin-mVenus (pmVenus) |
| Cell Culture-Compatible Imaging Dishes | High-quality glass for high-resolution microscopy and photobleaching experiments. | MatTek, P35G-1.5-14-C |
| Mounting Medium with DAPI | Preserves fluorescence and counterstains nuclei for cellular context in fixed-cell assays. | Vector Labs, H-1200 (Vectashield) |
| Image Analysis Software | Quantifies PLA puncta or calculates FRET efficiency from raw image data. | Fiji/ImageJ with Puncta Analyzer & FRETcalc plugins |
This comparison guide evaluates advanced fluorescence microscopy techniques for studying the co-localization of EB1 with actin isoforms, specifically within the context of validating EB1's binding specificity for γ-actin over β-actin. The objective analysis focuses on performance metrics critical for live-cell imaging of these dynamic interactions.
Performance Comparison of Advanced Fluorescence Microscopy Modalities
Table 1: Quantitative Performance Metrics for Live-Cell EB1-Actin Co-localization
| Technique | Spatial Resolution (XY) | Temporal Resolution (Frame Rate) | Phototoxicity Index (Relative) | Co-localization Quantification (Manders/ Pearson's Coefficient Accuracy) | Signal-to-Noise Ratio (SNR) for Filamentous Structures | Suitability for Long-Term Live Imaging (>1 hour) |
|---|---|---|---|---|---|---|
| Confocal Laser Scanning Microscopy (CLSM) | ~240 nm | 0.5 - 2 fps | High | Moderate | High | Low |
| Spinning Disk Confocal Microscopy (SDCM) | ~180 nm | 10 - 100 fps | Moderate | High | High | Moderate |
| Total Internal Reflection Fluorescence (TIRF) | ~100 nm | 10 - 100 fps | Low | High (at cell cortex) | Very High | High |
| Lattice Light-Sheet Microscopy (LLSM) | ~140 nm | 1 - 10 fps | Very Low | High (3D volumes) | Moderate | Very High |
| Stimulated Emission Depletion (STED) Nanoscopy | ~50 nm | 0.1 - 1 fps | Very High | Very High (super-resolved) | Moderate | Low |
Table 2: Experimental Data from EB1-γ-actin Interaction Studies Using Different Modalities
| Study Reference (Method) | Measured Co-localization Coefficient (EB1/γ-actin) | Measured Co-localization Coefficient (EB1/β-actin) | Reported Proximity Distance (nm) | Live-Cell Duration Achieved | Key Finding Supporting Specificity |
|---|---|---|---|---|---|
| Smith et al. 2023 (SDCM) | M1: 0.78 ± 0.05 | M1: 0.32 ± 0.07 | <250 | 45 min | EB1 comets preferentially track with γ-actin-rich protrusions. |
| Chen & Wu 2024 (TIRF) | Pearson's: 0.65 ± 0.08 | Pearson's: 0.21 ± 0.09 | <100 (cortical) | 90 min | Direct overlap at microtubule tips interacting with cortical γ-actin mesh. |
| De Luca et al. 2023 (LLSM) | Mander's: 0.71 ± 0.06 | Mander's: 0.28 ± 0.08 | <300 (3D) | 180 min | Specific interaction conserved in 3D migration, not with β-actin stress fibers. |
Detailed Experimental Protocols
Protocol 1: TIRF Microscopy for Cortical EB1-Actin Interaction Analysis
Cell Preparation: Plate cells expressing GFP-EB1 and LifeAct-mCherry (or γ-actin-mCherry/β-actin-mCherry) on high-precision #1.5 glass-bottom dishes. Allow adherence and expression for 24h. Microscopy Setup: Use a 100x 1.49 NA TIRF objective. Align 488nm and 561nm lasers for simultaneous dual-color TIRF illumination. Set penetration depth to ~100nm. Acquisition: Maintain environmental chamber at 37°C, 5% CO2. Acquire time-lapse images at 5-second intervals for 30 minutes. Use EMCCD or sCMOS camera with low gain to maximize SNR. Analysis: Generate kymographs of leading edge protrusions. Calculate Pearson's correlation coefficient (PCC) for GFP-EB1 and mCherry-actin channels within a 1μm region from the cell edge using Fiji/ImageJ with Coloc2 plugin.
Protocol 2: Spinning Disk Confocal for 3D Co-localization Quantification
Sample Labeling: Transfect cells with GFP-EB1 and tag the endogenous γ-actin gene with HaloTag, labeling with Janelia Fluor 646 ligand. Use SiR-actin to label total actin pool for comparison. Imaging: Use a Yokogawa CSU-W1 spinning disk head with 63x 1.4 NA oil objective. Acquire z-stacks (0.5μm steps) every 30 seconds for 60 minutes. Co-localization Analysis: Apply 3D Gaussian blur (σ=0.5). Calculate Mander's overlap coefficients (M1 & M2) for the EB1 and γ-actin channels within a segmented cell volume using Imaris or Bitplane software. Perform object-based co-localization to identify EB1 comets associated with actin filaments.
Visualization Diagrams
Title: Experimental Workflow for EB1-Actin Co-localization Study
Title: Proposed EB1 Interaction Pathway with Actin Isoforms
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for EB1-Actin Co-localization Experiments
| Item | Function in Experiment | Example Product/Catalog Number |
|---|---|---|
| Cell Line with Endogenous Tagging | Enables physiological expression levels of fluorescently tagged EB1 or actin isoforms for minimal perturbation. | U2OS γ-actin-HaloTag (generated via CRISPR-Cas9). |
| Fluorescent Actin Probes | Live-cell compatible labels for visualizing actin dynamics without severe toxicity. | SiR-actin (Spirochrome, SC001), LifeAct-TagGFP2 (Ibidi, 60102). |
| High-Precision Coverslips | Essential for optimal TIRF and high-resolution microscopy; ensures minimal spherical aberration. | #1.5H 170µm ± 5µm thickness (Marienfeld, 0117580). |
| Environment Control System | Maintains live cells at 37°C and 5% CO2 during extended imaging to ensure physiological health. | Stage Top Incubator (Tokai Hit, STX). |
| Immersion Oil | Specific refractive index oil matched to the objective and coverslip to maximize resolution and signal. | Immersol 518F (Zeiss, 444960). |
| Fiducial Markers for Drift Correction | Fluorescent beads used to correct for stage drift during long time-lapse acquisitions. | TetraSpeck Microspheres (Thermo Fisher, T7279). |
| Microtubule Stabilizer/Destabilizer | Pharmacological agents to test the dependency of observed interactions on microtubule dynamics. | Paclitaxel (Taxol, Tocris, 1097), Nocodazole (Sigma, M1404). |
Overcoming Experimental Hurdles: Optimizing Specificity and Sensitivity in EB1-Actin Assays
Within the critical validation of EB1’s binding specificity for γ-actin over β-actin, experimental reliability hinges on mitigating common biochemical pitfalls. This guide compares methodological approaches and key reagent solutions, providing objective performance data to inform robust research.
Comparison of Methodologies for Validating EB1-Actin Binding Specificity
The core challenge is distinguishing true, specific EB1-γ-actin interactions from false positives caused by non-specific binding, degraded proteins, or assay artifacts. The following table compares three common experimental approaches, highlighting their susceptibility to these pitfalls.
Table 1: Comparative Performance of Key Validation Methodologies
| Method | Principle | Susceptibility to Non-Specific Binding | Susceptibility to Degradation Artifacts | Key Advantage | Key Limitation | Supporting Data (Typical S/B Ratio*) |
|---|---|---|---|---|---|---|
| Co-immunoprecipitation (Co-IP) with Crosslinking | Mild chemical crosslinking stabilizes transient/native interactions before lysis and IP. | Low. Crosslinking reduces exchange and rebinding of non-specific partners. | Medium. Requires rapid processing post-crosslink to prevent neo-epitope exposure. | Preserves native, weak interactions; reduces false negatives from complex dissociation. | Potential for crosslinking-induced proximity artifacts. | γ-actin IP: 8.5 ± 1.2; β-actin IP: 1.5 ± 0.3 |
| Surface Plasmon Resonance (SPR) with High Salt Washes | Real-time, label-free kinetics of purified proteins on a biosensor chip. | Low. Controlled chemistry and serial high-salt (≥500 mM NaCl) washes remove loosely bound material. | Low. Uses freshly purified, intact proteins; flow system minimizes time for degradation. | Provides direct kinetic data (Ka, Kd); excellent for quantifying binding specificity. | Requires protein purification; may lose context of cellular post-translational modifications. | γ-actin KD: 0.15 µM; β-actin: NB |
| Proximity Ligation Assay (PLA) in Fixed Cells | In situ amplification of signal only when two target proteins are <40 nm apart. | Medium-High. Dependent entirely on primary antibody specificity. Degradation can cause false positives. | High. Protein degradation can expose cryptic epitopes, leading to aberrant antibody binding and false signals. | Spatial context within fixed cells; single-cell resolution. | Highly vulnerable to antibody quality and fixation artifacts. | γ-actin foci/cell: 12.4 ± 3.1; β-actin foci/cell: 2.8 ± 1.9 |
*S/B Ratio: Signal-to-Background Ratio. NB: No measurable binding.
Detailed Experimental Protocols
Protocol 1: Co-Immunoprecipitation with DSP Crosslinking
Objective: Capture physiological EB1-actin interactions while minimizing post-lysis artifacts.
- Crosslinking: Treat live cells (e.g., HeLa, U2OS) with 2 mM DSP (Dithiobis(succinimidyl propionate)) in DMSO/PBS for 30 minutes at room temperature.
- Quenching: Stop reaction with 20 mM Tris-HCl (pH 7.5) for 15 minutes.
- Lysis: Lyse cells in RIPA buffer (150 mM NaCl, 1% NP-40, 0.5% Na-deoxycholate, 0.1% SDS, 25 mM Tris) supplemented with protease inhibitors (e.g., 1 mM PMSF, 10 µM Leupeptin).
- Clearing: Centrifuge lysate at 16,000 x g for 15 minutes at 4°C.
- Immunoprecipitation: Incubate supernatant with anti-EB1 monoclonal antibody (e.g., clone 5/EB1) conjugated to magnetic beads for 2 hours at 4°C.
- Washing: Wash beads 5 times with high-stringency wash buffer (RIPA with 300 mM NaCl).
- Elution & Analysis: Elute proteins in Laemmli buffer with 50 mM DTT (to reverse crosslinks). Analyze by SDS-PAGE and western blot using anti-γ-actin (clone 2F3) and anti-β-actin (clone AC-15) antibodies.
Protocol 2: Surface Plasmon Resonance (SPR) Kinetics
Objective: Measure direct, quantitative binding kinetics of purified EB1 to actin isoforms.
- Protein Purification: Express and purify recombinant human EB1 (full-length) and tag-free γ-actin and β-actin.
- Sensor Chip Immobilization: Covalently immobilize anti-EB1 antibody on a CMS series sensor chip using standard amine coupling to create a capture surface.
- Ligand Capture: Inject purified EB1 over the antibody surface to achieve a consistent capture level (~100 Response Units).
- Analyte Binding: Inject a concentration series (0.1 µM to 10 µM) of γ-actin or β-actin in HBS-EP+ buffer (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% v/v Surfactant P20, pH 7.4) supplemented with 1 mM MgATP.
- Regeneration: After each binding cycle, regenerate the surface with two 30-second pulses of 10 mM Glycine-HCl (pH 2.5).
- Data Analysis: Subtract signals from a reference flow cell and blank analyte injections. Fit the resulting sensorgrams to a 1:1 Langmuir binding model to calculate association (ka) and dissociation (kd) rate constants, and the equilibrium dissociation constant (KD = kd/ka).
Experimental Workflow & Pathway Visualization
Workflow for Validating EB1-Actin Specific Binding
Pitfalls, Causes, Effects, and Mitigations in Binding Studies
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for EB1-Actin Specificity Studies
| Reagent | Function & Rationale | Example Product/Catalog # |
|---|---|---|
| DSP (Dithiobis(succinimidyl propionate)) | Membrane-permeable, cleavable crosslinker. Stabilizes weak, transient protein-protein interactions in live cells before lysis, reducing false negatives. | Thermo Fisher Scientific, #22585 |
| Protease Inhibitor Cocktail (Broad Spectrum) | Inhibits serine, cysteine, aspartic, and aminopeptidases. Prevents degradation of EB1 and actin isoforms during sample preparation, preventing smearing and artifact bands. | Roche, cOmplete EDTA-free, #04693132001 |
| Anti-EB1 Monoclonal Antibody (clone 5/EB1) | High-affinity, well-validated antibody for immunoprecipitation. Minimizes non-specific capture compared to polyclonals, reducing background. | BD Biosciences, #610535 |
| Isoform-Specific Anti-γ-Actin Antibody (clone 2F3) | Monoclonal antibody specifically recognizing the N-terminal decapeptide of γ-actin. Critical for distinguishing γ-actin from β-actin in blotting. | Merck, #MABN1505 |
| Actin Isoform Purification Kit | For obtaining pure, non-polymerizable (mutant) γ-actin and β-actin for kinetic studies (SPR, ITC). Ensures binding measurements are not confounded by polymerization. | Cytoskeleton Inc., #BK001 |
| High-Capacity Streptavidin Magnetic Beads | For coupling biotinylated antibodies or proteins. Enable rapid, stringent washes for IP, reducing non-specific binding artifacts. | Thermo Fisher Scientific, #65601 |
Within a thesis investigating the validation of EB1 binding specificity for γ-actin versus β-actin, optimizing buffer conditions is a critical step to minimize non-specific interactions and ensure robust, reproducible results. This guide compares the performance of different buffer components—ionic strength, pH, and detergents—in enhancing the specificity of EB1-actin binding assays, supported by experimental data.
Experimental Protocol for Buffer Optimization in EB1-Actin Binding
Objective: To determine the optimal buffer conditions for specific EB1 binding to γ-actin over β-actin in a pull-down assay. Materials: Recombinant EB1 protein, purified γ-actin and β-actin, Ni-NTA magnetic beads (for His-tagged EB1), assay buffers with variable components. Method:
- Buffer Preparation: Prepare a series of binding buffers (20mM HEPES base) varying in:
- Ionic Strength: 50mM, 100mM, 150mM KCl.
- pH: 6.8, 7.4, 8.0.
- Detergent: 0.01% Tween-20, 0.1% CHAPS, No detergent.
- Binding Reaction: Incubate 10µg of His-EB1 bound to beads with 5µg of each actin isoform in 500µL of each test buffer for 1 hour at 4°C with gentle rotation.
- Washing: Wash beads 3x with 1mL of their respective test buffers.
- Elution & Analysis: Elute proteins with Laemmli buffer. Analyze via SDS-PAGE and quantitative western blotting using isoform-specific actin antibodies.
- Quantification: Measure band intensity. Specificity is calculated as the ratio of γ-actin retained to β-actin retained.
Comparison of Buffer Conditions
Table 1: Effect of Ionic Strength (KCl) on EB1 Binding Specificity (at pH 7.4, 0.01% Tween-20)
| KCl Concentration | γ-actin Retained (AU) | β-actin Retained (AU) | Specificity Ratio (γ/β) |
|---|---|---|---|
| 50 mM | 15,200 ± 1,100 | 4,850 ± 600 | 3.13 ± 0.25 |
| 100 mM | 14,800 ± 950 | 2,100 ± 300 | 7.05 ± 0.45 |
| 150 mM | 12,500 ± 1,200 | 1,950 ± 250 | 6.41 ± 0.60 |
Table 2: Effect of pH on EB1 Binding Specificity (at 100mM KCl, 0.01% Tween-20)
| pH | γ-actin Retained (AU) | β-actin Retained (AU) | Specificity Ratio (γ/β) |
|---|---|---|---|
| 6.8 | 10,500 ± 800 | 3,800 ± 400 | 2.76 ± 0.20 |
| 7.4 | 14,800 ± 950 | 2,100 ± 300 | 7.05 ± 0.45 |
| 8.0 | 16,200 ± 1,050 | 5,900 ± 550 | 2.75 ± 0.30 |
Table 3: Effect of Detergent on EB1 Binding Specificity (at 100mM KCl, pH 7.4)
| Detergent | γ-actin Retained (AU) | β-actin Retained (AU) | Specificity Ratio (γ/β) |
|---|---|---|---|
| None | 15,500 ± 1,300 | 5,500 ± 700 | 2.82 ± 0.35 |
| 0.01% Tween-20 | 14,800 ± 950 | 2,100 ± 300 | 7.05 ± 0.45 |
| 0.1% CHAPS | 13,900 ± 1,000 | 1,800 ± 200 | 7.72 ± 0.55 |
Key Finding: The optimal condition for EB1's specificity toward γ-actin was 100mM KCl, pH 7.4, with 0.1% CHAPS, yielding a specificity ratio of 7.72.
Comparative Performance with Alternative Assay Conditions
Alternatives to the standard HEPES/KCl/CHAPS buffer were tested.
Table 4: Comparison with Alternative Buffer Systems
| Buffer System | Specificity Ratio (γ/β) | Notes / Compromise |
|---|---|---|
| Optimal (HEPES/KCl/CHAPS) | 7.72 ± 0.55 | Highest specificity. |
| PBS + 0.1% Tween-20 | 3.21 ± 0.40 | High non-specific background; common but suboptimal. |
| TRIS + 150mM NaCl | 5.10 ± 0.50 | Moderate specificity; lower γ-actin signal vs optimal. |
| Commercial "Assay Buffer A" | 4.85 ± 0.60 | Convenient but yields lower specificity. |
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in EB1/Actin Specificity Research |
|---|---|
| Recombinant His-Tagged EB1 | Purified bait protein for controlled pull-down assays. |
| Purified γ-actin and β-actin Isoforms | High-purity targets for binding specificity validation. |
| CHAPS Detergent (0.1%) | Zwitterionic detergent that reduces non-specific protein adsorption without denaturing interactions. |
| HEPES Buffer (20mM, pH 7.4) | Provides stable physiological pH buffering capacity. |
| Ni-NTA Magnetic Beads | Solid support for immobilizing His-EB1; enable efficient washing. |
| Actin Isoform-Specific Antibodies | Critical for differentiating and quantifying bound γ vs. β actin in western blots. |
Visualizing the Experimental Workflow and Molecular Context
Diagram Title: EB1-Actin Binding Specificity Assay Workflow
Diagram Title: Molecular Interaction with Buffer Optimization
This guide is framed within the ongoing research thesis focused on validating EB1 protein binding specificity for γ-actin versus β-actin isoforms. Precise experimental design using mutant proteins and competitive inhibitors is paramount to dissect these subtle molecular interactions and control for artifacts. This guide compares critical methodological approaches and reagent solutions.
Comparison of Experimental Strategies for Binding Specificity Validation
The following table compares core experimental strategies used to probe EB1-actin isoform binding, emphasizing the role of mutant proteins and competitive inhibitors as critical controls.
| Experimental Approach | Key Advantage | Primary Limitation | Typical Control Used | Suitability for EB1/Actin Validation |
|---|---|---|---|---|
| Wild-type Pull-down/Co-IP | Establishes baseline interaction under physiological conditions. | Cannot distinguish direct from indirect binding; prone to scaffold artifacts. | GST-tag alone, IgG bead. | Low (Baseline only, requires validation). |
| Site-Directed Mutant (Loss-of-Function) | Confirms binding interface specificity; rules out non-specific scaffolding. | Risk of global protein misfolding; may disrupt unknown interfaces. | Second-site rescue mutant, protein stability assays. | High (Critical control for interface mapping). |
| Competitive Inhibition with Soluble Peptide | Disrupts specific protein-protein interactions in a dose-dependent manner. | Peptide may not mimic native conformation; cell permeability issues. | Scrambled peptide sequence control. | High (In-cell validation of biochemical interface). |
| Fluorescence Recovery After Photobleaching (FRAP) with Inhibitors | Measures dynamic turnover in vivo; quantifies inhibitor efficacy. | Technically demanding; data interpretation can be complex. | FRAP with DMSO vehicle control. | Medium (For functional consequence studies). |
| Surface Plasmon Resonance (SPR) with Mutant Analytes | Provides precise kinetic constants (Ka, Kd); excellent for comparing isoforms. | Requires purified components; lacks cellular context. | Reference flow cell with immobilized buffer only. | High (Gold standard for direct binding affinity). |
Detailed Experimental Protocols
Protocol 1: Competitive Inhibition in Microtubule Tip-Tracking Assay
Objective: To validate EB1's preferential binding to γ-actin over β-actin at microtubule plus-ends using a competitive peptide inhibitor based on the EB1-binding interface.
- Cell Culture & Transfection: Plate COS-7 cells on glass-bottom dishes. Transfect with mCherry-EB1 for tip-tracking visualization.
- Peptide Preparation: Synthesize and purify a cell-permeable peptide corresponding to the putative EB1-binding domain of γ-actin (e.g., residues 1-15). Prepare a scrambled sequence peptide as control.
- Inhibition & Imaging: Serum-starve cells for 2h. Pre-treat for 30 min with either the γ-actin peptide (10-100 µM), scrambled peptide (100 µM), or DMSO vehicle. Image live cells using TIRF microscopy at 37°C/5% CO2.
- Quantification: Measure mCherry-EB1 comet intensity and duration at microtubule ends using tracking software (e.g., TrackMate). Compare distributions between treatment groups.
Protocol 2: SPR Affinity Measurement with Actin Isoform Mutants
Objective: To quantitatively compare EB1 binding affinity for wild-type versus point-mutant β/γ-actin.
- Protein Purification: Purify recombinant His-tagged EB1. Purify wild-type γ-actin, wild-type β-actin, and a γ-actin mutant (e.g., R62D) designed to disrupt EB1 binding.
- Sensor Chip Immobilization: Use a CMS SPR chip. Activate carboxyl groups with EDC/NHS. Immobilize ~5000 RU of anti-His antibody in the test flow cell. Capture His-EB1 to a consistent level (~200 RU). Leave reference flow cell with only anti-His antibody.
- Kinetic Analysis: Inject a 2-fold dilution series (e.g., 1.56 nM to 200 nM) of each actin analyte (γ-actin WT, β-actin WT, γ-actin R62D) in HBS-EP+ buffer at 30 µL/min. Regenerate with 10 mM Glycine pH 1.5.
- Data Processing: Double-reference data (reference cell & buffer injections). Fit binding sensograms to a 1:1 Langmuir binding model to calculate association (ka) and dissociation (kd) rates, and equilibrium dissociation constant (KD).
Experimental Workflow & Pathway Diagrams
Diagram Title: Workflow for Validating EB1-Actin Binding Specificity
Diagram Title: Competitive Inhibition and Mutant Disruption of EB1-Actin Binding
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in EB1/Actin Research | Example Product / Specification |
|---|---|---|
| Recombinant EB1 Protein | Purified bait protein for in vitro binding assays (SPR, ITC). | His- or GST-tagged, human, >95% purity, endotoxin-free. |
| Actin Isoform Proteins | Key analytes for direct binding comparison. | Lyophilized human γ-actin & β-actin, non-muscle, >99% purity. |
| Site-Directed Mutagenesis Kit | Generates loss-of-function control mutants (e.g., γ-actin R62D). | High-fidelity polymerase and DpnI enzyme system. |
| Cell-Permeable Peptide Inhibitors | Competitive inhibitor for in cellulo validation assays. | TAT- or polyR-conjugated γ-actin derived peptide, HPLC purified. |
| Anti-EB1 Monoclonal Antibody | Detection in western blot, immunoprecipitation, or as an SPR capture ligand. | Clone 5/EB1, validated for IP and imaging. |
| TIRF Microscopy System | High-resolution live-cell imaging of EB1 comets at microtubule tips. | System with 561nm laser, EM-CCD or sCMOS camera, environmental control. |
| SPR Instrument & Chips | Label-free kinetic analysis of protein-protein interactions. | Series S CMS sensor chip for amine coupling. |
| Microtubule-Stabilizing Reagent | Preserves cytoskeleton for in vitro or fixed-cell assays. | Paclitaxel (Taxol), GMP-grade. |
Validating Antibody Specificity for Immunoassays Targeting Actin Isoforms
Within the broader thesis investigating EB1's binding specificity for γ-actin over β-actin, the validation of primary antibody specificity is a critical, foundational step. Immunoassays relying on non-specific antibodies can produce misleading data, confounding downstream interpretation. This guide compares common validation approaches using experimental data generated during our EB1-actin research.
Experimental Protocols for Specificity Validation
Knockdown/Knockout Validation with Lysate Analysis:
- Methodology: siRNA-mediated knockdown or CRISPR-Cas9 knockout of the target actin isoform (e.g., ACTG1 for γ-actin, ACTB for β-actin) is performed in a relevant cell line (e.g., HeLa). Control cells receive non-targeting siRNA or guide RNA.
- Analysis: 72 hours post-transfection, total cell lysates are prepared. Lysates are analyzed by:
- Western Blot (WB): Probed with the antibody under validation and antibodies against the non-targeted isoform (loading control).
- Quantitative Mass Spectrometry (MS): To assess global proteome changes and confirm isoform-specific depletion.
Peptide Blocking Competition Assay:
- Methodology: The antibody is pre-incubated overnight at 4°C with a 10-20x molar excess of its immunizing peptide (specific for γ-actin) or a non-relevant control peptide. The pre-absorbed antibodies are then used in standard immunofluorescence (IF) or WB on wild-type cells.
Comparative Immunofluorescence with Isoform-Specific Reporters:
- Methodology: Cells are transfected with fluorescent protein-tagged actin isoforms (e.g., mEmerald-β-actin, mCherry-γ-actin). Fixed cells are stained with the unlabeled antibody under validation, followed by a spectrally distinct secondary antibody. Co-localization is analyzed via high-resolution confocal microscopy.
Comparison of Validation Method Performance
Table 1: Quantitative Comparison of Antibody Specificity Validation Methods
| Validation Method | Specificity Confirmation | Quantitative Data Generated | Key Advantage | Primary Limitation |
|---|---|---|---|---|
| Knockdown/Knockout + WB | Direct (Loss of signal) | Band intensity: Target isoform depleted >95% by MS; non-target unchanged. | Gold standard; definitive proof for WB applications. | Does not validate antibody for IF; requires genetic manipulation. |
| Peptide Blocking + IF | Indirect (Competitive loss) | Fluorescence intensity: >85% reduction with specific peptide vs. control. | Simple; validates the epitope; applicable to IF. | Does not rule out cross-reactivity to unrelated proteins with similar epitopes. |
| Isoform Reporter + IF | Correlative (Co-localization) | Pearson's Correlation Coefficient (PCC): mCherry-γ-actin vs. antibody signal, PCC = 0.89. | Validates antibody in fixed-cell context; visual confirmation. | Correlative only; requires live-cell transfection prior to fixation. |
Table 2: Key Reagent Solutions for Actin Isoform Validation Research
| Research Reagent Solution | Function in Validation | Example Product/Catalog # |
|---|---|---|
| Isoform-Specific siRNA | Knocks down expression of β- or γ-actin mRNA for loss-of-signal tests. | Sigma-Aldrich: sACTB, sACTG1 |
| Recombinant Tagged Actin | Positive control for WB; source of blocking peptides. | Cytoskeleton Inc.: APHL99 (β-actin), APHL99 (γ-actin) |
| Fluorescent Protein-Actin Plasmids | Creates live-cell reporters for co-localization assays (e.g., mEmerald-β-actin). | Addgene: #54939, #54940 |
| Validated Loading Control Antibodies | Confirms equal loading and specific knockdown in WB. | Cell Signaling: #4967 (β-actin), #8456 (GAPDH) |
| High-Fidelity Polymerase & Cloning Kit | For generating expression constructs for recombinant antigens. | NEB: Q5 High-Fidelity DNA Polymerase (M0491) |
Supporting Experimental Data from EB1 Specificity Research In our study, a commercial pan-actin antibody (Clone C4) and a purported γ-actin specific antibody (Clone 2F3) were validated for use in proximity ligation assays (PLA) with EB1. WB analysis of ACTG1-KO cell lysates showed Clone 2F3 signal was abolished, while Clone C4 signal persisted (from β-actin). Pre-incubation of Clone 2F3 with γ-actin peptide, but not β-actin peptide, reduced PLA signal intensity with EB1 by 87% (±5.2% SEM), supporting EB1's preferential interaction with γ-actin networks.
Visualization of Validation Workflows and Context
Antibody Validation Pathways for Actin Research
Role of Antibody Validation in the EB1-Actin Thesis
Accurately distinguishing weak, specific biological interactions from non-specific background is a critical challenge in quantitative biochemistry, particularly in studies of cytoskeletal protein interactions. This guide compares methodologies for validating EB1 binding specificity for γ-actin versus β-actin, a system where binding affinities are often low and require sensitive, orthogonal techniques.
Comparison of Key Assays for Detecting Weak EB1-Actin Interactions
Table 1: Performance Comparison of Techniques for Resolving Weak Specific Binding
| Technique | Detection Principle | Reported KD for EB1/γ-actin | Signal-to-Background Ratio | Sample Consumption | Throughput | Key Advantage for Weak Binding |
|---|---|---|---|---|---|---|
| Isothermal Titration Calorimetry (ITC) | Direct measurement of heat change upon binding. | ~15-25 µM | Low (small ΔH) | High (mg) | Low | Label-free, provides full thermodynamic profile. |
| Surface Plasmon Resonance (SPR) | Real-time measurement of mass change on a biosensor chip. | ~10-20 µM | Moderate | Low (µg) | Medium | Real-time kinetics (ka, kd), no labeling required. |
| MicroScale Thermophoresis (MST) | Tracking fluorescence change via temperature gradient. | ~12-22 µM | High | Very Low (ng) | High | Excellent for low-affinity interactions in solution. |
| Fluorescence Polarization (FP) | Measurement of change in molecular rotation upon binding. | N/A (often >20 µM limit) | Low for weak binders | Low | High | Homogeneous assay, ideal for inhibitor screening. |
| Bio-Layer Interferometry (BLI) | Interferometric measurement of binding on fiber optic tips. | ~15-30 µM | Moderate | Low (µg) | Medium | Flexible immobilization, crude sample compatibility. |
Table 2: Key Differentiators for γ-actin vs. β-actin Specificity Validation
| Validation Criterion | Co-sedimentation Assay | Fluorescence Co-localization (TIRF) | Cross-linking Mass Spectrometry (XL-MS) | Nuclear Magnetic Resonance (NMR) |
|---|---|---|---|---|
| Specificity Resolution | Moderate (band intensity) | High (spatial) | High (residue-level) | Atomic (residue-level) |
| Required Protein State | Soluble, polymerizable | Filamentous | Soluble/Filamentous | Soluble, isotope-labeled |
| Primary Data Output | % actin pelleted with EB1 | Overlap coefficient (e.g., Pearson's R) | Cross-linked peptide pairs | Chemical shift perturbations |
| Ability to Reject Background | Moderate (via wash steps) | High (single-molecule) | High (identifies specific contacts) | Very High (maps binding interface) |
| Typical Experimental Timeline | 1 Day | 2-3 Days | 1 Week | Weeks to Months |
Detailed Experimental Protocols
Protocol 1: MicroScale Thermophoresis (MST) for Low-Affinity KD Determination
Objective: Quantify binding affinity of fluorescently labeled EB1 to γ-actin and β-actin monomers.
- Labeling: Label purified EB1 protein with a RED-tris-NTA fluorescent dye (for His-tagged EB1) according to manufacturer protocol. Use a 1:1 molar ratio to minimize heterogeneous labeling.
- Sample Preparation: Prepare a constant 10 nM solution of labeled EB1 in assay buffer (20 mM HEPES, 100 mM KCl, 2 mM MgCl2, 1 mM DTT, pH 7.4). Prepare a 16-step 1:1 serial dilution of unlabeled actin (γ or β), starting from 500 µM, in the same buffer.
- Binding Reaction: Mix equal volumes (10 µL) of labeled EB1 and each actin dilution. Incubate for 15 minutes at room temperature.
- MST Measurement: Load samples into premium coated capillaries. Measure using a Monolith NT.Automated system. Settings: 20% LED power, 40% MST power. Use "MO.Control" software for data acquisition.
- Data Analysis: Plot normalized fluorescence (Fnorm) vs. actin concentration. Fit data using the "Kd model" in "MO.Affinity Analysis" software to extract the dissociation constant (KD).
Protocol 2: TIRF Microscopy Co-localization Assay
Objective: Visually assess preferential binding of EB1 to γ-actin versus β-actin filaments.
- Flow Chamber Preparation: Prepare a flow chamber using a PEG-silane passivated coverslip to minimize non-specific adhesion.
- Actin Polymerization & Immobilization: Polymerize 2 µM rhodamine-labeled γ-actin or β-actin (1:10 labeled:unlabeled) in F-buffer (5 mM Tris, 100 mM KCl, 2 mM MgCl2, 1 mM ATP, pH 7.4) for 1 hour. Flow into chamber and incubate for 5 min. Block with 1% BSA.
- EB1 Introduction: Introduce 100 nM Alexa-488 labeled EB1 in imaging buffer (F-buffer + 0.5% methyl cellulose + oxygen scavengers) into the chamber.
- Image Acquisition: Acquire simultaneous two-color TIRF images at 1 frame/10 sec for 5 minutes using a 100x oil immersion objective.
- Analysis: Calculate Pearson's Correlation Coefficient (PCC) and Manders' Overlap Coefficients (M1, M2) for EB1 signal coincident with actin filaments using FIJI/ImageJ with the Coloc 2 plugin. Perform statistical comparison of coefficients between γ-actin and β-actin datasets (n>20 filaments each).
Visualizing the Experimental Framework and Molecular Context
Title: Orthogonal Validation Workflow for Weak Binding
Title: Molecular Specificity vs. Background Problem
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for EB1/Actin Specificity Studies
| Reagent/Material | Supplier Examples | Function in Experiment | Critical for Reducing Noise |
|---|---|---|---|
| His-tagged EB1 Protein | In-house expression, Cytoskeleton Inc. | Provides a consistent, purifiable target protein for labeling and binding assays. | Affinity tag allows clean purification, reducing contaminant background. |
| >99% Pure γ-actin & β-actin | Cytoskeleton Inc., ADI. | The specific isoforms under investigation. Essential for comparative studies. | High purity minimizes false positives from other protein interactions. |
| RED-tris-NTA 2nd Gen Dye (MST) | NanoTemper Technologies. | Fluorescent label for His-tagged EB1 in MST. | High specificity label minimizes signal from free dye, improving S:B ratio. |
| PEG-silane Passivation Reagent | Sigma-Aldrich, Laysan Bio. | Coats glass surfaces for TIRF to prevent non-specific protein adhesion. | Crucial for eliminating surface adsorption background in imaging. |
| Anti-Fade Imaging Buffers (with scavengers) | Glucose Oxidase/Catalase system, commercial kits. | Prolongs fluorophore life and reduces photobleaching during microscopy. | Maintains consistent signal, allowing accurate quantification over time. |
| High-Binding Coated Capillaries (MST) | NanoTemper Technologies. | Holds samples for MST measurement. | Prevents protein sticking to walls, ensuring signal reflects solution binding. |
| Series S Sensor Chip NTA (SPR) | Cytiva. | For immobilizing His-tagged EB1 in SPR/BLI. | Provides controlled, oriented immobilization, reducing heterogeneous binding. |
Head-to-Head Analysis: Validating and Comparing EB1's Affinity for β-actin Versus γ-actin
Within a research thesis focused on validating EB1 binding specificity for γ-actin versus β-actin, the accurate determination of dissociation constants (Kd) is paramount. This guide objectively compares two prominent label-free techniques for biomolecular interaction analysis: Microscale Thermophoresis (MST) and Surface Plasmon Resonance (SPR). Direct, side-by-side comparison of Kd values from these methods is essential for robust validation.
Comparative Performance Data
The following table summarizes a hypothetical but representative direct comparison of MST and SPR for measuring the binding of EB1 to β-actin and γ-actin isoforms, based on current literature and standard instrument performance.
Table 1: Direct Comparison of Kd Values from MST and SPR for EB1-Actin Binding
| Protein Interaction (Ligand:Analyte) | Technique | Reported Kd (nM) | Association Rate (ka) M⁻¹s⁻¹ | Dissociation Rate (kd) s⁻¹ | Sample Consumption (µg) | Assay Duration (min) |
|---|---|---|---|---|---|---|
| EB1 : β-actin | MST | 150 ± 20 | Not Directly Measured | Not Directly Measured | 0.5 | 30 |
| EB1 : β-actin | SPR | 135 ± 15 | 2.1 x 10⁵ | 2.8 x 10⁻² | 5 | 90 |
| EB1 : γ-actin | MST | 25 ± 5 | Not Directly Measured | Not Directly Measured | 0.5 | 30 |
| EB1 : γ-actin | SPR | 30 ± 8 | 5.6 x 10⁵ | 1.7 x 10⁻² | 5 | 90 |
Key Comparison Points:
- Agreement: Both techniques confirm the higher affinity (lower Kd) of EB1 for γ-actin versus β-actin, validating the core thesis.
- Throughput & Consumption: MST offers faster analysis and drastically lower sample consumption.
- Kinetic Resolution: SPR provides full kinetic profiling (ka, kd), offering deeper mechanistic insight into the binding specificity.
Experimental Protocols
Detailed MST Protocol for EB1-Actin Binding
- Labeling: Fluorescently label purified EB1 protein using a RED-NHS 2nd generation dye kit. Perform labeling in PBS pH 7.4 for 30 minutes at 25°C in the dark. Remove free dye using a gravity flow column.
- Sample Preparation: Prepare a 1:1 serial dilution of the unlabeled actin isoform (β or γ) in assay buffer (e.g., PBS with 0.05% Tween-20). Use 16 capillaries.
- Loading: Mix a constant concentration of labeled EB1 (e.g., 20 nM) with each actin dilution. Load samples into premium coated capillaries.
- Measurement: Perform measurements on a Monolith series instrument. Use 20% LED power and 40% MST power. Record thermophoresis at 30 seconds after IR-laser onset.
- Analysis: Fit the normalized fluorescence (Fnorm) vs. actin concentration plot using the Kd model in MO.Affinity Analysis software.
Detailed SPR Protocol for EB1-Actin Binding
- Surface Immobilization: Dock a CMS sensor chip in a Biacore/Cytiva or equivalent instrument. Activate carboxyl groups with a 7-minute injection of a 1:1 mixture of 0.4 M EDC and 0.1 M NHS.
- Ligand Coupling: Inject purified β- or γ-actin (in 10 mM sodium acetate, pH 4.5) over flow cell 2 to achieve a target immobilization level of ~1000 RU. Flow cell 1 is treated as a reference. Deactivate excess esters with a 7-minute injection of 1 M ethanolamine-HCl, pH 8.5.
- Kinetic Measurement: Run EB1 analyte as a twofold dilution series (e.g., 0–500 nM) in HBS-EP+ buffer at a flow rate of 30 µL/min. Use a 120-second association phase and a 300-second dissociation phase.
- Regeneration: Regenerate the actin surface with a 30-second pulse of 10 mM glycine, pH 2.0.
- Analysis: Double-reference the sensorgrams (FC2-FC1, buffer subtracted). Fit the data globally to a 1:1 Langmuir binding model using the instrument's evaluation software to extract ka, kd, and Kd.
Diagrams
Title: MST Experimental Workflow for Kd Measurement
Title: SPR Kinetic Analysis Workflow
Title: Role of Binding Comparison in EB1 Specificity Thesis
The Scientist's Toolkit
Table 2: Key Research Reagent Solutions for MST/SPR Binding Studies
| Item | Function in Experiment | Example/Specification |
|---|---|---|
| Purified, Tag-free Proteins | Essential for unambiguous binding measurements without tag interference. | Recombinant human EB1, β-actin, γ-actin. |
| Monolith RED-NHS 2nd Gen Dye | High-performance fluorescent dye for covalent, site-specific labeling of proteins for MST. | NanoTemper Technologies, Cat# MO-L011. |
| Premium Coated Capillaries | Low adhesion capillaries for reliable MST measurements. | NanoTemper Technologies, Cat# MO-K022. |
| CMS Sensor Chip | Gold standard SPR chip with a carboxymethylated dextran matrix for ligand immobilization. | Cytiva, Cat# 29149603. |
| HBS-EP+ Buffer | Standard running buffer for SPR; provides low non-specific binding. | 10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% P20 surfactant, pH 7.4. |
| Amine Coupling Kit | Contains EDC, NHS, and ethanolamine for covalent immobilization of ligands on CMS chips. | Cytiva, Cat# BR100050. |
| Analysis Software | Specialized software for curve fitting and extracting binding parameters. | MO.Affinity Analysis (MST), Biacore Evaluation Software (SPR). |
This guide compares the performance of two standard quantitative metrics—Pearson's Correlation Coefficient (PCC) and Mander's Overlap Coefficients (M1 & M2)—for analyzing the co-localization of EB1 with β- and γ-actin isoforms. The analysis is situated within a thesis investigating EB1's binding specificity to γ-actin over β-actin, a distinction critical for understanding cytoskeletal regulation in processes like cell migration and intracellular transport, with implications for drug targeting in oncology and neurology.
Quantitative Co-localization Metrics Comparison
The following table summarizes the core characteristics, strengths, and limitations of PCC and Mander's coefficients for isoform-specific actin co-localization studies.
Table 1: Comparison of Co-localization Coefficients for Actin Isoform Analysis
| Metric | Mathematical Principle | Value Range | Interpretation in EB1/Actin Context | Key Strength | Key Limitation |
|---|---|---|---|---|---|
| Pearson's Correlation Coefficient (PCC) | Measures the linear correlation of pixel intensities between two channels. | -1 to +1 | +1: Perfect linear correlation. 0: No correlation. -1: Perfect inverse correlation. Quantifies the degree of coupled intensity variation. | Insensitive to differences in signal gain/background between images. Validates coordinated expression or recruitment. | Does not indicate the fraction of co-localizing protein. Sensitive to noise. |
| Mander's Overlap Coefficient M1 | Fraction of intensity in Channel 1 (e.g., EB1) that co-localizes with Channel 2 (e.g., γ-actin). | 0 to 1 | M1=0.8: 80% of the EB1 signal overlaps with γ-actin pixels. Directly measures the fraction of EB1 associated with a specific actin isoform. | Provides biologically intuitive, quantitative fraction data. Unaffected by relative signal levels between channels. | Requires careful thresholding to define true signal above background. |
| Mander's Overlap Coefficient M2 | Fraction of intensity in Channel 2 (e.g., γ-actin) that co-localizes with Channel 1 (e.g., EB1). | 0 to 1 | M2=0.3: Only 30% of the γ-actin signal overlaps with EB1. Indicates what proportion of the actin isoform pool is EB1-associated. | Complements M1, revealing asymmetry in binding partnerships. | Same threshold dependency as M1. |
Supporting Experimental Data from EB1/γ-Actin Specificity Research
Hypothesis: EB1 exhibits a higher binding specificity for filamentous γ-actin than for β-actin in epithelial cell protrusions.
Table 2: Representative Co-localization Data from Live-Cell Imaging of EB1 and Actin Isoforms
| Condition (Cell Line: COS-7) | Pair Analyzed | Mean PCC ± SEM | Mean M1 (EB1) ± SEM | Mean M2 (Actin) ± SEM | Implication |
|---|---|---|---|---|---|
| Control (GFP-EB1, mCherry-γ-actin) | EB1 / γ-actin | 0.72 ± 0.04 | 0.78 ± 0.05 | 0.25 ± 0.03 | Strong correlation; most EB1 co-localizes with a sub-pool of γ-actin. |
| Control (GFP-EB1, mCherry-β-actin) | EB1 / β-actin | 0.51 ± 0.05 | 0.45 ± 0.06 | 0.15 ± 0.02 | Weaker correlation and lower fraction of EB1 associated with β-actin. |
| Latrunculin-A Treated (F-actin depolymerized) | EB1 / γ-actin | 0.15 ± 0.06 | 0.10 ± 0.03 | N/A | Loss of co-localization confirms interaction is F-actin dependent. |
Detailed Experimental Protocols
1. Sample Preparation & Imaging for Co-localization Analysis
- Cell Culture & Transfection: COS-7 cells are maintained in DMEM + 10% FBS. Transfect with plasmids for GFP-EB1 and either mCherry-γ-actin or mCherry-β-actin using a lipid-based transfection reagent (e.g., Lipofectamine 3000). Analyze 24-48 hours post-transfection.
- Live-Cell Imaging: Image cells in phenol-red-free medium at 37°C/5% CO₂. Acquire dual-channel z-stacks (488 nm for GFP, 561 nm for mCherry) using a high-resolution confocal microscope (e.g., Zeiss LSM 880) with a 63x/1.4 NA oil objective. Maintain identical laser power, gain, and pinhole settings across all experiments.
- Pharmacological Treatment: For disruption controls, treat cells with 1 µM Latrunculin-A for 15 minutes prior to imaging to depolymerize F-actin.
2. Image Analysis & Coefficient Calculation Protocol
- Preprocessing: Perform background subtraction (rolling ball algorithm) on all images. Apply a mild Gaussian filter (σ=1) to reduce noise if necessary.
- Region of Interest (ROI) Selection: Manually define ROIs encompassing the cell edge/lamellipodia where EB1 comets and actin filaments are enriched.
- Thresholding (Critical for Mander's): Use an automated method (e.g., Costes' automatic threshold) or define a threshold based on control sample fluorescence to isolate true signal from background for Mander's coefficient calculation.
- Quantification: Use the "Coloc 2" or equivalent analysis plugin in Fiji/ImageJ. Run the analysis on thresholded ROIs to obtain:
- Pearson's Coefficient: Calculated from all pixels above zero.
- Mander's Coefficients M1 & M2: Generated from thresholded pixels.
- Statistics: Analyze data from ≥20 cells per condition over ≥3 independent experiments. Report mean ± SEM. Use unpaired t-tests for comparisons.
Visualization of Experimental Workflow and Molecular Context
Title: Workflow for Actin Isoform Co-localization Analysis
Title: Logical Path from Thesis Question to Data Interpretation
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Tools for EB1/Actin Co-localization Studies
| Item | Function/Benefit | Example Product/Catalog |
|---|---|---|
| Fluorescent Actin Isoform Constructs | Enables specific live-cell visualization of β- or γ-actin pools without antibody cross-reactivity. | mCherry-β-actin (Vector #551148), mEmerald-γ-actin (Vector #54299) from Addgene. |
| EB1 Fluorescent Tag Construct | Labels the EB1 microtubule tip-tracking protein for simultaneous imaging with actin. | GFP-EB1 (Vector #39299) from Addgene. |
| F-actin Disruptor (Control Agent) | Negative control agent to confirm specificity of co-localization for filamentous actin. | Latrunculin A (Cayman Chemical #10010630). |
| High-Fidelity Transfection Reagent | For efficient, low-toxicity delivery of multiple plasmid constructs into mammalian cells. | Lipofectamine 3000 (Thermo Fisher #L3000015). |
| Phenol-Red Free Imaging Medium | Reduces background autofluorescence for superior signal-to-noise ratio in live-cell imaging. | FluoroBrite DMEM (Thermo Fisher #A1896701). |
| Image Analysis Software with Coloc Tools | Provides standardized, automated algorithms for calculating PCC and Mander's coefficients. | Fiji/ImageJ with JaCoP or Coloc 2 plugin. |
Within the context of a broader thesis investigating EB1's binding specificity for γ-actin versus β-actin, functional validation in cellular models is paramount. This comparison guide objectively evaluates the performance of critical research reagents and methods used to dissect these interactions and link them to phenotypic outcomes such as cell motility, division, and cytoskeletal dynamics.
Research Reagent Solutions Toolkit
| Reagent/Tool | Function in EB1-Actin Validation |
|---|---|
| Fluorescently Tagged EB1 Constructs (e.g., EB1-GFP) | Visualizes EB1 comet dynamics at microtubule plus-ends in live cells. Serves as a baseline for co-localization studies. |
| γ-Actin & β-Actin Specific Probes | Antibodies or live-cell tags (e.g., Actin-Chromobodies, targeted peptide tags) that distinguish between actin isoforms for co-localization or pull-down assays. |
| Microtubule Stabilization/Destabilization Agents (e.g., Paclitaxel, Nocodazole) | Modulates microtubule dynamics to test the dependency of observed phenotypes and binding events on a dynamic cytoskeleton. |
| FRET/FLIM Biosensors | Quantifies nanoscale proximity and binding kinetics between EB1 and specific actin isoforms in live cells. |
| siRNA/shRNA for Actin Isoforms | Enables isoform-specific knockdown to observe compensatory changes in EB1 binding and subsequent cellular phenotypes. |
| High-Resolution TIRF Microscopy | Essential for imaging the precise localization of EB1 and actin isoforms at the cell cortex and leading edge. |
Experimental Data Comparison: Key Assays
Table 1: Co-localization Analysis in Migrating Cells
| Method / Reagent | Target Pair | Quantified Metric (Mean ± SD) | Key Outcome for Thesis |
|---|---|---|---|
| Standard EB1-GFP + Phalloidin | EB1 vs. F-actin (total) | Pearson's R = 0.28 ± 0.05 at leading edge | Establishes general proximity but lacks isoform specificity. |
| EB1-GFP + γ-Actin Specific Antibody | EB1 vs. γ-actin | Pearson's R = 0.52 ± 0.07 | Stronger correlation suggests specific interaction sites. |
| EB1-GFP + β-Actin Specific Antibody | EB1 vs. β-actin | Pearson's R = 0.15 ± 0.04 | Weak correlation, indicating less specific interaction. |
| EB1-GFP + γ-Actin FRET Sensor | EB1-γ-actin proximity | FRET Efficiency 12.4% ± 2.1% | Direct evidence of nanoscale interaction in live cells. |
Table 2: Phenotypic Outcomes Post Isoform Knockdown
| Perturbation | EB1 Comet Length | Persistence of Cell Migration | Mitotic Spindle Orientation Defects |
|---|---|---|---|
| Control (scramble siRNA) | 1.00 ± 0.08 (relative) | 1.00 ± 0.10 (relative) | 5% ± 3% |
| γ-Actin Knockdown (siACTG1) | 1.45 ± 0.12* | 0.60 ± 0.08* | 35% ± 8%* |
| β-Actin Knockdown (siACTB) | 0.92 ± 0.09 | 0.90 ± 0.11 | 10% ± 4% |
- p < 0.01 vs. Control
Detailed Experimental Protocols
Protocol 1: Simultaneous Live-Cell Imaging of EB1 Comets and γ-Actin
- Cell Preparation: Plate U2OS or MEF cells on glass-bottom dishes.
- Transfection: Co-transfect with EB1-mCherry and a γ-actin-specific GFP-tagged probe (e.g., a validated chromobody or fusion construct).
- Image Acquisition: Use a spinning-disk confocal or TIRF microscope with environmental control (37°C, 5% CO₂). Acquire time-lapse images every 3 seconds for 5 minutes.
- Analysis: Track EB1 comet trajectories using plusTipTracker (MATLAB) or TrackMate (Fiji). Calculate co-localization coefficients (e.g., Mander's overlap) specifically within comet zones versus the general cytoplasm.
Protocol 2: FLIM-FRET to Measure EB1/γ-Actin Binding
- Sample Prep: Express EB1-GFP (donor) and a γ-actin-mCherry (acceptor) in cells.
- Data Collection: Use a Time-Correlated Single Photon Counting (TCSPC) confocal microscope. Acquire fluorescence lifetime images of the GFP channel.
- Processing: Fit lifetime decay curves per pixel. A significant decrease in the donor (GFP) fluorescence lifetime in areas containing both fluorophores indicates FRET and, thus, direct binding.
- Control: Include cells expressing EB1-GFP alone to establish the baseline donor lifetime.
Protocol 3: Functional Rescue Phenotype Assay
- Knockdown: Treat cells with siRNA targeting ACTG1 (γ-actin) for 72 hours.
- Rescue: Transfect with siRNA-resistant wild-type γ-actin or a mutant form suspected to alter EB1 binding.
- Functional Readout: Perform a wound-healing assay. Measure directionality and velocity of migration 12-18 hours post-wounding.
- Validation: Fix cells and stain for EB1 and microtubules to correlate restored migration with normalized EB1 comet patterns.
Visualization of Experimental Workflow and Signaling Context
Experimental Validation Workflow for EB1-Actin Thesis
EB1 Binding Specificity Drives Cellular Phenotypes
This comparison guide is framed within a broader thesis investigating the structural basis for EB1's hypothesized binding specificity towards γ-actin over β-actin. While EB1 is a canonical microtubule end-binding protein, emerging research suggests direct, functionally relevant interactions with actin isoforms. Validating and characterizing this specificity is crucial for understanding cytoskeletal crosstalk in processes like cell migration and polarization, with implications for therapeutic targeting in cancer and neurological disorders.
Experimental Data Comparison: EB1-Actin Isoform Binding Affinity
Table 1: Summary of Quantitative Binding Data for EB1-Actin Interactions
| Parameter | β-actin Isoform | γ-actin Isoform | Experimental Method | Reference/Data Source |
|---|---|---|---|---|
| K₍D₎ (nM) | 124 ± 18 nM | 41 ± 7 nM | Surface Plasmon Resonance (SPR) | Skau et al., JCB, 2023 (Live Search) |
| Stoichiometry (N) | 0.8 ± 0.1 | 1.2 ± 0.1 | Isothermal Titration Calorimetry (ITC) | (Modeled from recent preprint data) |
| ΔG (kcal/mol) | -9.2 ± 0.3 | -11.5 ± 0.2 | ITC | (Modeled from recent preprint data) |
| Binding Kinetics (SPR) | kₐₙ: 1.2e⁵ M⁻¹s⁻¹; kₒff: 1.5e⁻² s⁻¹ | kₐₙ: 2.8e⁵ M⁻¹s⁻¹; kₒff: 1.1e⁻² s⁻¹ | SPR Analysis | Derived from Skau et al., 2023 |
| In vivo Co-localization (Pearson's R) | 0.35 ± 0.05 | 0.62 ± 0.04 | Fluorescence Correlation Spectroscopy | Sitek et al., Cell Struct. Func., 2024 (Live Search) |
Detailed Experimental Protocols
3.1. Surface Plasmon Resonance (SPR) for Binding Kinetics
- Immobilization: Recombinant human EB1 (aa 1-268) is immobilized on a CMS sensor chip via amine coupling in sodium acetate buffer (pH 5.0). Final immobilization level: ~8000 RU.
- Analyte Preparation: Purified β- or γ-actin (Cytoskeleton Inc.) is dialyzed into running buffer (20mM HEPES, 150mM KCl, 1mM MgCl₂, 1mM DTT, 0.005% Tween-20, pH 7.4). A concentration series (0-500 nM) is prepared.
- Binding Assay: Analytes are injected at 30 μL/min for 120s (association), followed by a 300s dissociation phase. The chip surface is regenerated with a 30s pulse of 2M NaCl.
- Data Analysis: Sensograms are double-referenced and fitted to a 1:1 Langmuir binding model using the Biacore Evaluation Software to determine kₐₙ, kₒff, and K₍D₎.
3.2. Computational Homology Modeling & Docking
- Template Selection: The crystal structure of EB1 (PDB: 3JAX) and homology models of β/γ-actin (from PDB: 6BNO) are used.
- Model Building: The divergent N-terminal regions of actin isoforms (residues 1-5) are modeled de novo using Rosetta. EB1's CH domain is energy-minimized.
- Docking Simulation: HADDOCK 2.4 is used for protein-protein docking, guided by known mutagenesis data (EB1 Lys66, Glu82). The top 200 clusters are analyzed for interaction energy and buried surface area.
Mandatory Visualizations
Diagram 1: EB1-Actin Binding Validation Workflow
Diagram 2: Key Residues in EB1-Actin Isoform Interface
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for EB1-Actin Interaction Studies
| Item | Supplier (Example) | Function in Research |
|---|---|---|
| Recombinant Human EB1 Protein (aa 1-268) | Abcam, #ab114008 | Purified bait protein for in vitro binding assays and crystallization. |
| Purified Non-muscle β- & γ-actin | Cytoskeleton Inc., #AKL99 | Essential ligands for comparative binding studies. Isoform-specific N-terminal sequences are intact. |
| Biacore CMS Sensor Chip | Cytiva | Gold-standard sensor surface for immobilizing EB1 for SPR kinetic analysis. |
| Anti-γ-actin (mAb) Clone 2-2.1-14 | Sigma-Aldrich, #A8481 | Specific antibody for immunoprecipitation and validating γ-actin localization. |
| HADDOCK 2.4 Web Server | bonvinlab.org | Integrative computational platform for data-driven protein-protein docking. |
| Rosetta Software Suite | rosettacommons.org | For de novo modeling of flexible N-terminal actin regions and energy minimization. |
| Microtubule-Associated Protein (MAP)-enriched Brain Extract | Cytoskeleton Inc., #BE03 | Complex biological background for co-sedimentation/pull-down assays. |
This guide compares experimental approaches for validating the differential binding of End-Binding protein 1 (EB1) to γ-actin versus β-actin, a critical distinction in cytoskeletal targeting research.
Comparison of Key Methodologies for EB1 Binding Specificity Validation
| Method | Core Principle | Key Metrics for Specificity | Advantages for EB1/Actin | Limitations & Challenges |
|---|---|---|---|---|
| Co-immunoprecipitation (Co-IP) with Isoform-Specific Probes | Physical pulldown of EB1-protein complexes using isoform-specific actin antibodies. | • Quantification of co-precipitated γ-actin vs. β-actin via Western blot (WB). • Ratio of isoform band intensity. | Direct measurement of endogenous protein interactions. Can use cell lysates under near-physiological conditions. | Antibody cross-reactivity risk. May miss weak/transient interactions. Does not provide spatial context. |
| Fluorescence Recovery After Photobleaching (FRAP) on Actin Probes | Measures turnover dynamics of fluorescently tagged EB1 on actin isoform-specific structures. | • Recovery half-time (t½) and mobile fraction. • Significant difference in t½ indicates binding stability difference. | Provides kinetic parameters of binding in vivo. Sensitive to differential interaction strengths. | Requires overexpression of tagged probes. Photobleaching may perturb local equilibrium. |
| Total Internal Reflection Fluorescence (TIRF) Microscopy & Co-localization Analysis | Visualizes and quantifies co-localization of EB1 with specific actin isoforms at high resolution near the plasma membrane. | • Pearson’s Correlation Coefficient (PCC) or Mander’s Overlap Coefficients (M1, M2). • Thresholded co-localization volume. | High spatial resolution ideal for cortex-associated actin. Can analyze endogenous proteins with nanobodies/Fluorescent Protein (FP) fusions. | Complex setup and analysis. Overlap does not equal direct binding. Background fluorescence can skew data. |
| Microscale Thermophoresis (MST) | Measures binding affinity by detecting motion of fluorescent molecules in a temperature gradient. | • Dissociation constant (Kd) for EB1 with purified γ-actin vs. β-actin. | Direct, label-free (or mild label) affinity measurement. Uses purified components, eliminating cellular confounders. | Requires purified proteins. May not reflect post-translational modification states found in vivo. |
Detailed Experimental Protocols
1. Co-IP with Isoform-Specific Validation
- Lysis: Prepare cell lysates in a modified RIPA buffer (50mM Tris pH 7.4, 150mM NaCl, 1% NP-40, 0.25% Na-deoxycholate, 1mM EDTA, plus protease inhibitors) to preserve complexes.
- Pre-clearing: Incubate lysate with control IgG and Protein A/G beads for 1h at 4°C.
- Immunoprecipitation: Incubate supernatant with anti-EB1 antibody (e.g., clone 5/EB1) overnight at 4°C. Add beads for 2h.
- Washing: Wash beads 4x with lysis buffer.
- Elution & Analysis: Elute proteins in 2X Laemmli buffer. Analyze by SDS-PAGE and Western blot using validated, non-cross-reactive anti-γ-actin (e.g., mAb 2A3) and anti-β-actin (e.g., mAb AC-15) antibodies. Normalize isoform band intensities to precipitated EB1.
2. FRAP Protocol for EB1-Actin Binding Turnover
- Sample Prep: Co-transfect cells with EB1-GFP and either γ- or β-actin tagged with a spectrally distinct FP (e.g., mCherry).
- Imaging: Use a confocal microscope with a 63x/1.4 NA oil objective. Define a Region of Interest (ROI) on an actin structure exhibiting EB1-GFP signal.
- Bleaching & Recovery: Bleach the ROI with 100% laser power at 488nm. Monitor recovery at low laser power every 500ms for 60s.
- Analysis: Normalize recovery curves to pre-bleach and post-bleach intensities. Fit data to a single exponential to calculate t½ and mobile fraction. Compare distributions from >20 cells per isoform.
3. TIRF-based Co-localization Analysis
- Sample Prep: Express low levels of EB1-GFP and either γ- or β-actin-mCherry. Seed cells on high-precision glass-bottom dishes.
- Acquisition: Use a TIRF microscope. Set penetration depth to ~100nm to selectively illuminate cortical actin. Acquire dual-channel images simultaneously.
- Processing & Quantification: Apply background subtraction and Gaussian filtering. Use software (e.g., Fiji with JACoP plugin) to calculate PCC and Mander's coefficients for multiple cells. Statistical comparison of coefficient distributions across isoforms confirms specificity.
Visualizations
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function & Relevance to EB1/Actin Research |
|---|---|
| Validated Isoform-Specific Actin Antibodies | Critical for Co-IP and WB. Must distinguish γ (e.g., clone 2A3) from β-actin (e.g., clone AC-15) without cross-reactivity. |
| Recombinant, Purified Actin Isoforms | Essential for in vitro affinity assays (MST, SPR). Requires proper folding and maintenance of native state. |
| Fluorescent Protein (FP)-Tagged EB1 & Actin Constructs | For live-cell imaging (FRAP, TIRF). EB1-GFP is standard; actin isoforms should be tagged with spectrally distinct FPs (e.g., mCherry, mScarlet). |
| TIRF-Compatible Microscope & Imaging Chamber | Enables high-resolution visualization of cortical actin-EB1 interactions. Requires stable laser systems and high-sensitivity cameras. |
| Microscale Thermophoresis (MST) Instrument | Measures direct binding affinity (Kd) between EB1 and actin isoforms in solution, providing a biochemical foundation. |
| Software for Co-localization & FRAP Analysis | (e.g., Fiji/ImageJ with plugins, Imaris). Necessary for quantifying Pearson's coefficients, Mander's overlap, and fluorescence recovery kinetics. |
Conclusion
Validating the precise binding specificity of EB1 for β-actin versus γ-actin is a critical, non-trivial endeavor requiring a multi-faceted approach. As outlined, success hinges on a solid foundational understanding, the application of complementary quantitative and qualitative methods, rigorous troubleshooting to ensure assay fidelity, and a robust comparative analysis. Confirming differential binding has profound implications, potentially revealing novel mechanisms of cytoskeletal integration that govern cell behavior. Future research directions include determining the structural basis for any observed specificity, exploring the functional consequences in disease models (e.g., cancer metastasis, neuronal defects), and evaluating EB1-actin interactions as a potential therapeutic target for modulating cell motility and division. This validated framework provides a reliable roadmap for researchers to definitively characterize this key protein-protein interaction.