Beyond Structure: How Actin Cytoskeleton Dynamics Orchestrate Immune Cell Signaling and Therapeutic Potential
This comprehensive review synthesizes current research on the multifaceted role of the actin cytoskeleton in immune cell signaling.
Beyond Structure: How Actin Cytoskeleton Dynamics Orchestrate Immune Cell Signaling and Therapeutic Potential
Abstract
This comprehensive review synthesizes current research on the multifaceted role of the actin cytoskeleton in immune cell signaling. Targeting researchers, scientists, and drug development professionals, it explores foundational principles, from actin's role as a mechanical and biochemical scaffold to its regulation of immune synapse formation and receptor clustering. We examine cutting-edge methodologies for imaging and manipulating actin dynamics in immune contexts, address common experimental challenges and optimization strategies, and critically compare models and validation techniques. The article concludes by highlighting translational opportunities for targeting actin regulators in immunotherapies and autoimmune diseases, providing a roadmap for future mechanistic and applied research.
The Molecular Choreography: Foundational Principles of Actin Remodeling in Immune Signal Initiation
Within the broader thesis on actin cytoskeleton dynamics in immune cell signaling research, actin's role is classically understood as a structural scaffold. However, contemporary research establishes it as a dynamic, information-rich signaling platform. It integrates mechanical and biochemical cues to direct immune cell functions like migration, phagocytosis, and T-cell activation. This whitepaper provides a technical guide to actin's signaling functions, current methodologies, and quantitative data.
Core Signaling Pathways and Molecular Mechanisms
Actin nucleation, polymerization, and architecture are regulated by signaling pathways, and in turn, actin directly influences signal transduction.
Nucleation Pathways and Their Regulators
Three major nucleators initiate actin polymerization, each activated by distinct signals.
| Nucleation Factor | Upstream Regulator (Signal) | Key Effector Complex/Protein | Primary Immune Function |
|---|---|---|---|
| Arp2/3 Complex | WASP/N-WASP (via Cdc42, PIP2) | Actin filament branch | Phagocytosis, podosome formation |
| WAVE Regulatory Complex (via Rac) | Actin filament branch | Lamellipodia, cell migration | |
| Formins (mDia1/2) | RhoA, RhoC (GTP-binding) | Linear, unbranched filaments | Immunological synapse, cytokineis |
| Profilin | PIP2, Poly-Proline binding | Actin monomer delivery | General polymerization, T-cell activation |
Figure 1: Key Signaling Pathways to Actin Nucleation in Immune Cells
Actin as a Regulator of Transcription (MRTF-SRF Pathway)
Actin dynamics directly control gene expression via the MRTF-SRF pathway, linking cytoskeletal state to transcriptional programs.
Figure 2: Actin-MRTF-SRF Transcriptional Regulation Pathway
Table 1: Kinetic Parameters of Actin Nucleators in Immune Cells
| Parameter | Arp2/3 Complex | Formin (mDia1) | Profilin-Actin | Measurement Method |
|---|---|---|---|---|
| Nucleation Rate | ~0.1 branches/min/Arp2/3 | ~1.5 filaments/min/formin | N/A (monomer delivery) | TIRF Microscopy, Pyrene Actin |
| Elongation Rate | Dependent on mother filament | 10-20 subunits/sec | N/A | TIRF Microscopy |
| Critical Conc. (Cc) | ~0.1 µM (at branch points) | ~0.1 µM (barbed end) | N/A | In vitro Reconstitution |
| Key Inhibitor | CK-666 (IC50 ~25 µM) | SMIFH2 (IC50 ~10 µM) | N/A | Biochemical Assay |
Table 2: Impact of Actin Perturbation on Immune Cell Functions
| Function | Perturbation (Drug/Target) | Measured Effect (% Reduction vs. Control) | Key Readout |
|---|---|---|---|
| T-cell Cytotoxicity | Latrunculin B (G-actin sequester) | 65-80% | Target cell lysis |
| Neutrophil Chemotaxis | CK-666 (Arp2/3 inhibitor) | ~70% | Directional persistence, speed |
| Macrophage Phagocytosis | Cytochalasin D (Capping) | ~90% (FcγR-mediated) | Phagocytic index |
| DC Antigen Uptake | Jasplakinolide (Stabilization) | 50-60% | Dextran-FITC internalization |
Experimental Protocols
Protocol 1: Visualizing Actin Dynamics in Live T-Cells via TIRF Microscopy
Objective: To quantify actin polymerization flux at the immunological synapse (IS). Key Reagents: Human Jurkat T-cells, Anti-CD3/CD28 coated coverslips, LifeAct-GFP or Actin-GFP plasmid, Imaging medium (RPMI without phenol red). Procedure:
- Transfection: Transfect Jurkat cells with LifeAct-GFP using nucleofection. Culture for 24h.
- Stimulatory Coverslip: Prepare #1.5 coverslip by coating with 5 µg/mL anti-CD3 and 2 µg/mL anti-CD28 for 2h at 37°C. Block with 1% BSA.
- Imaging Chamber: Assemble coverslip in a live-cell chamber. Add transfected cells in imaging medium.
- TIRF Microscopy: Use a 488nm laser on a TIRF system with a 100x/1.49 NA objective. Acquire images every 2-5 seconds for 5-10 minutes immediately upon cell contact.
- Analysis: Use FIJI/ImageJ with plugins (e.g., TIRF Analyzer) to measure fluorescence intensity over time in the contact zone. Calculate polymerization rates from fluorescence increase slopes, normalized to basal levels.
Protocol 2: Co-immunoprecipitation of Actin-Associated Signaling Complexes
Objective: To isolate and identify signaling proteins bound to actin filaments in activated macrophages. Key Reagents: RAW 264.7 macrophages, LPS (100 ng/mL), DNase I-agarose beads (binds G-actin), Phalloidin-agarose beads (binds F-actin), Lysis buffer (1% Triton X-100, 150 mM NaCl, 10 mM PIPES pH 6.8, with protease/phosphatase inhibitors). Procedure:
- Stimulation: Stimulate macrophages with LPS for 0-30 min. Wash with cold PBS.
- Differential Extraction: Lyse cells in Triton-based buffer (37°C, 5 min) to preserve F-actin. Centrifuge at 16,000xg for 10 min at 37°C. The supernatant contains G-actin and soluble proteins. The pellet contains Triton-insoluble F-actin and associated proteins.
- Affinity Pulldown:
- G-actin Complexes: Incubate supernatant with DNase I-agarose for 2h at 4°C.
- F-actin Complexes: Resuspend pellet in lysis buffer + 10 µM phalloidin. Sonicate briefly. Incubate with Phalloidin-agarose for 2h at 4°C.
- Wash & Elution: Wash beads 5x with lysis buffer. Elute bound proteins with SDS-PAGE sample buffer at 95°C.
- Analysis: Analyze by Western Blot for suspected interactors (e.g., WASp, ARPC2, mTOR) or by mass spectrometry for discovery.
Protocol 3: FRET-based Biosensor for Rho GTPase Activity at Actin Sites
Objective: To spatially map the activation of Rho GTPases (Rac1, Cdc42) relative to actin structures. Key Reagents: Raichu-Rac1 or Raichu-Cdc42 FRET biosensor plasmid, Neonatal Human Dermal Fibroblasts (NHDFs) or Dendritic Cells, Fibronectin-coated dishes. Procedure:
- Cell Preparation: Seed cells on fibronectin. Transfect with the FRET biosensor using appropriate transfection reagent (e.g., Lipofectamine 3000).
- FRET Imaging: After 24-48h, image live cells on a confocal microscope with capabilities for FRET (e.g., using CFP excitation at 433nm and simultaneous collection of CFP (475nm) and YFP (527nm) emission).
- Stimulation: Acquire baseline images, then stimulate with a chemoattractant (e.g., SDF-1α for DCs) and continue time-lapse imaging.
- Ratio Analysis: Calculate the FRET ratio (YFP emission / CFP emission) for each pixel over time using software (e.g., MetaMorph). Co-stain with SiR-actin or LifeAct-RFP to correlate high FRET ratio zones (high GTPase activity) with actin structures like lamellipodia.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Actin Signaling Research
| Item | Function / Target | Example Product/Catalog # | Key Application Notes |
|---|---|---|---|
| CK-666 | Allosteric inhibitor of Arp2/3 complex | Sigma-Aldrich, SML0006 | Use at 50-100 µM; control with inactive analog CK-689. |
| Latrunculin A/B | Sequesters G-actin, prevents polymerization | Cayman Chemical, 10010630 | Highly toxic; use low nM to µM range; effects rapid. |
| Jasplakinolide | Stabilizes F-actin, promotes polymerization | Tocris, 2792 | Can induce apoptosis with prolonged treatment. |
| LifeAct-TagGFP2 | Live-cell F-actin label, minimal perturbation | Ibidi, 60102 | Preferred over GFP-actin for reduced artifact. |
| Phalloidin (Alexa Fluor conjugates) | High-affinity stain for F-actin (fixed cells) | Thermo Fisher, various (e.g., A12379) | Gold standard for fixed-cell imaging; multiple colors. |
| Rhotekin-RBD Agarose | Pull-down assay for active, GTP-bound Rho | Cytoskeleton, Inc., RT02 | Specific for Rho (A,B,C); use with positive control GTPγS. |
| G-LISA RhoA Activation Assay | ELISA-based quantitation of active RhoA | Cytoskeleton, Inc., BK124 | Higher throughput than traditional pull-down + WB. |
| SiR-Actin / -Tubulin Kit | Far-red live-cell actin stain (cell-permeable) | Cytoskeleton, Inc., CY-SC001 | Low cytotoxicity, excellent for long-term imaging. |
| Recombinant Human WASP | Purified protein for in vitro actin assays | Novus Biologicals, H00007454-P01 | Critical for reconstitution of Arp2/3 branching. |
Moving beyond its canonical structural role, actin is a central processor of signaling information in immune cells. Its polymerization status directly feeds back on transcriptional programs and receptor signaling, creating dynamic feedback loops. Deciphering these mechanisms requires the integrated use of live-cell imaging, biochemical fractionation, and targeted pharmacological tools as outlined. This positions actin and its regulatory networks as promising, albeit complex, therapeutic targets for modulating immune responses in autoimmunity, cancer immunotherapy, and inflammation.
The actin cytoskeleton is a fundamental determinant of cellular architecture and mechanical behavior. In immune cells, its rapid, precise remodeling underpins critical functions: chemotaxis, phagocytosis, immunological synapse formation, and cytotoxic granule release. This dynamic restructuring is primarily orchestrated by two classes of nucleation factors: the Arp2/3 complex and formins. The Arp2/3 complex generates branched actin networks that produce protrusive forces, essential for lamellipodia and phagocytic cup formation. Formins generate linear, unbranched actin filaments, crucial for filopodia, contractile rings, and microtubule capture. Their activity is tightly regulated by convergent signaling inputs from cell surface receptors, including GPCRs, antigen receptors, and cytokine receptors. Dysregulation of these pathways contributes to immunodeficiencies, autoimmunity, and cancer metastasis. This whitepaper provides a technical guide to these key regulators, their signaling inputs, and methodologies for their study within immune cell research.
The Arp2/3 Complex: Architecture and Activation
The Arp2/3 complex is a stable assembly of seven subunits (Arp2, Arp3, ARPC1-5). In its inactive state, Arp2 and Arp3 are held apart. Activation requires simultaneous binding to a nucleation-promoting factor (NPF) and the side of a pre-existing ("mother") actin filament.
Key Signaling Inputs to Arp2/3 in Immune Cells:
- GPCR (e.g., Chemokine Receptors): Signals through Rac GTPases (Rac1, Rac2), which activate WAVE Regulatory Complex (WRC). WAVE is a major NPF that relieves autoinhibition and presents the WCA domain (WH2-Central-Acidic) to Arp2/3.
- Immunoreceptors (TCR, BCR, FcRs): Engage multiple pathways. One major route involves activation of N-WASP via Cdc42 and phosphorylation by kinases like Nck and Src-family kinases (e.g., Lck). N-WASP is another critical NPF.
- Integrins: During adhesion, integrin engagement activates Src and FAK, leading to Rac and WASP/WAVE activation.
- Pathogen Sensors: NOD-like receptors can nucleate actin around invading bacteria via N-WASP.
Diagram 1: Arp2/3 Activation Pathways in Immune Cells
Formins: Processive Elongation and Regulation
Formins are a large family of multi-domain proteins characterized by formin homology 1 and 2 (FH1, FH2) domains. The FH2 dimer processively caps the barbed end, enabling rapid filament elongation while protecting from capping protein. The FH1 domain recruits profilin-actin for delivery. Auto-inhibition is mediated by intra-molecular binding between the diaphanous inhibitory domain (DID) and diaphanous autoregulatory domain (DAD).
Key Signaling Inputs to Formins in Immune Cells:
- Rho GTPases: Primary activators. RhoA activates mDia1/2; Rac activates mDia2 and FMNL isoforms; Cdc42 activates mDia2 and FMNL1. GTPase binding to the N-terminal GTPase-binding domain (GBD) relieves autoinhibition.
- Immunoreceptor Signaling: TCR engagement activates both RhoA and Rac, leading to mDia1 and FMNL1 activation, critical for T cell polarization and synapse stability.
- Cytokine Receptors (e.g., IL-2R): Signal through Rho GTPases to modulate formin activity during cell cycle progression and cytotoxicity.
Diagram 2: Formin Activation and Elongation Mechanism
Comparative Quantitative Analysis
Table 1: Key Characteristics of Arp2/3 Complex and Formins
| Feature | Arp2/3 Complex | Formins (e.g., mDia1, FMNL1) |
|---|---|---|
| Core Activity | Nucleates branched networks, caps pointed ends. | Nucleates linear filaments, processively caps barbed ends. |
| Nucleation Rate | ~0.1 filaments/complex/min (slow initiator). | Highly variable; up to ~1.5 μm/min elongation per filament. |
| Structural Trigger | Binding to NPFs (WCA domain) and mother filament. | Relief of autoinhibition via Rho GTPase binding. |
| Primary Immune Signal Inputs | GPCR→Rac→WAVE; TCR→Cdc42/Nck→N-WASP. | GPCR/TCR→RhoA/Rac→mDia/FMNL. |
| Key Inhibitors | CK-666 (prevents active conformation), CK-869 (binds Arp3). | SMIFH2 (targets FH2 domain), small-molecule Rho inhibitors. |
| Dominant Cellular Structures | Lamellipodia, phagocytic cups, dendritic spines. | Filopodia, stress fibers, contractile rings, microtubule tracks. |
| Typical Filament Lifetime | Shorter, highly dynamic due to capping protein. | Longer, stabilized by processive capping. |
Table 2: Exemplary Signaling Inputs and Functional Outcomes in Immune Cells
| Immune Process | Primary Receptor | Key Nucleator | Major Upstream Signal | Cytoskeletal Output | Functional Outcome |
|---|---|---|---|---|---|
| Chemotaxis | Chemokine GPCR (e.g., CXCR4) | Arp2/3 (via WAVE) | Rac activation | Leading-edge lamellipodia | Directional migration |
| Phagocytosis | Fcγ Receptor | Arp2/3 (via N-WASP) | Cdc42 & PI(4,5)P2 | Phagocytic cup | Pathogen engulfment |
| Immunological Synapse | T Cell Receptor | Formin (mDia1, FMNL1) | RhoA & Rac activation | Radial F-actin array | TCR clustering, signaling |
| NK Cell Cytotoxicity | Activating Receptors (e.g., NKG2D) | Formin (mDia1) & Arp2/3 | Rho & Cdc42 | Synaptic F-actin mesh | Cytolytic granule convergence |
Essential Experimental Protocols
Protocol 1: In Vitro Pyrene-Actin Polymerization Assay
Purpose: Quantify nucleation activity of purified Arp2/3 or formins with/without activators.
Key Reagents & Workflow:
- Materials: Purified actin (10% pyrene-labeled), nucleation factor (Arp2/3 + NPF, or formin), activator (GTPase•GTPγS, PIP2 liposomes), polymerization buffer (1 mM MgCl2, 50 mM KCl, 1 mM EGTA, 10 mM imidazole pH 7.0).
- Procedure:
- Prepare actin in G-buffer (low salt) on ice.
- In a 96-well plate, mix nucleation factor ± activator in polymerization buffer.
- Initiate polymerization by adding MgCl2/KCl and actin to final 2-4 μM.
- Immediately measure pyrene fluorescence (ex 365 nm, em 407 nm) every 5-10 sec in a plate reader at 25°C.
- Plot fluorescence vs. time. Nucleation activity is derived from the time to half-maximal polymerization or initial slope.
Diagram 3: Pyrene-Actin Assay Workflow
Protocol 2: Immunofluorescence-Based Quantification of Cortical F-actin in T Cells
Purpose: Assess the role of specific nucleation factors upon TCR stimulation using pharmacological inhibition.
Key Reagents & Workflow:
- Materials: Primary T cells, anti-CD3/CD28 coated coverslips, inhibitors (CK-666 for Arp2/3, SMIFH2 for formins), fixation/permeabilization buffer, phalloidin-Alexa Fluor 488, confocal microscope.
- Procedure:
- Pre-treat cells with DMSO, CK-666 (100 μM), or SMIFH2 (25 μM) for 30 min.
- Seed onto stimulatory coverslips for desired time (e.g., 2-5 min for early actin burst).
- Fix (4% PFA), permeabilize (0.1% Triton X-100), and stain with phalloidin.
- Acquire z-stacks at the cell-coverslip interface using consistent settings.
- Quantify mean phalloidin fluorescence intensity at the cortical region (peripheral ~1 μm) using ImageJ.
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Studying Actin Nucleation in Immune Signaling
| Reagent | Target/Function | Example Use-Case | Key Consideration |
|---|---|---|---|
| CK-666 | Arp2/3 complex inhibitor (allosteric). | Inhibiting branched network formation in chemotaxis. | Reversible; control with inactive analog CK-689. |
| SMIFH2 | Formin FH2 domain inhibitor. | Assessing formin contribution to filopodia. | Can have off-target effects at high浓度; use siRNA rescue. |
| Recombinant Rho GTPases (Constitutively Active/Dominant Negative) | To modulate upstream signaling. | Directly activating formins or WRC in reconstitution assays. | Always load with GTPγS (active) or GDP (inactive). |
| siRNA/shRNA Libraries | Knockdown of specific NPFs or formins. | Determining isoform-specific roles in immune cells. | Transfection efficiency in primary immune cells can be low. |
| Latrunculin A/B | Binds G-actin, prevents polymerization. | Negative control for actin-dependent processes. | Highly toxic; effects are reversible upon washout. |
| PIP2 (PI(4,5)P2) Liposomes | Membrane lipid signaling component. | Activating N-WASP in in vitro motility assays. | Must prepare fresh and characterize size. |
| Profilin-1 Purified Protein | Enhances formin-mediated elongation. | In vitro pyrene assays with mDia1. | Critical for efficient actin delivery to FH1 domain. |
| Anti-phospho-WASP/WAVE Antibodies | Detect activation status of NPFs. | Monitoring N-WASP activation by flow cytometry after TCR crosslinking. | Requires careful fixation/permeabilization protocols. |
Within the broader thesis on actin cytoskeleton dynamics in immune cell signaling research, the immune synapse (IS) stands as a paramount example of spatial organization. This intricate, transient structure forms at the interface between an antigen-presenting cell (APC) and a lymphocyte, orchestrating the specific, directed secretion of effector molecules and the integration of signaling cascades. Its precise assembly, stability, and function are fundamentally driven by the dynamic remodeling of the actin cytoskeleton. This whitepaper provides an in-depth technical guide to the core principles, experimental dissection, and quantitative analysis of actin's role in immune synapse biology.
Core Principles of Actin Dynamics at the Synapse
The IS is characterized by a concentric bullseye pattern: a central supramolecular activation cluster (cSMAC) surrounded by a peripheral pSMAC, all encircled by a distal SMAC (dSMAC). Actin dynamics are spatially segregated: Arp2/3 complex-driven branched actin polymerization generates a outward-moving, contractile network in the dSMAC, facilitating TCR microcluster centripetal transport. Meanwhile, formin (e.g., mDia1)-driven linear actin filaments stabilize the central region. Myosin II motor activity provides the contractile force. This spatial choreography ensures sustained signaling and polarized secretion.
Table 1: Key Actin Regulators at the Immune Synapse
| Protein/Complex | Primary Function | Spatial Localization | Perturbation Effect |
|---|---|---|---|
| WASP/N-WASP | Activates Arp2/3 complex for branched actin nucleation. | dSMAC, sites of TCR microclusters. | Inhibits TCR cluster centripetal flow, reduces synapse stability. |
| Arp2/3 Complex | Nucleates branched actin networks. | dSMAC, lamellipodial region. | Disrupts peripheral actin cloud, abrogates central clearing. |
| mDia1 (Formin) | Nucleates linear, unbranched actin filaments. | cSMAC/pSMAC interface. | Impairs central stability and microtubule organizing center (MTOC) polarization. |
| Myosin IIA | ATP-dependent motor; provides contractility. | pSMAC/dSMAC interface. | Blocks TCR microcluster movement to center, alters synapse symmetry. |
| Cofilin | Severs and depolymerizes actin filaments. | Throughout, active at disassembling regions. | Leads to excessive actin accumulation, disrupts turnover and flow. |
| HS1 | Stabilizes branched actin networks, recruits signaling proteins. | dSMAC, associated with WASP. | Reduces actin density and stability at synapse periphery. |
Experimental Protocols for Investigating Actin at the IS
Protocol 1: High-Resolution Live-Cell Imaging of Actin Dynamics
Objective: To visualize and quantify F-actin flow and turnover in real-time during immune synapse formation.
- Cell Preparation: Isolate primary human T-cells or use a suitable T-cell line (e.g., Jurkat). Transfect with a fluorescent actin marker (e.g., LifeAct-GFP, expressed via nucleofection) or label F-actin with a cell-permeable dye (e.g., SiR-actin).
- Synapse Formation: Prepare stimulatory surfaces. Coat #1.5 glass-bottom dishes with anti-CD3ε (5 µg/mL) and anti-CD28 (2 µg/mL) antibodies in PBS for 2 hours at 37°C. Block with 1% BSA. Alternatively, use supported lipid bilayers (SLBs) containing GPI-linked ICAM-1 and fluorescently labeled antigenic pMHC.
- Image Acquisition: Seed cells onto the stimulatory surface and immediately image on a spinning-disk confocal or TIRF microscope equipped with a 37°C/5% CO₂ environmental chamber. Acquire time-lapse images (1-5 second intervals) for 10-30 minutes using a 100x oil immersion objective.
- Analysis: Use particle image velocimetry (PIV) or kymograph analysis along a line scan from the synapse periphery to the center to quantify actin flow velocity (typically 0.05 - 0.2 µm/s). Measure fluorescence recovery after photobleaching (FRAP) in a defined synaptic region to calculate actin turnover half-time.
Protocol 2: Spatial Mapping of Actin Regulator Localization
Objective: To correlate the nanoscale localization of actin-binding proteins with F-actin architecture.
- Sample Fixation: Form synapses as in Protocol 1. At desired time points (e.g., 5 min for early, 20 min for mature synapse), fix cells with 4% paraformaldehyde (w/v) in cytoskeleton buffer (CB: 10 mM MES, 138 mM KCl, 3 mM MgCl₂, 2 mM EGTA, pH 6.9) for 15 minutes to preserve actin structures.
- Immunostaining: Permeabilize with 0.1% Triton X-100 in CB for 5 min. Block with 5% normal goat serum. Incubate with primary antibodies (e.g., anti-WASP, anti-p34-Arc, anti-HS1) overnight at 4°C. Use species-specific secondary antibodies conjugated to spectrally distinct fluorophores.
- Super-Resolution Imaging: Acquire images using 3D-SIM (Structured Illumination Microscopy) or STORM (Stochastic Optical Reconstruction Microscopy). For STORM, use Alexa Fluor 647-conjugated secondary antibodies and imaging buffer containing 100 mM cysteamine, 5% glucose, glucose oxidase, and catalase.
- Colocalization Analysis: Calculate Pearson's correlation coefficient or Manders' overlap coefficients between the actin channel and each regulator channel within defined synaptic subregions using software like Fiji/ImageJ with the JACoP plugin.
Protocol 3: Functional Perturbation via CRISPR-Cas9 or Pharmacological Inhibition
Objective: To determine the functional requirement of specific actin regulators in synapse formation.
- Genetic Knockout: Generate knockout T-cell lines using CRISPR-Cas9. Design sgRNAs targeting human WAS (WASP), WASL (N-WASP), or DIAPH1 (mDia1). Transfect via nucleofection, single-cell clone, and validate knockout by western blot and sequencing.
- Pharmacological Inhibition: Use small molecule inhibitors: CK-666 (100 µM) to inhibit Arp2/3 complex; SMIFH2 (25 µM) to inhibit formins; Blebbistatin (50 µM) to inhibit Myosin II.
- Functional Readouts:
- Calcium Flux: Load control and perturbed cells with Fluo-4 AM dye. Flow onto anti-CD3 coated plates and monitor intracellular Ca²⁺ flux via fluorescence increase over time using a plate reader or imager.
- Cytolytic Granule Polarization: Co-culture cytotoxic T lymphocytes (CTLs) with target cells. Fix, stain for perforin/granzyme B and actin. Score percentage of cells with granules polarized to the contact site.
- Signaling Microcluster Tracking: Image synapses on SLBs. Track movement of fluorescently tagged TCRζ or ZAP-70 microclusters via single-particle tracking algorithms to determine velocity and directionality.
Signaling Pathway Visualization
(Diagram Title: Actin Signaling Pathways in Immune Synapse Assembly)
Table 2: Measured Parameters of Actin Dynamics at the T-cell Immune Synapse
| Parameter | Typical Value (Range) | Measurement Technique | Biological Significance |
|---|---|---|---|
| Actin Retrograde Flow Velocity | 0.08 - 0.15 µm/s | PIV analysis of LifeAct movies. | Drives centripetal transport of TCR microclusters. |
| F-actin Half-life (Turnover) | 30 - 90 seconds | FRAP of actin-GFP in synaptic region. | Indicates dynamic remodeling essential for adaptability. |
| TCR Microcluster Speed | 0.05 - 0.12 µm/s | Single-particle tracking of TCRζ-GFP. | Coupled to, but slower than, actin flow. |
| Synaptic F-actin Density (Peak) | 2-3 fold over cytoplasmic baseline | Integrated fluorescence intensity in TIRF. | Reflects magnitude of cytoskeletal reorganization. |
| Time to Central Clearing (Actin Depletion) | 3 - 8 minutes post-contact | Time-lapse microscopy. | Marks transition to mature synapse architecture. |
| MTOC Polarization Latency | 5 - 15 minutes | Imaging of microtubule markers (e.g., EB1). | Dependent on central actin stabilization. |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Immune Synapse Actin Research
| Reagent Name | Supplier Examples (Catalog #) | Function/Application |
|---|---|---|
| SiR-Actin Kit | Cytoskeleton, Inc. (CY-SC001) | Live-cell, far-red fluorescent F-actin staining with low cytotoxicity. |
| Anti-CD3ε (OKT3) & Anti-CD28 | BioLegend, Tonbo Biosciences | Coating antibodies for planar stimulatory surfaces to trigger synapse formation. |
| CK-666 | Sigma-Aldrich (SML0006), Tocris (3950) | Cell-permeable, selective inhibitor of the Arp2/3 complex. |
| SMIFH2 | Sigma-Aldrich (S4826) | Small molecule inhibitor of formin homology 2 (FH2) domain activity. |
| (-)-Blebbistatin | Cayman Chemical (13013) | Selective, reversible inhibitor of non-muscle myosin II ATPase. |
| LifeAct-TagGFP2 Plasmid | ibidi (60101) | Fluorescent probe for labeling F-actin structures in live cells via transfection. |
| Dylight 650 NHS Ester | Thermo Fisher Scientific | For custom fluorescent labeling of proteins (e.g., pMHC) for SLB incorporation. |
| CellTrace Violet / CFSE | Thermo Fisher Scientific | Cell proliferation dyes useful for distinguishing immune cells in co-culture assays. |
| Supported Lipid Bilayer (SLB) Kit | Nanion Technologies | Pre-formulated vesicles for creating fluid bilayers containing adhesion and antigen molecules. |
| CRISPR-Cas9 T-cell Nucleofector Kit | Lonza (VPA-1002) | System for efficient delivery of CRISPR ribonucleoproteins into primary human T-cells. |
The immune synapse epitomizes the sophisticated spatial control exerted by the actin cytoskeleton in immune cell function. Its study requires a multidisciplinary approach combining high-resolution live imaging, spatial proteomics, and precise genetic and pharmacological perturbations. Understanding this actin-driven organization is not merely an academic pursuit; it provides a critical framework for developing novel immunotherapies that modulate immune cell communication, such as enhancing cytotoxic T-cell function in solid tumors or dampening aberrant signaling in autoimmunity. Future research, guided by the quantitative and methodological principles outlined here, will continue to decode this masterclass in cellular organization.
Thesis Context: This whitepaper details the proximal signaling events linking surface receptor stimulation to actin cytoskeleton remodeling, a critical process underpinning immune cell functions such as migration, phagocytosis, and immune synapse formation. Understanding these cascades is fundamental to manipulating immune responses in therapeutic contexts.
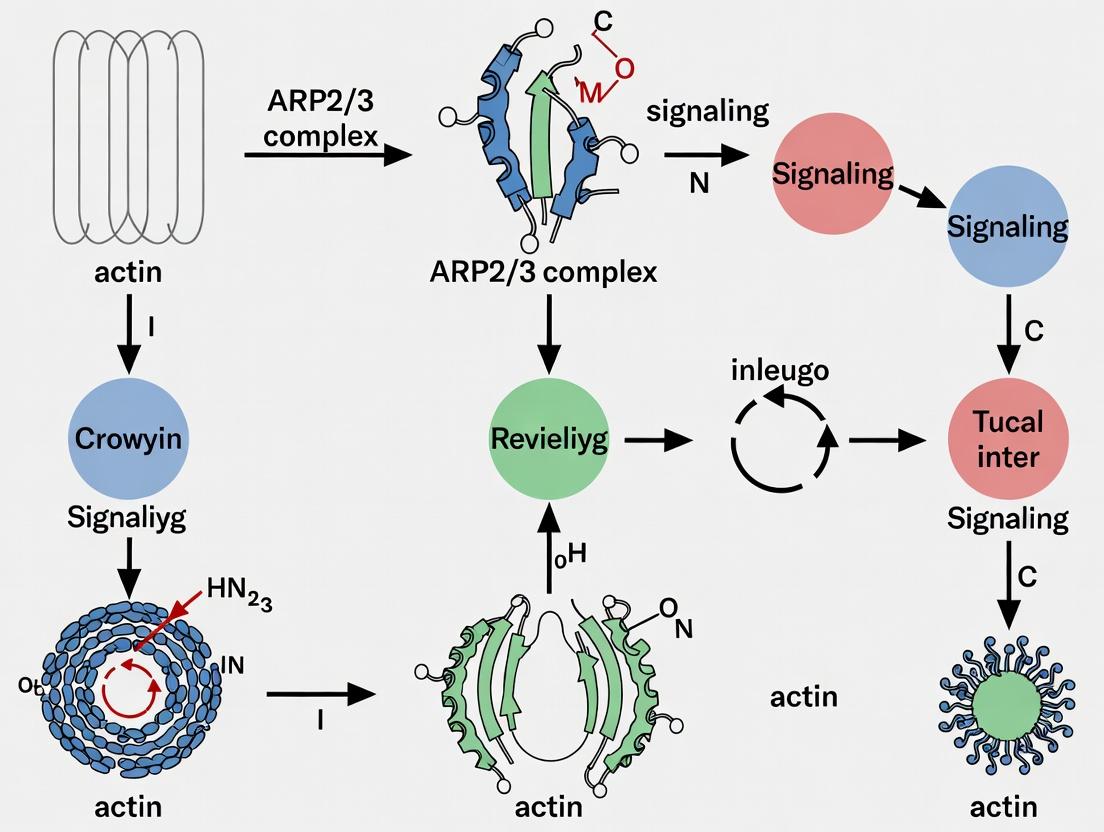
The nucleation of new actin filaments via the Arp2/3 complex is the central event in receptor-driven cytoskeletal rearrangement. This process is tightly regulated by the Wiskott-Aldrich Syndrome protein (WASp) family, their binding partners like WIP, and the lipid second messenger phosphatidylinositol 4,5-bisphosphate (PIP₂).
- WASp/N-WASp: Auto-inhibited nucleation-promoting factors (NPFs). Activation relieves the intramolecular interaction, exposing the VCA domain (Verprolin homology, Cofilin homology, Acidic region) that binds both Arp2/3 complex and G-actin.
- WIP (WASp-Interacting Protein): Stabilizes WASp, protects it from degradation, and participates in its activation and translocation. It is also a direct effector of actin dynamics.
- PIP₂: A membrane phospholipid that serves as both a docking site and a direct allosteric activator of numerous actin regulators, including WASp, N-WASp, and profilin.
Key Signaling Pathway: TCR/CD28 to Actin Polymerization
A canonical pathway in T cell activation illustrates the cascade from receptor engagement to actin rearrangement.
Quantitative Data on Molecular Interactions & Dynamics
Table 1: Key Biophysical Parameters in Early Actin Signaling
| Molecule/Interaction | Measured Parameter | Typical Value (Range) | Experimental Method | Significance |
|---|---|---|---|---|
| PIP₂ (in plasma membrane) | Concentration | ~1 mol% of inner leaflet lipids | Mass spectrometry, Lipid binding probes | Threshold for effector protein recruitment. |
| WASp Auto-inhibition | Kd (Intramolecular) | ~0.1 - 1 µM | ITC, FRET, NMR | Indicates stability of inactive state. |
| Cdc42-GTP to WASp | Kd (Binding) | ~50 - 100 nM | SPR, Fluorescence Polarization | Strength of primary activating signal. |
| PIP₂ to WASp (Basic Region) | Kd (Binding) | ~5 - 20 µM | Lipid co-sedimentation, SPR | Membrane tethering and co-activation. |
| Active WASp VCA to Arp2/3 | Kd (Binding) | ~0.1 - 5 µM | Pyrene Actin Assembly, SPR | Efficiency of complex nucleation. |
| Actin Branching Rate (with WASp/Arp2/3) | Branch formation | ~0.1 - 0.3 branches/µm/sec | TIRF Microscopy | Kinetics of network expansion. |
Detailed Experimental Protocols
Protocol: Assessing WASp Activation by Co-sedimentation
Objective: To evaluate the release of WASp auto-inhibition in vitro upon addition of activators (Cdc42-GTP and PIP₂-containing vesicles).
Key Reagents:
- Purified full-length N-WASp protein.
- Recombinant GTP-loaded Cdc42 (use GTPγS for non-hydrolyzable form).
- Synthetic liposomes containing 5% PIP₂ in a PC/PS background.
- Ultracentrifuge with TLA-100 rotor.
- SDS-PAGE and Coomassie staining/Western blot apparatus.
Procedure:
- Incubation: Combine 1 µM N-WASp with/without 5 µM Cdc42-GTPγS and/or 100 µM PIP₂ liposomes in a 100 µL reaction buffer (20 mM HEPES pH 7.4, 100 mM NaCl, 1 mM MgCl₂, 1 mM DTT).
- Sedimentation: Incubate 30 min at 25°C. Centrifuge at 100,000 x g for 30 min at 4°C to pellet liposomes and any bound protein.
- Analysis: Carefully separate supernatant (unbound) and pellet fractions. Resuspend pellet in equal volume of buffer. Load supernatant and pellet samples on SDS-PAGE.
- Interpretation: Auto-inhibited WASp remains in supernatant. Successful activation by Cdc42/PIP₂ leads to membrane binding and appearance in pellet fraction.
Protocol: Visualizing Actin Nucleation via TIRF Microscopy
Objective: To directly observe the kinetics of actin branch formation by the WASp/Arp2/3 complex.
Key Reagents:
- Purified actin (10% labeled with Alexa Fluor 488 or 647).
- Purified Arp2/3 complex, N-WASp VCA domain (or active full-length + activators).
- Profilin, CP (Capping Protein).
- Flow chamber passivated with PEG-biotin/NeutrAvidin.
- TIRF microscope with temperature control and sensitive EMCCD/sCMOS camera.
Procedure:
- Chamber Preparation: Create a biotinylated surface in a flow chamber. Introduce NeutrAvidin, followed by biotinylated poly-L-lysine or N-WASp to tether nucleation points.
- Reaction Mix: Prepare G-actin (2 µM, 10% labeled) in TIRF buffer (10 mM Imidazole pH 7.4, 50 mM KCl, 1 mM MgCl₂, 1 mM EGTA, 0.2 mM ATP, 50 mM DTT, 0.5% methylcellulose) with 50 nM Arp2/3, 100 nM profilin, 50 nM CP.
- Initiation & Imaging: Flow reaction mix into chamber. Immediately place on TIRF microscope. Acquire images at 1-5 second intervals for 10-20 minutes using appropriate laser lines.
- Analysis: Use tracking software (e.g., FIJI/ImageJ plugins) to quantify filament elongation rates and the appearance of new branches (Y-junctions).
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Studying Early Actin Signaling Cascades
| Reagent Category | Specific Example(s) | Function & Application |
|---|---|---|
| Lipid Probes/Resins | PIP₂ DiC₈ (soluble), PIP₂-coated beads, PI(4,5)P₂ Agarose beads | To mimic membrane recruitment, perform pull-down assays, or directly activate PIP₂-binding proteins in vitro. |
| GTPase Tools | Recombinant Cdc42/Rac1 (wild-type, constitutively active Q61L, dominant negative T17N), GEF/GAP assays kits. | To precisely manipulate and monitor the activity of small GTPases upstream of WASp/N-WASp. |
| Actin Polymerization Kits | Pyrene-actin-based fluorescence kits, Actin Binding Protein Spin-Down kits. | Standardized, quantitative measurement of bulk actin assembly kinetics and effector protein activity. |
| Biosensors | FRET biosensors for Cdc42/Rac1 activity (e.g., Raichu), PIP₂ (e.g., PH-PLCδ-GFP), Lifeact-GFP for F-actin. | Live-cell, spatiotemporal visualization of signaling molecule activity and cytoskeletal dynamics. |
| Inhibitors/Agonists | wiskostatin (WASp inhibitor), CK-666/ CK-689 (Arp2/3 inhibitors), SCV-07 (WIP inhibitor). | Pharmacological disruption of specific nodes in the pathway to establish causal relationships. |
| Antibodies | Phospho-specific antibodies (e.g., pY291-WASp), antibodies for N-WASp, WIP, Arp2/3 subunits. | Detection of protein expression, localization, and activation states via WB, IF, IP. |
| Recombinant Proteins | Purified full-length & truncation mutants of WASp/N-WASp, WIP, Arp2/3 complex. | For structural studies, in vitro reconstitution assays, and defining direct molecular interactions. |
Within the broader thesis on actin cytoskeleton dynamics in immune cell signaling, this whitepaper details the molecular mechanisms by which the actin cytoskeleton senses, transduces, and responds to mechanical forces. In immune cells, this process is critical for functions such as migration through tissues, antigen recognition, and synapse formation. This guide provides a technical dissection of the core mechanotransductive machinery.
The actin cytoskeleton is not merely a structural scaffold; it is a dynamic, force-sensitive network. In immune cells, external physical cues—such as substrate stiffness, shear flow, or the rigidity of a target cell—are converted into biochemical signals through actin's remodeling. This mechanotransduction directs pivotal processes like T-cell activation, phagocytosis, and chemotaxis.
Core Molecular Mechanisms
Force Sensing via Actin-Binding Proteins
Key proteins that tether actin to the membrane and to other structures undergo conformational changes under force, exposing cryptic binding sites or altering enzymatic activity.
- Vinculin & Talin: At integrin-mediated adhesion sites (focal adhesions in migrating cells or the immunological synapse), talin unfolds under tension, revealing vinculin-binding sites. Vinculin recruitment stabilizes the link to actin, reinforcing the adhesion.
- α-Actinin & Filamin: These actin cross-linkers sense and respond to cytoskeletal tension by altering their bundling or scaffolding properties, often recruiting signaling effectors.
- ERM Proteins (Ezrin/Radixin/Moesin): Link the cortical actin mesh to the plasma membrane. Phosphorylation and activation of ERM proteins are mechanosensitive, regulating cell cortex tension and membrane trafficking.
Force Generation and Feedback via Myosin II
Non-muscle myosin II (NMII) is the primary motor generating contractile force on actin networks. Its activity is regulated by phosphorylation of its regulatory light chain (RLC). Crucially, actin polymerization itself can stimulate myosin contractility, creating a positive feedback loop essential for sustained migration and immune synapse maturation.
Signaling Outputs: From Mechanics to Biochemistry
The mechanical perturbations of the actin network modulate several key signaling pathways:
- YAP/TAZ Signaling: In response to high cytoskeletal tension and a stiff microenvironment, F-actin polymerization inhibits the LATS1/2 kinase, preventing YAP/TAZ phosphorylation and degradation. Nuclear YAP/TAZ act as transcriptional co-activators for genes promoting proliferation and migration.
- SRF Signaling: G-actin monomers sequester the transcription factor co-activator MAL (MRTF-A). Force-induced actin polymerization depletes the G-actin pool, releasing MAL to translocate to the nucleus and activate Serum Response Factor (SRF)-driven transcription.
- Local Biochemical Cascades: Mechanical reinforcement of adhesion sites leads to the sustained activation of Src-family kinases, FAK, and Rho-family GTPases (e.g., RhoA, Rac1), creating spatially confined signaling hubs.
Table 1: Key Actin-Based Mechanosensors and Their Functions in Immune Cells
| Protein | Structural Role | Mechanosensitive Action | Immune Cell Context |
|---|---|---|---|
| Talin | Links integrins to F-actin | Unfolds under tension, exposing vinculin sites | T-cell synapse stability, leukocyte adhesion |
| Vinculin | Actin-membrane adapter | Binds unfolded talin, reinforces adhesion | Strengthens adhesion during extravasation |
| α-Actinin | Actin bundler | Alters bundling efficiency under load | Organizes actin at the phagocytic cup |
| Non-Muscle Myosin II (NMII) | Actin contractile motor | RLC phosphorylation regulated by tension | Cytotoxic T-cell killing, synapse contraction |
| Ezrin | Cortex-membrane linker | Phosphorylation/activation by tension | Regulates uropod retraction in migration |
Detailed Experimental Protocols
Protocol: Traction Force Microscopy (TFM) for Immune Cell Contractility
Objective: Quantify the forces exerted by an immune cell (e.g., a dendritic cell or T-cell) on its deformable substrate.
- Substrate Preparation: Fabricate polyacrylamide (PA) gels (~1-10 kPa stiffness) with embedded fluorescent microbeads (0.2 µm diameter). Coat the gel surface with ECM protein (e.g., ICAM-1, fibronectin).
- Cell Plating: Plate cells of interest onto the functionalized PA gel and allow to adhere (15-30 min).
- Imaging: Acquire high-resolution confocal z-stacks of the fluorescent beads with the cell present.
- Detachment: Gently remove the cell using trypsin-EDTA or a detergent lysis buffer.
- Reference Image: Acquire a second image of the beads in their relaxed, non-displaced state.
- Analysis: Use particle image velocimetry (PIV) algorithms (e.g., in MATLAB or open-source PIV software) to calculate the displacement field of beads between the two images. Input the gel's known elastic modulus into a Fourier transform traction cytometry (FTTC) algorithm to convert displacements into traction force vectors and maps.
Protocol: FRET-Based Tension Sensors in Live Cells
Objective: Visualize molecular-scale tension across a specific protein (e.g., vinculin) in real-time.
- Sensor Design: Transfect cells with a construct encoding the protein of interest (e.g., vinculin) harboring a tension sensor module (TSMod) inserted in a force-sensitive domain. TSMod consists of a FRET pair (e.g., mTFP1 and Venus) connected by an elastic spider silk peptide.
- Live-Cell Imaging: Use a confocal or TIRF microscope equipped with FRET capability. Image donor (mTFP1, Ex: 458nm) and acceptor (Venus, Ex: 514nm) emission channels.
- Force Calibration: Calculate the FRET ratio (Acceptor emission / Donor emission) for each pixel. A high FRET ratio indicates low tension (sensor compressed); a low FRET ratio indicates high tension (sensor extended, separating FRET pair).
- Application: Stimulate cells mechanically (e.g., shear flow) or biochemically (e.g., chemokine) and observe spatiotemporal changes in molecular tension at adhesions or the cell cortex.
Table 2: Quantitative Data from Key Mechanotransduction Studies
| Parameter Measured | Experimental System | Typical Value Range | Key Implication |
|---|---|---|---|
| Traction Force | T-cell on APL-coated gel | 1 - 50 nN/µm² | Force correlates with T-cell receptor signaling strength |
| Substrate Stiffness | Macrophage differentiation | 0.1 kPa (soft) to 100 kPa (stiff) | Stiff substrates promote M1 (inflammatory) phenotype |
| Molecular Tension (via TSMod) | Vinculin in migrating neutrophil | 2 - 6 pN | Peak tension at trailing edge uropod during retraction |
| Actin Polymerization Rate | Dendritic cell leading edge | ~100 subunits/µm/s | Increased by chemotactic gradient and substrate rigidity |
| Myosin II Contractility (pRLC) | NK cell during killing | 2-3 fold increase over basal | Required for perforin/granzyme delivery into target cell |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function/Application |
|---|---|
| Polyacrylamide Hydrogel Kits (e.g., BioPAE, CytoSoft) | Tunable substrates for traction force microscopy and stiffness sensing experiments. |
| Fluorescent Beads (Carboxylate-modified, 0.2µm) | Fiducial markers for quantifying substrate deformation in TFM. |
| FRET-based Tension Sensor (TSMod) Plasmids | Genetically encoded reporters for molecular tension (e.g., for vinculin, talin, spectrin). |
| Rho GTPase Activity Assays (G-LISA, FRET Biosensors) | Quantify activation of RhoA, Rac1, Cdc42 in response to mechanical stimuli. |
| Inhibitors: Y-27632 (ROCK), Blebbistatin (Myosin II), Latrunculin A (Actin) | Pharmacological tools to dissect the contribution of specific cytoskeletal components. |
| Atomic Force Microscopy (AFM) Cantilevers | For applying precise point forces or measuring cortical stiffness of single cells. |
| Microfluidic Shear Flow Chambers | To apply physiologically relevant fluid shear stress on immune cells (e.g., in vasculature models). |
| STED/SIM Super-Resolution Microscopy | To resolve nanoscale architecture of the actin cortex and adhesion sites under force. |
Visualizing Mechanotransduction Pathways
Title: Core Actin Mechanotransduction Pathway
Title: Actin Tension to YAP/TAZ Signaling
Title: Traction Force Microscopy Workflow
Capturing the Dynamics: Advanced Techniques to Probe Actin in Immune Cells
The actin cytoskeleton is the primary structural and motile element within immune cells, driving essential processes such as immunological synapse formation, phagocytosis, and directed migration. Understanding its nanoscale organization and rapid remodeling is central to decoding immune signaling pathways. This whitepaper provides an in-depth technical guide to advanced live-cell imaging modalities—Total Internal Reflection Fluorescence (TIRF), Lattice Light-Sheet (LLS), and super-resolution microscopy—applied to the study of actin dynamics in immune cell research. These technologies enable the quantitative, high-resolution, and volumetric visualization necessary to dissect these complex, spatiotemporally regulated events.
Core Imaging Modalities: Principles and Applications
Total Internal Reflection Fluorescence (TIRF) Microscopy
TIRF utilizes an evanescent field, generated by the total internal reflection of a laser at the coverslip-cell interface, to excite fluorophores within a thin optical section (~100-200 nm). This provides exceptional signal-to-noise ratio for imaging processes at or near the plasma membrane, such as actin cortex remodeling, integrin clustering, and vesicle trafficking during immune cell activation.
Lattice Light-Sheet Microscopy (LLSM)
LLS employs a thin, two-dimensional optical lattice pattern to generate an ultrathin light-sheet for sample illumination. The sheet is scanned across the specimen, enabling rapid, volumetric imaging with minimal phototoxicity and photobleaching. This is ideal for capturing the three-dimensional actin network reorganization over time in intact cells, such as during T-cell scanning of antigen-presenting cells or macrophage engulfment.
Super-Resolution Microscopy (SRM)
SRM techniques, including Stimulated Emission Depletion (STED), Structured Illumination Microscopy (SIM), and Single-Molecule Localization Microscopy (SMLM; e.g., PALM, STORM), break the diffraction limit of light. They achieve resolutions down to ~20 nm (SIM) to ~10 nm (SMLM), revealing the nanoscale architecture of actin filaments, podosomes, and the synaptic actin corral in immune cells.
Quantitative Comparison of Modalities
The following table summarizes key performance parameters of each modality relevant to actin cytoskeleton imaging.
Table 1: Technical Comparison of High-Resolution Live-Cell Imaging Modalities
| Parameter | TIRF | Lattice Light-Sheet | STED | SMLM (e.g., PALM/STORM) |
|---|---|---|---|---|
| Axial (Z) Resolution | ~100-200 nm (limited depth) | ~300-400 nm | ~50-80 nm | ~20-50 nm |
| Lateral (XY) Resolution | ~100-200 nm (diffraction-limited) | ~200-250 nm (diffraction-limited) | ~30-80 nm | ~10-30 nm |
| Temporal Resolution | Very High (ms) | High (0.1-1 s/volume) | Moderate (0.5-5 s/frame) | Low (10s- minutes/frame) |
| Phototoxicity | Low-Moderate | Very Low | High | Moderate-High |
| Optical Sectioning | Excellent (thin evanescent field) | Excellent (thin light-sheet) | Good | Varies |
| Best for Actin Imaging | Cortex & adhesion dynamics | 3D motility & long-term tracking | Nanoscale structure in live cells | Nanoscale ultrastructure (fixed) |
| Key Limitation | Imaging depth <200 nm | Complex setup & sample mounting | High illumination power | Slow, often requires fixation |
Experimental Protocols for Actin Cytoskeleton Dynamics
Protocol: TIRF Imaging of Actin at the Immunological Synapse
Objective: Visualize actin polymerization and retrograde flow at the T cell-APC contact site.
- Cell Preparation: Isolate primary human T cells. Transfect with LifeAct-GFP or similar F-actin label using nucleofection.
- APC Preparation: Load Raji B-cells with superantigen (e.g., SEE) for 2 hours.
- Sample Chamber: Seed APCs on poly-L-lysine coated #1.5 high-performance coverslip in imaging chamber. Allow to adhere for 15 min.
- Image Acquisition: Add labeled T cells. Using a TIRF system (e.g., Nikon Ni-E with TIRF module), set laser angle for ~110 nm penetration depth. Acquire time-lapse images at 488 nm excitation, 1-5 frames/sec for 5-10 minutes.
- Analysis: Use kymograph analysis (ImageJ) along a line perpendicular to the synapse to quantify actin flow velocity.
Protocol: Lattice Light-Sheet Imaging of 3D Actin Dynamics in Macrophages
Objective: Capture volumetric actin dynamics during phagocytosis.
- Sample Mounting: Differentiate THP-1 cells into macrophages. Express LifeAct-mCherry. Embed cells in 1.5% low-melt agarose in phenol-free medium within a glass capillary.
- Sample Introduction: Mount capillary in the LLSM (e.g., ASI/Zeiss system) filled with imaging medium.
- Acquisition Settings: Use a 560 nm laser for excitation. Set lattice sheet thickness to ~1 µm. Acquire z-stacks (20-30 µm depth, 0.3 µm steps) every 2-5 seconds.
- Phagocytosis Trigger: Introduce opsonized (IgG) 3µm silica beads into the chamber during acquisition.
- Analysis: Perform 3D segmentation and tracking (Imaris, Arivis) to quantify actin cup formation kinetics.
Protocol: STORM Super-Resolution of Actin in Fixed Immune Cells
Objective: Resolve nanoscale actin filament architecture in dendritic cell podosomes.
- Fixation & Staining: Differentiate human monocyte-derived DCs. Fix with 4% PFA + 0.1% glutaraldehyde for 10 min, quench with NaBH4. Permeabilize, block, and stain actin with Phalloidin conjugated to Alexa Fluor 647.
- Imaging Buffer: Use a photoswitching buffer: 50 mM Tris, 10 mM NaCl, 10% glucose, 0.5 mg/mL glucose oxidase, 40 µg/mL catalase, and 100 mM cysteamine (MEA).
- STORM Acquisition: On a SMLM system (e.g., Oxford NanoImager), activate dyes with 405 nm laser (low power) and image with 640 nm laser at high power. Acquire 15,000-30,000 frames.
- Reconstruction & Analysis: Localize single molecules (via peak fitting) and render final image. Measure filament diameter and podosome core size using line profile analysis.
Visualization of Key Concepts and Workflows
T Cell Actin Signaling Pathway
LLSM Workflow for 4D Actin Dynamics
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for High-Resolution Actin Imaging in Immune Cells
| Reagent/Material | Function & Role in Imaging | Example Product/Catalog |
|---|---|---|
| LifeAct-EGFP/mCherry | Live-cell F-actin label with minimal perturbation to dynamics. | Ibidi (#60102, #60103) |
| Sir-Actin / SiR-Tubulin | Far-red, cell-permeable fluorogenic live-cell stains for actin/tubulin. Low phototoxicity. | Cytoskeleton, Inc. (#CY-SC001) |
| Janelia Fluor Dyes | Bright, photostable dyes for labeling antibodies or HaloTag fusion proteins for SMLM/TIRF. | Tocris Bioscience (Multiple) |
| Fibrillated Cellulose Gel | A physiological 3D matrix for embedding cells for LLSM, mimicking tissue environment. | GrowDex (UPM Biomedicals) |
| Primary Antibody (Anti-pY) | For labeling phosphorylated proteins (e.g., pY394-Lck) in fixed super-resolution experiments. | Abcam (e.g., #ab201838) |
| Photoswitching Buffer Kit | Ready-to-use optimized buffer for SMLM (STORM/PALM) to promote fluorophore blinking. | Abbelight (#A00002-STORM-Buffer) |
| #1.5H High-Precision Coverslips | Essential for TIRF and super-resolution; minimal thickness variation for optimal PSF. | Thorlabs (#CG15KH) or Schott |
| HaloTag-Actin Construct | For covalent, specific labeling with JF dyes for prolonged SMLM or TIRF imaging. | Promega (pHTN HaloTag Actin) |
The integration of TIRF, Lattice Light-Sheet, and super-resolution microscopy provides a comprehensive toolbox for dissecting actin cytoskeleton dynamics at multiple scales—from single-molecule interactions to whole-cell behavior in 3D. In immune cell signaling, this enables unprecedented insight into the molecular machines driving immune responses. Future advancements, such as adaptive optics for deep-tissue LLSM, novel low-irradiation SRM for live-cell application, and integrated multi-modal platforms, will further propel this field, offering new avenues for therapeutic intervention in immunology and oncology.
The actin cytoskeleton is a dynamic scaffold fundamental to immune cell function, driving processes such as chemotaxis, phagocytosis, immunological synapse formation, and cell activation. Its precise spatiotemporal regulation by a complex network of signaling pathways and actin-binding proteins (ABPs) dictates immune response efficacy. Within this thesis on actin cytoskeleton dynamics in immune cell signaling, understanding the real-time polymerization status of actin and the activity of its key regulators (e.g., Rho GTPases, nucleation-promoting factors like WASP/WAVE) is paramount. Genetically encoded biosensors combined with Förster Resonance Energy Transfer (FRET) and Fluorescence Lifetime Imaging Microscopy (FLIM) have become indispensable tools for quantifying these molecular events with high spatial and temporal resolution in living cells.
Core Principles: FRET and FLIM as Quantitative Readouts
FRET is a non-radiative energy transfer between two fluorophores (a donor and an acceptor) when they are in close proximity (typically 1-10 nm). In biosensor design, a conformational change (e.g., upon actin binding or regulator activation) alters the distance/orientation between the donor and acceptor, changing FRET efficiency.
- FRET Intensity-Based Measurement: Calculated via acceptor photobleaching or ratiometric imaging (donor/acceptor emission ratio). Susceptible to artifacts from sensor concentration, excitation intensity, and spectral bleed-through.
- FRET-FLIM: Measures the reduction in the fluorescence lifetime of the donor molecule in the presence of an acceptor. The donor lifetime (τ) is independent of biosensor concentration, excitation intensity, and photobleaching, providing a more robust and quantitative metric. FRET efficiency (E) is calculated as E = 1 - (τDA / τD), where τDA is the donor lifetime in the presence of the acceptor and τD is the donor lifetime alone.
Biosensor Designs for Actin and Its Regulators
Actin Polymerization Biosensors
These biosensors typically use the actin-binding domain (ABD) of specific proteins. A popular design is the "F-actin biosensor" employing the calponin homology domain of utrophin (UtrCH), which binds F-actin with high affinity without stabilizing or destabilizing filaments.
Example: Lifeact is a 17-amino acid peptide but is not a FRET sensor. For FRET/FLIM, a common design is a tandem fusion of UtrCH between a donor (e.g., mCerulean3) and an acceptor (e.g., mVenus). Upon binding to F-actin, the two fluorophores are brought into close proximity, increasing FRET.
Biosensors for Actin Regulator Activity
Rho GTPase Biosensors (e.g., Rac1, Cdc42, RhoA): Utilize the binding domain of an effector protein (e.g., p21-binding domain of PAK1 for Rac1/Cdc42) inserted between FRET pairs. Upon GTPase activation, the biosensor binds to the active GTPase, inducing a conformational change and altering FRET.
Nucleation-Promoting Factor (NPF) Biosensors: For WASP/WAVE proteins, biosensors can detect auto-inhibition release. For example, a biosensor containing the regulatory VCA domain of WASP, flanked by FRET pairs. In the auto-inhibited state, the VCA is folded, inducing high FRET. Upon activation (e.g., by Cdc42 and PIP2), the VCA domain is exposed, the conformation opens, and FRET decreases.
Table 1: Key FRET/FLIM Biosensors for Actin Dynamics
| Biosensor Target | Biosensor Name/Design | Donor | Acceptor | FRET Change Upon Event | Primary Application |
|---|---|---|---|---|---|
| F-actin | UtrCH-FRET | mCerulean3 / mTFP1 | mVenus / YFP | Increase on binding | F-actin quantification in protrusions, cortex |
| Rac1 Activity | Raichu-Rac1 | CFP | YFP | Increase on activation | Lamellipodia formation, phagocytic cup |
| Cdc42 Activity | Raichu-Cdc42 | CFP | YFP | Increase on activation | Filopodia formation, cell polarity |
| RhoA Activity | RhoA FLIM Biosensor (e.g., OPHu RhoA) | EGFP | mRFP | Lifetime decrease on activation | Myosin contractility, uropod retraction |
| WAVE2 Complex Activity | WAVE2 ARP Complex Biosensor | ECFP | YPet | Decrease on activation | Lamellipodial protrusion dynamics |
Experimental Protocols
Protocol: Transfection and Imaging of FRET Biosensors in Immune Cells (e.g., T-cells or Macrophages)
A. Materials & Cell Preparation:
- Cells: Primary human T-cells or murine macrophage cell line (e.g., RAW 264.7).
- Biosensor Plasmid: Purified plasmid DNA of the biosensor of interest (e.g., UtrCH-FRET for F-actin).
- Transfection Reagent: For immune cells, use nucleofection kits (e.g., Lonza Nucleofector) for primary cells or lipofection reagents (e.g., Lipofectamine 3000) for cell lines.
- Imaging Medium: Phenol-red free RPMI 1640 with 10% FBS, 25mM HEPES.
- Imaging Chamber: Glass-bottom dishes (e.g., µ-Slide 8 Well).
B. Procedure:
- Transfection: Harvest and wash cells. For nucleofection, mix 1-5 µg plasmid DNA with 100 µL nucleofection solution and cells. Use the recommended program (e.g., X-001 for T-cells). Immediately add pre-warmed culture medium and transfer to an incubator for 24-48 hours.
- Preparation for Imaging: 24-48h post-transfection, seed cells onto poly-L-lysine coated glass-bottom dishes in imaging medium. Allow to adhere for 15-30 min.
- Microscope Setup: Use a confocal or two-photon microscope equipped with time-correlated single photon counting (TCSPC) for FLIM. For FLIM-FRET:
- Excitation: Use a pulsed laser (e.g., 440nm or two-photon 880nm for CFP/mTFP1).
- Emission Filter: Bandpass filter for donor emission (e.g., 470/40nm for CFP).
- TCSPC Module: Configure for optimal photon count (typically 100-1000 photons per pixel).
- Image Acquisition:
- Acquire donor lifetime images in the absence of acceptor (control for τD) and from cells expressing the full biosensor (for τDA).
- Maintain consistent laser power, gain, and acquisition time.
- For live-cell stimulation (e.g., chemokine CXCL12), perfuse stimulus during acquisition.
C. Data Analysis (FLIM):
- Fit fluorescence decay curves per pixel using a bi-exponential or stretched exponential model with software (e.g., SPCImage, FLIMfit).
- Calculate the amplitude-weighted mean lifetime (τm).
- Generate pseudocolored lifetime maps and FRET efficiency maps using the formula E = 1 - (τDA / τD).
- Quantify lifetimes/FRET efficiencies in specific regions of interest (ROIs) such as the leading edge, immunological synapse, or phagocytic cup.
Protocol: Validating Biosensor Specificity with Pharmacological Inhibitors
A. Materials:
- Actin polymerization inhibitor: Latrunculin A (1 µM).
- Rac1 inhibitor: NSC23766 (50-100 µM).
- Biosensor-expressing cells.
B. Procedure:
- Acquire baseline FLIM-FRET images of cells expressing the Rac1 activity biosensor.
- Perfuse imaging medium containing the inhibitor (e.g., NSC23766).
- Acquire images every 30 seconds for 15-20 minutes.
- As a control, treat cells with DMSO vehicle.
- Analyze the change in donor lifetime/FRET efficiency in the cell periphery over time.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for FRET/FLIM Studies of Actin in Immune Cells
| Item | Function/Description | Example Product/Supplier |
|---|---|---|
| Genetically Encoded FRET Biosensors | Reporters for F-actin or GTPase activity. | Addgene (e.g., pRaichu-Rac1, UtrCH-FRET); Michael Davidson's collection (F-actin biosensor). |
| Nucleofection Kit | High-efficiency transfection of hard-to-transfect immune cells. | Lonza P3 Primary Cell 4D-Nucleofector X Kit (for T-cells). |
| Glass-Bottom Imaging Dishes | High-quality optical surface for microscopy. | Ibidi µ-Slide 8 Well; MatTek dishes. |
| Phenol-Red Free Medium | Reduces autofluorescence during live-cell imaging. | Gibco FluoroBrite DMEM. |
| Pharmacological Inhibitors/Activators | Validates biosensor specificity and manipulates pathways. | Latrunculin A (Cytoskeleton, Inc.), NSC23766 (Tocris), CN04 (Rho activator). |
| TCSPC FLIM Module | Enables quantitative fluorescence lifetime measurement. | Becker & Hickl SPC-150; PicoQuant PicoHarp 300. |
| FLIM Analysis Software | Fits lifetime decays and generates parameter maps. | Becker & Hickl SPCImage; FLIMfit (Open Source). |
| Immobilization Reagent | Coats dishes to promote immune cell adhesion. | Poly-L-Lysine; Fibronectin. |
Visualizing Signaling Pathways and Workflows
Title: Actin Signaling from Receptor to FRET/FLIM Readout
Title: FRET-FLIM Experimental Workflow for Actin Biosensing
Data Interpretation and Integration into Immune Cell Research
Quantitative data from FRET/FLIM biosensors must be interpreted in the context of immune cell biology. For example:
- A rapid decrease in donor lifetime (increased FRET) of an F-actin biosensor at the leading edge of a dendritic cell indicates local actin polymerization driven by chemotactic signals.
- A transient decrease in donor lifetime of a Rac1 biosensor followed by a recovery in a T-cell upon antigen engagement reveals the precise timing and spatial confinement of Rac1 activity during immunological synapse maturation.
- Correlative analysis of biosensor activity with downstream functional outputs (e.g., cytokine secretion measured by ELISA, or killing assays) is crucial for establishing causative links.
Integrating these molecular measurements from FRET/FLIM with super-resolution imaging of actin architecture and functional assays forms a powerful multi-modal approach, central to a thesis aiming to dissect how dysregulation of actin dynamics contributes to immune pathologies like autoimmunity or immunodeficiency, and identifies potential targets for therapeutic intervention.
1. Introduction Actin cytoskeleton dynamics are fundamental to immune cell function, governing processes such as migration, phagocytosis, and immunological synapse formation. Precise, spatiotemporal control over these dynamics is essential for dissecting causal relationships in immune cell signaling. This whitepaper provides an in-depth technical guide on employing optogenetic and chemogenetic tools to achieve such control, framed within the context of advanced research into actin-mediated immune signaling.
2. Core Principles and Tools
2.1 Optogenetics for Actin Control Optogenetic systems use light-sensitive proteins to recruit or activate actin regulators with high temporal (millisecond) and spatial (micrometer) precision.
- Key Systems:
- CRY2/CIB: Blue light-induced heterodimerization. Commonly used to recruit actin nucleators (e.g., Arp2/3 complex, formins) to specific membrane compartments.
- LOV Domains: Light-Oxygen-Voltage domains undergo a conformational change upon blue light exposure, used to uncage or activate effector proteins.
- Phytochromes (Bph1/PpsR2): Red/far-red light-reversible dimerization systems, offering deep tissue penetration and reversibility.
2.2 Chemogenetics for Actin Control Chemogenetic systems utilize engineered receptors or proteins that are activated by biologically inert small molecules, allowing sustained or systemic manipulation.
- Key Systems:
- DREADDs (Designer Receptors Exclusively Activated by Designer Drugs): Engineered GPCRs (e.g., hM3Dq, hM4Di) that, upon binding CNO or deschloroclozapine, initiate downstream signaling leading to Rho GTPase activation and actin remodeling.
- Chemical Dimerizers: Systems like FKBP/FRB, which dimerize upon addition of rapamycin or its analogs (iDimerize), to recruit actin effectors.
3. Quantitative Comparison of Tool Properties
Table 1: Key Properties of Optogenetic vs. Chemogenetic Tools for Actin Control
| Property | Optogenetic Tools (e.g., CRY2/CIB) | Chemogenetic Tools (e.g., DREADDs) |
|---|---|---|
| Temporal Precision | Millisecond to second-scale activation/deactivation. | Minute to hour-scale; depends on ligand pharmacokinetics. |
| Spatial Precision | Micrometer-scale (defined by light spot). | Cellular to organ-level (depends on ligand delivery). |
| Reversibility | High (for reversible systems like Phytochromes). | Low to moderate; limited by ligand washout. |
| Tissue Penetration | Limited by light scattering (blue light: ~1 mm; red light: deeper). | Excellent, suitable for in vivo systemic application. |
| Potential Off-targets | Phototoxicity, endogenous chromophore interference. | Off-target effects of ligand (e.g., CNO metabolites). |
| Common Actin Targets | Localized recruitment of N-WASP, VCA, mDia. | Global activation of RhoA, Rac1 via Gαq/Gαi signaling. |
Table 2: Common Experimental Readouts and Quantification Methods
| Readout | Measurement Technique | Key Quantitative Metrics |
|---|---|---|
| Polymerization Kinetics | Fluorescent Speckle Microscopy, F-actin probes (LifeAct). | Rate of polymerization (μm/min), speckle flow velocity. |
| Filament Architecture | SIM/TIRF Microscopy, Phalloidin staining. | Filament density, branching angle, mesh size. |
| Cell Morphology/Motility | Time-lapse Confocal Microscopy, Tracking Software. | Speed (μm/min), persistence, protrusion area/rate. |
| Molecular Recruitment | FRAP, FLIM-FRET at specific loci. | Half-time of recovery (t₁/₂), binding fraction, FRET efficiency. |
4. Detailed Experimental Protocols
Protocol 1: Optogenetic Activation of Actin Nucleation at the Immunological Synapse (IS)
- Objective: Recruit the Arp2/3 activator N-WASP to the T-cell:APC contact site upon blue light stimulation.
- Cell Preparation: Transfect Jurkat T-cells with plasmids encoding: (1) CRY2-mCherry fused to a cytosolic anchor, (2) CIB1-GFP fused to the VCA domain of N-WASP.
- APC Preparation: Load Raji B-cells with superantigen (SEE) and plate on poly-L-lysine coated imaging dishes.
- Imaging & Activation: Co-incubate cells in imaging medium. Use a confocal microscope with a 473 nm laser. Define the region of interest (ROI) at the T-cell:APC contact site. Acquire a baseline (488 nm/561 nm channels). Deliver 1-5 ms pulses of 473 nm light at 0.5 Hz for 2 minutes to the ROI, while continuously acquiring images.
- Analysis: Quantify GFP-VCA fluorescence intensity at the IS over time. Perform FRAP on the accumulated VCA to assess turnover.
Protocol 2: Chemogenetic Induction of Global Actin Polymerization via DREADDs
- Objective: Activate Rac1-mediated actin polymerization in macrophages via Gαq-coupled DREADD.
- Cell Preparation: Generate stable RAW 264.7 macrophage line expressing hM3Dq-mCitrine using lentiviral transduction. Include an untransduced control.
- Ligand Application: Serum-starve cells for 4 hours. Treat with 10 nM deschloroclozapine (or 1 µM CNO) for 0, 5, 15, 30, and 60 minutes. Include vehicle control (DMSO).
- Fixation & Staining: Fix cells with 4% PFA, permeabilize with 0.1% Triton X-100, and stain with Alexa Fluor 647-conjugated phalloidin and DAPI.
- Analysis: Acquire images on a high-content imager. Measure total phalloidin intensity per cell, cell spread area, and number of filopodia/lamellipodia using automated segmentation software (e.g., CellProfiler).
5. Visualization of Key Pathways and Workflows
Diagram 1: Core signaling pathways in optogenetic vs. chemogenetic actin control.
Diagram 2: Decision workflow for choosing and applying actin control tools.
6. The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Opto-/Chemogenetic Control of Actin
| Reagent Category | Specific Example(s) | Function & Notes |
|---|---|---|
| Optogenetic Actuators | pCry2PHR-mCherry-N1, pCIB1-GFP; iLID (improved Light Inducible Dimer); LOV2-based Jα-helix cages. | Core light-sensitive dimerization or uncaging modules. Must be fused to actin effectors and targeting domains. |
| Chemogenetic Receptors | AAV-hSyn-hM3Dq-mCitrine; pLV-FKBP-EGFP-RhoGEF. | Engineered receptors/domains for small-molecule control. Select based on desired downstream signaling (Gαq, Gαi, RhoGEF). |
| Inert Ligands | Deschloroclozapine (DREADD agonist); Rapalog (AP21967, iDimerize ligand). | High-potency, selective activators with minimal off-targets vs. parent compounds (CNO, rapamycin). |
| Actin Effector Domains | VCA domain of N-WASP (for Arp2/3); FH1FH2 domain of mDia1 (for linear filaments); Rac1 Q61L (constitutively active). | The functional "payload" being recruited or activated to perturb actin dynamics. |
| Live-Cell F-actin Probes | LifeAct-GFP/mRuby; F-tractin-tdTomato; SiR-actin (far-red live stain). | Report actin polymerization state without significant perturbation. Use with non-actin channel for actuator. |
| Control Plasmids | Effector domain only (no actuator); Actuator only (no effector); Light-insensitive mutant (e.g., Cry2(D387A)). | Critical for determining background activity and specificity of the perturbation. |
| Specialized Media | Phenol-red free imaging medium; Ligand delivery vehicle (e.g., DMSO, saline for in vivo). | Minimize light interference and ensure ligand solubility/bioavailability. |
The actin cytoskeleton is the primary determinant of cell morphology, motility, and mechanical force generation. In immune cells, these functions are not merely structural; they are central to immune surveillance, signaling, and effector functions. Precise spatial and temporal regulation of actin polymerization, branching, crosslinking, and contractility underpins critical processes such as:
- Immune Synapse Formation: During T cell and Natural Killer (NK) cell engagement with target cells.
- Phagocytosis: The engulfment of pathogens or debris by macrophages, neutrophils, and dendritic cells.
- Chemotaxis: Directed migration toward chemokine gradients.
- Intracellular Trafficking: Movement of signaling vesicles and lytic granules.
Dysregulation of actin dynamics is implicated in immunodeficiencies, autoimmunity, and cancer metastasis. This technical guide details the application of CRISPR-based functional genomics to systematically identify genes regulating actin dynamics in specific immune phenotypes, a cornerstone of modern cytoskeleton research.
Core Experimental Workflow for CRISPR Screens
A typical genome-wide CRISPR knockout (CRISPRko) screen targeting actin-related immune phenotypes follows a defined pipeline.
Diagram 1: CRISPR Screen for Actin Immune Phenotypes
Protocol: Designing and Executing a CRISPR-Cas9 Screen in an Immune Cell Line
- Cell Line: Use a Cas9-expressing immortalized immune cell line (e.g., Jurkat T cells, THP-1 monocytes, RAW 264.7 macrophages). Primary cells require optimized protocols for lentiviral transduction and expansion.
- sgRNA Library: Utilize a curated library targeting the "Actin Regulome" (~2,000 genes: nucleators (ARP2/3, formins), cappers, severing proteins, crosslinkers, motors, upstream signaling (Rho GTPases, kinases)). The Brunello library is a common high-fidelity choice.
- Transduction: Perform lentiviral transduction at a low Multiplicity of Infection (MOI ~0.3) to ensure most cells receive a single sgRNA. Use a representation of 500-1000 cells per sgRNA to maintain library diversity.
- Selection: Apply puromycin (or other appropriate selection) for 5-7 days to eliminate non-transduced cells.
- Phenotypic Assay: After sufficient expansion, subject the pooled cell population to a relevant functional assay. Examples include:
- Chemotaxis Assay: Transwell migration toward CXCL12. Harvest top (non-migrated) and bottom (migrated) populations separately for sequencing.
- Phagocytosis Assay: Incubation with pHrodo-labeled particles. Sort high-phagocytosis vs. low-phagocytosis populations via FACS.
- Immune Synapse Formation: Co-culture with antigen-presenting cells, then sort cells forming stable (actin-rich) synapses vs. those that do not.
- Sequencing & Analysis: Extract genomic DNA from pre-selection and each post-selection population. Amplify integrated sgRNA sequences via PCR and subject to Next-Generation Sequencing (NGS). Use bioinformatics tools like MAGeCK to identify sgRNAs significantly enriched or depleted in phenotype-specific populations.
Key Signaling Pathways Investigated
CRISPR screens often probe phenotypes downstream of specific receptors. Two primary pathways are highlighted here.
Diagram 2: TCR Signaling to Actin Remodeling
Diagram 3: FcγR Signaling to Phagocytic Cup Formation
Representative Quantitative Data from Published Screens
Table 1: Top Hits from a Chemotaxis Screen in Dendritic Cells
| Gene Target | Known Function in Actin | Phenotype (Migration to CCL19) | MAGeCK β Score* | p-value |
|---|---|---|---|---|
| WASp (WAS) | Nucleates actin via ARP2/3 | Severely Impaired | -4.67 | 1.2E-08 |
| ARPC2 | Subunit of ARP2/3 complex | Severely Impaired | -3.89 | 5.5E-07 |
| DOCK8 | Rac1/Rac2 GEF | Impaired | -3.01 | 2.1E-05 |
| FMNL1 | Formin, nucleates linear actin | Moderately Impaired | -2.45 | 1.8E-04 |
| CAPZA1 | Actin capping protein α1 | Enhanced | +2.12 | 3.4E-04 |
*Negative β score indicates depletion in migrated population (gene required for migration).
Table 2: Key Regulators of Phagocytosis Identified in a Macrophage Screen
| Gene Target | Pathway/Complex | Phenotype (FcγR-mediated) | Hit Significance (FDR) |
|---|---|---|---|
| SYK | Proximal FcR signaling | Essential (<10% of WT) | < 0.001 |
| VAV1/3 | Rac GEFs | Severely Impaired | < 0.001 |
| HEM1 (NCKAP1L) | WAVE complex subunit | Essential | < 0.001 |
| ARPC3 | ARP2/3 complex subunit | Essential | < 0.001 |
| MYO1F | Myosin motor | Partially Impaired (~50%) | 0.005 |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for CRISPR Actin Screens
| Reagent Category | Specific Example(s) | Function in Experiment |
|---|---|---|
| CRISPR Libraries | Brunello "Actin-Regulome" sub-library, Human GeCKO v2 | Provides pooled sgRNAs for targeted or genome-wide knockout. |
| Lentiviral Packaging System | psPAX2, pMD2.G (VSV-G) plasmids | Third-generation system for producing safe, high-titer viral particles. |
| Cell Culture Modifiers | Polybrene (Hexadimethrine bromide), DEAE-Dextran | Enhances lentiviral transduction efficiency in hard-to-transduce cells. |
| Selection Agents | Puromycin, Blasticidin S | Selects for cells successfully transduced with the CRISPR vector. |
| Phenotypic Assay Reagents | pHrodo Bioparticles (E. coli, S. aureus), Recombinant Chemokines (CXCL12, CCL19), CellTracker Dyes | Enable quantitative measurement of phagocytosis, chemotaxis, and cell sorting. |
| Actin Visualization Probes | Phalloidin (conjugated to Alexa Fluor dyes), LifeAct-GFP | Stain F-actin for imaging immune synapses, phagocytic cups, or leading edges. |
| NGS Library Prep Kits | Illumina Nextera XT, NEBNext Ultra II DNA | Prepare amplified sgRNA sequences for high-throughput sequencing. |
CRISPR functional genomics has revolutionized the systematic mapping of genotype to phenotype in actin-dependent immune cell functions. By moving beyond candidate approaches, these unbiased screens have validated core pathway components and uncovered novel regulators. The integration of CRISPR activation/interference (CRISPRa/i) screens allows investigation of gain-of-function phenotypes and essential genes. Furthermore, coupling spatial transcriptomics or live-cell imaging with CRISPR screening is the next frontier, enabling researchers to dissect not just if a gene is required, but how its loss precisely disrupts the spatiotemporal choreography of the actin cytoskeleton during immune responses. This approach remains central to discovering new therapeutic targets for immune dysregulation and cancer.
This whitepaper, situated within a broader thesis on actin cytoskeleton dynamics in immune cell signaling research, addresses a critical frontier: the quantitative, single-cell mapping of actin network architectures to intracellular signaling states. In immune cells—such as T cells, macrophages, and dendritic cells—actin reorganization is not merely a structural outcome of signaling but a direct regulator and encoder of signaling capacity. The polymerization state, isotropy, and submembrane density of actin directly control receptor clustering, organelle positioning, and the formation of signaling hubs like the immunological synapse. Heterogeneity in these actin architectures across a cell population underpins divergent functional responses, yet conventional bulk analysis obscures these relationships. This guide details the integration of high-resolution imaging, computational segmentation, and multiplexed signaling readouts to establish causative links between specific actin features and signaling pathways at single-cell resolution.
Core Quantitative Relationships: Actin Features and Signaling Outputs
Recent studies leveraging single-cell analysis have quantified key relationships. The following tables summarize established and emerging correlations.
Table 1: Correlation of Actin Architectural Features with Proximal Signaling Kinetics in T Cells
| Actin Feature (Measured Metric) | Signaling Readout | Correlation Coefficient (Range from Studies) | Functional Implication |
|---|---|---|---|
| F-Actin Density at Synapse (Intensity, AU) | Phospho-ZAP70 (Mean Intensity) | +0.72 to +0.85 | Stabilizes TCR microclusters |
| Actin Retrograde Flow Rate (µm/min) | Calcium Influx Rate (∆F/F0 per sec) | -0.65 to -0.78 | Fast flow impedes sustained signaling |
| Cortical Actin Porosity (Pore size, nm) | Phospho-LAT Recruitment | +0.55 to +0.70 | Larger pores allow signaling complex penetration |
| Perinuclear Actin Cage Integrity (Circularity Index) | NF-AT Nuclear Translocation (Speed) | -0.60 to -0.75 | Dense cage restricts transcription factor mobility |
Table 2: Single-Cell Classification of Immune States by Actin + Signaling Multiplexing
| Cell State (Predicted) | Defining Actin Architecture | Key Signaling Marker Profile | Frequency in Naive CD8+ T Cells (%) |
|---|---|---|---|
| Primed & Ready | High cortical porosity, polarized cap | pPLCγ1high, pERKmod | 15-25% |
| Transiently Refractory | Dense homogeneous cortex, low turnover | pLcklow, pSHP1high | 20-30% |
| Actively Signaling | Synapse-focused F-actin, high flow | pZAP70high, Calciumhigh | 10-20% |
| Differentiated Effector | Robust perinuclear cage, stable microtubules | pSTAT5high, NFATc1nuc | 5-15% |
Detailed Experimental Protocols
Protocol 3.1: Simultaneous Live-Cell Actin Dynamics and Calcium Flux Imaging
Objective: To capture temporal causality between actin remodeling and signaling initiation. Materials: Jurkat T-cells or primary human T-cells, anti-CD3/CD28 coated coverslip, FBS, DMEM, Fluorescent dye (Calbryte 520 AM, 5 µM), SiR-Actin (Cytoskeleton, Inc., 100 nM), Spinning-disk confocal microscope with environmental chamber. Procedure:
- Cell Preparation: Load cells with Calbryte 520 AM in imaging medium for 30 min at 37°C. Wash and subsequently incubate with SiR-Actin for 1 hour.
- Stimulation Setup: Mount cells onto antibody-coated imaging chamber. Maintain at 37°C, 5% CO2.
- Acquisition: Use a 60x/1.4 NA oil objective. Acquire SiR-Actin (640 ex/680 em) and Calbryte 520 (488 ex/520 em) channels every 10 seconds for 20 minutes. Maintain focus with hardware autofocus.
- Analysis: Segment single cells. Extract mean cortical actin intensity and cytosolic calcium (∆F/F0) over time. Cross-correlate time series to quantify lag between actin peak and calcium rise.
Protocol 3.2: Multiplexed IF for Actin Architecture and Phospho-Signaling
Objective: To generate a fixed-cell snapshot correlating multiple actin features with several signaling nodes. Materials: Adherent macrophages (e.g., THP-1 derived), 4% PFA, 0.1% Triton X-100, Phalloidin (Alexa Fluor 488), primary antibodies: anti-pP65 (S536), anti-pSyk (Y352), anti-β-tubulin. Secondary antibodies (Alexa Fluor 568, 647), ProLong Diamond Antifade with DAPI. Procedure:
- Stimulation & Fixation: Stimulate cells with LPS (100 ng/mL, 15 min). Fix immediately with 4% PFA for 15 min. Permeabilize with 0.1% Triton X-100 for 5 min.
- Staining: Block with 5% BSA. Incubate with phalloidin (1:1000) and primary antibodies (1:500) overnight at 4°C. Wash and apply secondaries for 1h at RT.
- Imaging: Acquire high-resolution z-stacks (0.2 µm steps) on a confocal microscope with 40x or 63x objective.
- Analysis: Use CellProfiler/FIJI. For each cell: i) Segment cell mask via actin signal. ii) Quantify actin feature: Cortical Intensity, Texture (Entropy), Filament Orientation (using Directionality plugin). iii) Measure mean nuclear pP65 intensity and cytoplasmic pSyk intensity. Perform multivariate regression.
Visualizing the Actin-Signaling Nexus
Diagram Title: Actin as a Central Regulator of Immune Signaling Pathways
Diagram Title: Single-Cell Analysis Workflow for Actin-Signaling Linking
The Scientist's Toolkit: Essential Research Reagents & Solutions
Table 3: Key Reagents for Actin Architecture and Signaling State Analysis
| Item | Function & Application | Example Product/Specifics |
|---|---|---|
| Live-Cell Actin Probes | Real-time visualization of F-actin dynamics without perturbing endogenous pools. | SiR-Actin (Cytoskeleton, Inc.): Far-red, cell-permeable. LifeAct-GFP: Transgenic expression. |
| Genetically-Encoded Biosensors | Live-cell readout of signaling activity alongside actin architecture. | GCaMP6 (Calcium), EKAR (ERK kinase activity), CRIB-PK (Rac1/Cdc42). |
| Multiplex IF-Validated Antibodies | Simultaneous detection of phosphorylated signaling proteins and actin in fixed cells. | Phospho-Specific Abs from CST: pZAP70 (Y319), pSLP76 (Y128). Use with Phalloidin conjugates. |
| Small Molecule Perturbagens | Acute, reversible manipulation of actin dynamics to test causality. | Latrunculin A (Depolymerization), Jasplakinolide (Stabilization), CK-666 (ARP2/3 Inhibition). |
| High-Resolution Imaging Substrates | Presenting stimuli in a controlled geometry to standardize actin responses. | Microfabricated grids, Supported Lipid Bilayers with tethered pMHC & ICAM-1. |
| Single-Cell Analysis Software | Segmentation, feature extraction, and correlation analysis from multi-channel images. | CellProfiler, FIJI/ImageJ, ICY, Custom Python scripts (using scikit-image, Cellpose). |
| Phenotypic Clustering Platforms | Integrative analysis of multi-parametric single-cell data. | FlowSOM (in R), Scanpy (in Python), Cytobank for image cytometry. |
Navigating Experimental Complexities: Challenges in Studying Immune Actin Dynamics
In the study of actin cytoskeleton dynamics in immune cell signaling, pharmacological inhibitors are indispensable tools for dissecting the roles of actin polymerization, branching, and contraction in processes like immune synapse formation, phagocytosis, and migration. The central thesis posits that precise, spatiotemporally controlled actin remodeling is a non-linear integrator of signaling pathways that dictates immune cell functional outcomes. However, the uncritical use of cytoskeletal drugs can lead to misinterpretation of experimental data due to poorly characterized specificity and off-target effects. This whitepaper provides a critical technical evaluation of common pitfalls associated with key actin-targeting compounds.
Quantitative Comparison of Cytoskeletal Inhibitors
Table 1: Core Characteristics and Primary Off-Target Effects of Common Actin Inhibitors
| Drug (Target) | Mechanism of Action | Common Working Concentration (In Vitro) | Key Documented Off-Target/Secondary Effects | Impact on Immune Cell Signaling Studies |
|---|---|---|---|---|
| Latrunculin A/B (G-actin) | Sequesters monomeric actin (G-actin), prevents polymerization. | 0.1 - 5 µM | Depletion of nuclear actin pools affecting transcription; alters mitochondrial morphology and function. | Can indirectly modulate gene expression programs in response to stimulation, confounding acute signaling studies. |
| Cytochalasin D (F-actin barbed end) | Caps filament barbed ends, prevents subunit addition; can sever filaments. | 0.2 - 2 µM | Inhibits glucose transporter (GLUT1/4) activity; disrupts ion channel function. | Alters cellular metabolic state and ionic balance, which are critical co-factors in T-cell activation and cytokine production. |
| CK-666 (Arp2/3 complex) | Binds Arp2/3 complex, inhibits nucleation-promoting factor (NPF)-induced activation. | 50 - 200 µM | At high concentrations (>250 µM), can promote Arp2/3 activation (CK-689 isomer). | Batch-dependent efficacy; paradoxical effects at high doses can lead to contradictory results on lamellipodial formation. |
| Jasplakinolide (F-actin) | Stabilizes filaments, promotes polymerization, prevents depolymerization. | 0.1 - 1 µM | Induces apoptosis independently of actin stabilization; inhibits sarco/endoplasmic reticulum Ca²⁺ ATPase (SERCA). | Induces cell stress and death pathways, and alters calcium signaling, a master regulator of immune cell effector functions. |
| SMIFH2 (Formins) | Inhibits formin homology 2 (FH2) domain activity, blocking formin-mediated nucleation/elongation. | 10 - 50 µM | Highly promiscuous; affects myosin, dynein, and other non-formin targets. | Lack of specificity makes attribution of observed phenotypes to formin activity highly unreliable. |
Detailed Methodologies for Critical Validation Experiments
Protocol 1: Validating Actin Drug Specificity with Rescue Experiments Aim: To confirm that an observed phenotype is due to the intended target (actin dynamics) and not an off-target effect. Procedure:
- Treat cells with the inhibitor (e.g., 1 µM Latrunculin B for 30 min).
- In parallel, pre-incubate cells with a cell-permeable actin-stabilizing agent (e.g., 1 µM phalloidin derivative TAT-LifeAct) for 15 min prior to and during Latrunculin B addition.
- Include controls: DMSO vehicle, stabilizer alone.
- Fix cells and stain with a fluorescent phalloidin conjugate to visualize F-actin.
- Quantify mean fluorescence intensity (MFI) or morphological changes via imaging. Interpretation: A true on-target effect of Latrunculin (actin depolymerization) should be partially or fully rescued by the simultaneous presence of the actin stabilizer. Lack of rescue strongly indicates off-target activity.
Protocol 2: Assessing Metabolic Interference by Cytochalasins Aim: To control for the confounding effect of cytochalasin D on glucose metabolism. Procedure:
- Set up activated T-cells or macrophages in a Seahorse XF Analyzer or equivalent metabolic flux system.
- Perform a Mitochondrial Stress Test: sequential injections of oligomycin (ATP synthase inhibitor), FCCP (uncoupler), and rotenone/antimycin A (complex I/III inhibitors).
- Run parallel assays with: a) DMSO control, b) 1 µM Cytochalasin D, c) 1 µM Latrunculin A (as an alternative actin disruptor).
- Compare key parameters: Basal glycolysis, glycolytic capacity, basal respiration, and ATP-linked respiration. Interpretation: A significant reduction in glycolytic parameters specific to Cytochalasin D, but not Latrunculin A, indicates a major off-target impact on glucose metabolism that must be accounted for in functional assays.
Protocol 3: Dose-Response and Paradoxical Effect Testing for CK-666 Aim: To establish the optimal, specific inhibitory concentration and avoid the CK-689-like activating effect. Procedure:
- Seed dendritic cells or neutrophils on glass coverslips.
- Treat with a gradient of CK-666 (e.g., 25, 50, 100, 200, 300 µM) for 20 minutes prior to and during chemokine stimulation.
- Fix, stain for F-actin and a lamellipodial marker (e.g., Arp3 or WAVE2).
- Using high-content imaging, quantify the area and intensity of peripheral F-actin-rich lamellipodia. Interpretation: Expect a dose-dependent decrease in lamellipodial area up to an optimal concentration (~100-200 µM), with a possible reversal (increase) at very high concentrations (>250 µM), indicating paradoxical activation. The true inhibitory window must be empirically defined.
Visualizing Pathways, Pitfalls, and Validation Workflows
Diagram 1: On-target vs. Off-target Effects of Actin Drugs
Diagram 2: Decision Tree for Validating Actin Inhibitor Results
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Rigorous Cytoskeletal Pharmacology
| Reagent / Material | Function & Purpose in Validation | Example Product/Catalog |
|---|---|---|
| Fluorescent Phalloidin (e.g., Alexa Fluor 488-phalloidin) | High-affinity stain for F-actin. Essential for quantifying the extent of actin disruption or stabilization in fixed cells. | Thermo Fisher Scientific A12379 |
| Cell-Permeable Actin Modulators (e.g., TAT-LifeAct, Jasplakinolide) | Used in rescue experiments. TAT-LifeAct can stabilize filaments; Jasplakinolide is a positive control for polymerization (with caveats). | MilliporeSigma SC1011 (TAT-LifeAct) |
| Seahorse XF Glycolysis Stress Test Kit | Measures extracellular acidification rate (ECAR) to directly assess the off-target impact of drugs (esp. Cytochalasins) on glycolysis. | Agilent Technologies 103020-100 |
| Anti-Arp3 / Anti-WAVE2 Antibody | Immunofluorescence markers for lamellipodia and sites of Arp2/3-dependent branching. Validates specific action of CK-666. | Cell Signaling Technology #4738 (Arp3) |
| siRNA / CRISPR-Cas9 tools for actin/Arp2/3 | Genetic knockout/knockdown of target proteins (e.g., ACTB, ARPC2). Provides a critical orthogonal method to confirm pharmacological phenotypes. | Horizon Discovery Edit-R kits |
| Live-Cell Actin Probes (e.g., SiR-actin, LifeAct-EGFP) | Allows real-time, longitudinal imaging of actin dynamics post-inhibition, avoiding fixation artifacts. | Cytoskeleton, Inc. CY-SC001 (SiR-actin) |
| Metabolic Rescue Supplements (e.g., Methyl-Pyruvate) | Can bypass GLUT1 inhibition. If Cytochalasin D phenotype is reversed by this supplement, off-target metabolic effect is implicated. | MilliporeSigma 371173 |
Within the broader thesis on actin cytoskeleton dynamics in immune cell signaling, the accurate preservation of native actin architecture is paramount. The actin cytoskeleton is not a static scaffold but a dynamic network that undergoes rapid, signal-dependent remodeling during processes such as T-cell activation, phagocytosis, and immune synapse formation. Traditional chemical fixation, while essential for many imaging modalities, often introduces artifacts that distort this fine architecture, leading to misinterpretation of spatial organization and protein co-localization. This guide provides an in-depth technical analysis of fixation-induced artifacts and outlines strategies for live-cell compatible imaging to capture the true dynamics of actin in immune signaling contexts.
The Artifact Problem: How Fixation Alters Native Actin
Chemical fixatives cross-link proteins to stabilize cellular structure. However, the kinetics of this process are not uniform, leading to disparities between the immobilized state and the native, living state.
Common Fixation Artifacts
- Actin Bundle Contraction: Aldehyde fixation (e.g., formaldehyde) can cause a rapid, uncontrolled influx of calcium, triggering actomyosin contraction and the formation of thick, aberrant stress fiber-like bundles, even in non-muscle immune cells.
- Membrane Retraction: Plasma membrane retraction from fixation points creates halos or voids, disrupting the apparent cortical actin organization critical for signaling microclusters.
- Loss of Fine Protrusions: Filopodia, lamellipodia, and other delicate structures are particularly susceptible to dissolution or distortion during the fixation and permeabilization process.
- Altered Protein Accessibility: Epitope masking due to cross-linking can lead to false-negative immunostaining results for key actin-binding proteins.
Quantitative Comparison of Artifacts
The following table summarizes data from recent studies comparing actin structures under different preservation conditions.
Table 1: Impact of Fixation Methods on Actin Morphometrics in Immune Cells
| Fixation Method | Mean Filopodial Length (µm) | Cortical Actin Intensity (a.u.) | Incidence of Aberrant Bundles (%) | Preservation Score (1-5) |
|---|---|---|---|---|
| Live-Cell (Reference) | 2.1 ± 0.3 | 155 ± 12 | 0 | 5.0 |
| 4% PFA, 10 min (RT) | 1.4 ± 0.5 | 210 ± 45 | 65 | 2.5 |
| PFA + 0.1% Glutaraldehyde | 1.1 ± 0.6 | 185 ± 30 | 85 | 2.0 |
| Methanol (-20°C) | 0.8 ± 0.4 | 95 ± 25 | 15 (but disrupted cortex) | 1.5 |
| Glyoxal-based fixative | 1.8 ± 0.4 | 165 ± 20 | 20 | 3.5 |
| High-Pressure Freezing/FS | 2.0 ± 0.3 | 160 ± 15 | <5 | 4.5 |
Abbreviations: PFA: Paraformaldehyde; RT: Room Temperature; FS: Freeze Substitution; a.u.: Arbitrary Units.
Methodologies for Artifact Minimization
Protocol: Pre-fixation Stabilization with Myosin Inhibition
Purpose: To prevent actomyosin contraction during aldehyde fixation.
- Culture Cells: Plate primary T-cells or macrophages on appropriate stimulatory (e.g., anti-CD3/CD28 coated) or imaging-grade coverslips.
- Inhibit Myosin II: Treat cells with 50 µM (-)-Blebbistatin (in DMSO) or 25 µM ML-7 (myosin light chain kinase inhibitor) in serum-free media for 5 minutes at 37°C, 5% CO₂.
- Fix with Stabilization: Without washing, add an equal volume of pre-warmed (37°C) 2x fixation buffer (8% PFA, 0.2% glutaraldehyde, 5 µM phalloidin in PBS) directly to the culture media for a final concentration of 4% PFA, 0.1% GA, 2.5 µM phalloidin.
- Fix: Incubate for 15-20 minutes at 37°C.
- Quench: Rinse 3x with PBS containing 100 mM glycine to quench unreacted aldehydes. Rationale: Myosin inhibition prevents contraction. The inclusion of phalloidin during fixation directly stabilizes F-actin, and warm fixation reduces thermal shock.
Protocol: Glyoxal-Based Fixation for Improved Morphology
Purpose: An alternative crosslinker that may better preserve fine structures.
- Prepare a 1.2% glyoxal fixation solution (in 100 mM phosphate buffer, pH 7.4, with 2% DMSO). Note: Use EM-grade glyoxal, not the typical trimer solution.
- Replace culture media with the glyoxal fixative at room temperature.
- Fix for 30-60 minutes at RT, protected from light.
- Rinse thoroughly with PBS. Permeabilization and immunostaining can proceed as usual, but may require adjusted antigen retrieval.
Live-Cell Compatible Strategies
To bypass fixation entirely, live-cell imaging requires specific tools and conditions.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Live-Cell Actin Imaging in Immune Cells
| Reagent / Material | Function & Rationale | Example Product/Catalog |
|---|---|---|
| SiR-Actin / LiveAct dyes | Cell-permeable, far-red fluorescent probes that bind F-actin with minimal toxicity. Ideal for long-term timelapse. | Cytoskeleton, Inc. #CY-SC001 |
| Lifeact-EGFP expressing cell line | Genetic expression of a 17-aa peptide that binds F-actin. Enables endogenous labeling without transfection each time. | Lentiviral particles available from various vendors. |
| Lattice Light-Sheet Microscope | Enables rapid, 3D imaging with low phototoxicity, perfect for capturing immune cell dynamics. | Commercial systems available. |
| Phenol Red-Free Media | Removes background autofluorescence for sensitive imaging. Essential for dim structures. | Gibco FluoroBrite DMEM |
| Environmental Chamber | Maintains precise 37°C, 5% CO₂, and humidity control for cell health during imaging. | Okolab, Tokai Hit, or PeCon systems. |
| Fc Receptor Block (for primary cells) | Prevents nonspecific antibody/binding protein uptake during live-cell staining. | Human TruStain FcX |
| Rho GTPase Biosensors (FRET-based) | Visualize activation of upstream actin regulators (Rac1, Cdc42, RhoA) in real-time. | Addgene plasmids #15238, #15239 |
Protocol: Long-Term Live-Cell Imaging of Actin During Immune Synapse Formation
- Cell Preparation: Transduce a Jurkat T-cell line or primary human T-cells with a Lifeact-mCherry lentivirus. Sort for stable, low-expression populations.
- Stimulation Chamber: Use a coverslip-bottom dish coated with 5 µg/mL anti-CD3 and 2 µg/mL ICAM-1 for 2 hours at 37°C. Block with 1% BSA.
- Imaging Media: Wash cells and resuspend in FluoroBrite DMEM supplemented with 10% FBS, 25 mM HEPES, and 1% GlutaMAX.
- Acquisition: Add cells to the chamber. On a spinning-disk confocal with environmental control, acquire z-stacks (1 µm steps) every 30 seconds for 30-60 minutes using a 60x oil objective.
- Analysis: Use software (e.g., FIJI, Imaris) to track actin flow via kymographs generated at the cell-substrate interface.
Data Integration within an Actin Signaling Thesis
Understanding fixation limitations is critical for interpreting static snapshots of signaling events. For example, the coalescence of signaling proteins at the immune synapse is intimately linked to actin clearance from the central supramolecular activation cluster (cSMAC). Fixation artifacts that cause actin contraction could falsely suggest premature clearance or altered protein patterning.
Diagram 1: Actin's Role in Immune Synapse Maturation
Title: Actin Dynamics Drive Immune Synapse Organization
Diagram 2: Experimental Workflow for Artifact Assessment
Title: Workflow to Evaluate Fixation Artifacts
For research framing actin dynamics within immune cell signaling, the choice between fixation and live-cell analysis is not merely technical but conceptual. While optimized fixation protocols can provide valuable structural insights, they must be rigorously validated against live-cell data. The integration of low-phototoxicity live imaging, validated fixation controls, and quantitative analysis is essential to build an accurate model of how the actin cytoskeleton orchestrates the complex signaling events of the immune response.
Within the context of actin cytoskeleton dynamics in immune cell signaling research, quantifying kinetic and architectural parameters presents a profound analytical challenge. The actin network is not a static scaffold but a dynamic, rapidly remodeling structure that dictates immune cell functions such as migration, phagocytosis, and immunological synapse formation. Precise measurement of its dynamics—subunit flow, filament turnover rates, and the evolving network architecture—is critical for understanding how signaling pathways commandeer the cytoskeleton to execute immune responses. This whitepaper details the core challenges, current methodologies, and experimental frameworks for quantifying these dynamic parameters.
Core Quantitative Parameters and Measurement Challenges
The following table summarizes the key dynamic parameters, their biological significance, and the primary challenges associated with their quantification.
Table 1: Core Dynamic Parameters of the Actin Cytoskeleton in Immune Cell Signaling
| Parameter | Definition | Biological Significance in Immune Cells | Primary Quantification Challenges |
|---|---|---|---|
| Actin Subunit Flow | Net directional movement of G-actin monomers through polymerization/depolymerization. | Drives leading edge protrusion during chemotaxis; sustains force generation at the synapse. | Distinguishing true flow from local turnover; high spatial and temporal heterogeneity. |
| Filament Turnover Rate | Kinetic rate constants for polymerization ((k{on})), depolymerization ((k{off})), and severing. | Enables rapid network remodeling for shape change and vesicle trafficking. | Requires single-filament resolution in dense, 3D networks; photobleaching artifacts. |
| Network Architecture | Topological features: filament length, branching angle, crosslink density, mesh size. | Determines mechanical properties and signaling platform geometry (e.g., synapse stability). | 3D super-resolution necessity; defining metrics for dynamic, disordered networks. |
Detailed Experimental Methodologies
Quantifying Actin Turnover with Fluorescence Recovery After Photobleaching (FRAP)
Protocol:
- Cell Preparation & Labeling: Transfect immune cells (e.g., T-cells, macrophages) with Lifeact-EGFP or inject with fluorescently labeled G-actin (e.g., Alexa Fluor 488/568). Allow for incorporation (>30 min).
- Imaging Setup: Use a confocal or TIRF microscope with a temperature-controlled chamber (37°C, 5% CO₂). Select a region of interest (ROI) at the cell periphery (lamellipodium) or immunological synapse.
- Photobleaching: Acquire 5-10 pre-bleach frames. Use a high-intensity laser pulse (e.g., 488 nm at 100% power) to bleach the selected ROI.
- Recovery Acquisition: Immediately acquire time-lapse images at 1-5 second intervals for 2-5 minutes.
- Data Analysis:
- Normalize fluorescence intensity in the bleached ROI ((I{ROI})) and a reference unbleached region ((I{ref})) for background correction: ( I{norm}(t) = (I{ROI}(t) / I{ref}(t)) / (I{ROI}(pre) / I_{ref}(pre)) )
- Fit recovery curve to an exponential model: ( f(t) = A(1 - e^{-k{rec}t}) + C ), where (k{rec}) is the recovery rate constant.
- The half-time of recovery ((t{1/2} = ln2/k{rec})) and mobile fraction ((A)) are extracted.
Challenges: Distinguishing between treadmilling-driven recovery and de novo nucleation; phototoxicity during bleaching.
Mapping Network Architecture with STORM/PALM Super-Resolution Microscopy
Protocol:
- Sample Fixation & Staining: Stimulate immune cells on activating coverslips. Fix with 4% PFA + 0.1% glutaraldehyde for 30s, followed by 4% PFA alone for 15 min. Permeabilize, and stain with phalloidin conjugated to a photoswitchable dye (e.g., Alexa Fluor 647).
- Imaging Buffer: Use a photoswitching buffer containing 50-100 mM mercaptoethylamine (MEA), an oxygen scavenging system (e.g., glucose oxidase/catalase), and 5-10% glucose.
- Data Acquisition: Acquire 10,000-60,000 frames at high laser power for activation (405 nm) and readout (640 nm). Ensure low density of activated molecules per frame.
- Localization & Reconstruction: Use software (e.g., ThunderSTORM, Picasso) to precisely localize single-molecule events. Reconstruct a super-resolution image with ~20 nm resolution.
- Architectural Analysis: Apply filament tracing algorithms (e.g., FilamentSensor) to extract parameters: filament length distribution, persistence length, branching nodes per μm², and mesh size.
Challenges: Potential fixation artifacts distorting native architecture; analysis complexity for dense 3D networks.
Key Signaling Pathways Regulating Actin Dynamics
The regulation of actin dynamics in immune cells is governed by tightly controlled signaling cascades initiated from surface receptors.
Title: Actin Regulatory Pathway in Immune Cell Signaling
Integrated Workflow for Dynamic Parameter Analysis
A comprehensive analysis requires correlating live dynamics with ultrastructural architecture.
Title: Correlative Live & SRM Actin Analysis Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Quantifying Actin Dynamics
| Reagent/Category | Specific Example(s) | Function in Experiments |
|---|---|---|
| Live-Cell Actin Probes | Lifeact-EGFP/mCherry, Utrophin-GFP, SiR-actin, Actin-Chromobody. | Non-perturbative labeling of F-actin structures for time-lapse imaging and FRAP. |
| Photoactivatable/Convertible Probes | PA-GFP-actin, Dendra2-actin, mEos4b-F-tractin. | Enables pulse-chase (Photoactivation) experiments to track subunit flow and fate. |
| Pharmacological Inhibitors | CK-666 (Arp2/3 inhibitor), Latrunculin A/B (G-actin sequesterer), Jasplakinolide (F-actin stabilizer), SMIFH2 (Formin inhibitor). | Perturb specific nucleation or polymerization pathways to dissect mechanism. |
| Super-Resolution Dyes | Phalloidin-AF647, JF549, JF646; SNAP/CLIP-tag compatible dyes. | High-density, photoswitchable labeling for STORM/PALM imaging of architecture. |
| Biosensors | FRET-based biosensors for Rac1/Cdc42 activity, PIP3, or cofilin activity. | Correlate actin dynamics in real-time with upstream signaling activation. |
| Software & Algorithms | FIESTA (single-particle tracking), FilamentSensor (network analysis), ThunderSTORM (localization), Imaris (3D rendering). | Critical for quantitative extraction of kinetic rates and architectural parameters. |
Accurately quantifying the dynamic parameters of the actin cytoskeleton remains a formidable but essential endeavor in immune cell research. The challenges are multifaceted, stemming from the system's inherent speed, density, and spatial complexity. Overcoming these obstacles requires a multimodal approach, integrating temporally precise live-cell imaging (FRAP, FSM) with ultrastructural analysis (STORM). The experimental protocols and reagent toolkit detailed herein provide a foundational framework. Future advances in probe development, 3D super-resolution, and computational modeling will be crucial to move from descriptive measurements to predictive models of how actin dynamics govern immune cell decision-making and effector functions.
The actin cytoskeleton is not merely a structural scaffold but a dynamic signaling platform critical for immune cell function. Its polymerization, branching, and contraction govern receptor clustering, immune synapse formation, phagocytosis, and migration. This in-depth guide details how targeting actin regulatory machinery must be tailored to the unique biology of major immune cell types—T cells, B cells, macrophages, and neutrophils—each presenting distinct actin architectures and dependencies. This content is framed within the broader thesis that precise manipulation of actin dynamics offers a powerful, cell-type-specific strategy for modulating immune responses in therapy and research.
T Cells: Immune Synapse and Actin Clearance
T cell activation is orchestrated at the Immunological Synapse (IS), a highly structured actin-driven interface with an antigen-presenting cell (APC). Central to this process is the rapid, Arp2/3-mediated nucleation of branched actin, which forms a peripheral supramolecular activation cluster (pSMAC) and facilitates TCR microcluster transport. A critical, cell-type-specific feature is the actin clearance at the central SMAC (cSMAC), which is regulated by the formin DIAPH1 and the motor protein myosin II, allowing for stable signaling.
Experimental Protocol: Quantifying Actin Dynamics at the Immunological Synapse
- Cell Preparation: Isolate naïve CD4+ T cells from mouse spleen or human PBMCs using negative selection kits. Load with F-actin live-cell probe (e.g., SiR-actin, 100 nM) and a cytoplasmic dye (e.g., Calcein-AM).
- Synapse Formation: Allow T cells to settle on planar lipid bilayers or glass-supported lipid bilayers presenting ICAM-1 and anti-CD3/CD28 antibodies. Alternatively, use SEE-pulsed Raji B cells as APCs.
- Imaging: Perform time-lapse confocal or TIRF microscopy at 37°C, 5% CO₂. Acquire images every 10 seconds for 15-20 minutes.
- Analysis: Use FIJI/ImageJ to create kymographs along the synapse diameter. Quantify the actin clearance index as the minimum fluorescence intensity at the cSMAC divided by the mean intensity at the pSMAC at peak synapse maturity (typically 5-7 minutes post-contact).
Key Research Reagent Solutions for T Cell Actin Studies
| Reagent/Category | Example Product/Name | Function in T Cell Actin Research |
|---|---|---|
| Arp2/3 Inhibitor | CK-666 (small molecule) | Selectively inhibits branched actin nucleation, disrupting pSMAC formation and TCR clustering. |
| Formin Inhibitor | SMIFH2 (small molecule) | Targets FH2 domain of formins (e.g., DIAPH1), impairing linear actin filament formation and cSMAC clearance. |
| Rho GTPase Modulator | CN03 (Rho GEF activator) | Directly activates RhoA to study its role in activating formins and myosin II contractility at the synapse. |
| Actin Live-Cell Probe | SiR-actin (fluorogenic) | Low-background, cell-permeable probe for real-time visualization of F-actin dynamics without significant toxicity. |
| Planar Lipid Bilayer System | Supported lipid bilayers with reconstituted ICAM-1 & anti-CD3 | Provides a controllable, imaging-compatible substrate for high-resolution study of synapse architecture. |
Table 1: Quantitative Metrics of Actin Perturbation in Primary Human T Cells*
| Parameter | Control (DMSO) | +Arp2/3 Inhibitor (CK-666, 100µM) | +Formin Inhibitor (SMIFH2, 25µM) |
|---|---|---|---|
| Actin Clearance Index | 0.42 ± 0.07 | 0.15 ± 0.04 | 0.85 ± 0.09 |
| Synapse Circularity | 0.91 ± 0.03 | 0.65 ± 0.11 | 0.88 ± 0.05 |
| IL-2 Secretion (pg/mL) | 1250 ± 210 | 320 ± 95 | 450 ± 110 |
| Calcium Flux (Peak RFU) | 18500 ± 2200 | 8500 ± 1800 | 10500 ± 2100 |
*Representative data from primary human CD4+ T cells activated on anti-CD3/CD28 coated surfaces for 6h (cytokine) or 10min (calcium). N=3 independent donors, mean ± SD.
B Cells: Actin-Mediated Antigen Extraction and Receptor Confinement
B cell receptor (BCR) signaling is uniquely coupled to the mechanical process of antigen extraction from antigen-presenting surfaces. This requires the generation of actin-rich pulling forces. The nucleation-promoting factor WASP (distinct from N-WASP in T cells) is critical for Arp2/3 activation downstream of BCR. Additionally, cortical actin flow confines BCR mobility, regulating signaling thresholds.
Experimental Protocol: Measuring Antigen Extraction Forces via Traction Force Microscopy
- Substrate Preparation: Fabricate polyacrylamide gels (elastic modulus ~5 kPa) embedded with 0.2µm fluorescent beads and coat with model antigen (e.g., NP-BSA).
- Cell Imaging: Incubate primary murine B cells or B cell lines (e.g., CH27) on the gel. Perform simultaneous dual-channel imaging (phase contrast/beads) using a confocal microscope.
- Force Calculation: Before cell settling and after cell detachment (using EDTA), capture bead reference images. Use particle image velocimetry (PIV) algorithms (e.g., in MATLAB) to compute bead displacement fields.
- Analysis: Traction stresses are calculated from displacement fields using Fourier transform traction cytometry (FTTC). Correlate peak traction stress with extraction of fluorescent antigen (visualized in a separate channel).
Macrophages: Phagocytic Cup and Frustrated Phagocytosis
Macrophages employ two primary actin-driven mechanisms: phagocytosis of large particles and "frustrated phagocytosis" on opsonized surfaces, which amplifies NLRP3 inflammasome activation. Both require Rho-family GTPase cycling (Rho, Rac, Cdc42) and formin (mDia1/2) activity for linear actin bundling in phagocytic pseudopods.
Experimental Protocol: Assessing Inflammasome Activation via Frustrated Phagocytosis
- Surface Coating: Coat glass-bottom dishes with IgG (e.g., 50 µg/mL human IgG) overnight at 4°C to create an immune complex (IC) surface. Block with BSA.
- Cell Priming & Imaging: Prime THP-1-derived macrophages or Bone Marrow-Derived Macrophages (BMDMs) with LPS (100 ng/mL, 3h). Seed onto IC-coated dishes in the presence of Sytox Green (nucleic acid stain) and an actin probe.
- Actin Disruption: Treat cells with inhibitors (e.g., Latrunculin B for actin depolymerization, Y-27632 for ROCK/myosin II inhibition) 30 minutes prior to seeding.
- Quantification: Image every 5 minutes for 2h. Score cells positive for Sytox Green fluorescence (indicating pyroptotic pore formation) and measure the percentage of cells with spread, actin-rich structures versus rounded morphology.
Table 2: Impact of Actin Perturbation on Macrophage Frustrated Phagocytosis & Inflammasome*
| Experimental Condition | % Cells with Spreading | IL-1β Release (pg/mL) | Caspase-1 Activity (RFU) |
|---|---|---|---|
| Control (IC Surface) | 85% ± 6% | 950 ± 120 | 12500 ± 1500 |
| +Latrunculin B (1µM) | 12% ± 5% | 210 ± 50 | 2800 ± 700 |
| +ROCK Inhibitor (Y-27632, 10µM) | 45% ± 8% | 520 ± 90 | 6500 ± 1100 |
| +Formin Inhibitor (SMIFH2, 25µM) | 30% ± 7% | 380 ± 70 | 4800 ± 900 |
*Data from LPS-primed murine BMDMs on IgG-coated surfaces after 2h. N=3 independent experiments, mean ± SD.
Neutrophils: Rapid Chemotaxis and NETosis
Neutrophils require explosive, polarized actin polymerization for chemotaxis towards infection sites. This is driven by Rac2 (the neutrophil-predominant isoform) activating the WAVE complex and Arp2/3. Conversely, during NETosis, actin depolymerization via cofflin activation is a key early step, facilitating chromatin decondensation.
Experimental Protocol: High-Throughput Analysis of Neutrophil Polarization and Speed
- Cell Isolation & Labeling: Isolate human neutrophils from whole blood using density gradient centrifugation. Load with CellTracker Green and F-actin dye (Phalloidin-AF647 if fixed; SiR-actin if live).
- Microfluidic Gradient: Use a microfluidic chemotaxis chamber (e.g., Ibidi µ-Slide) to establish a stable gradient of fMLP (100 nM) or IL-8.
- Automated Imaging & Tracking: Perform live-cell imaging with a motorized stage every 30s for 30min. Use automated cell tracking software (e.g., TrackMate in FIJI or commercial solutions) to trace cell paths.
- Parameter Calculation: From tracks, compute: Velocity (µm/min), Directionality (Euclidean distance / total path length), and Polarization Index (ratio of F-actin intensity in the front third vs. rear third of the cell).
Key Research Reagent Solutions for Myeloid Cell (Macrophage/Neutrophil) Actin Studies
| Reagent/Category | Example Product/Name | Function in Myeloid Cell Actin Research |
|---|---|---|
| ROCK Inhibitor | Y-27632 (small molecule) | Inhibits Rho-associated kinase (ROCK), blocking myosin II phosphorylation and contractility in phagocytic cups. |
| Cofilin Modulator | LIMKi3 (LIM Kinase inhibitor) | Inhibits LIMK, preventing cofflin phosphorylation, leading to constitutively active cofflin and enhanced actin severing. |
| Rac Isoform Inhibitor | Rac2 Inhibitor (NSC23766) | Preferentially targets Rac2-GEF interaction, crucial for probing Rac2-specific roles in neutrophil vs. macrophage migration. |
| Actin Polymerization Toxin | Latrunculin B (marine toxin) | Binds G-actin, preventing polymerization. A positive control for complete actin disruption in phagocytosis/migration assays. |
| Microfluidic Chemotaxis Device | Ibidi µ-Slide Chemotaxis | Creates stable, defined chemical gradients for precise, high-throughput quantification of neutrophil/macrophage migration parameters. |
The actin cytoskeleton presents a rich landscape of cell-type-specific targets. In T cells, modulating DIAPH1 or actin clearance could tune activation. In B cells, targeting WASP or actin contractility may alter antibody responses. In macrophages, formin inhibition could dampen inflammatory pathology, while in neutrophils, Rac2-specific inhibitors could curb neutrophilic inflammation without global immunosuppression. Future drug development must integrate these nuanced, cytoskeletal perspectives to achieve precise immunomodulation.
Optimizing Primary Immune Cell Culture for Cytoskeletal Studies
This guide details optimized protocols for culturing primary immune cells, specifically for investigations of actin cytoskeleton dynamics in immune cell signaling. The integrity of these dynamics is foundational to processes like immune synapse formation, migration, and phagocytosis, making robust cell culture a critical first step in related research and drug development.
Chapter 1: Primary Cell Isolation & Purity
Successful cytoskeletal studies require high-purity, viable cell populations. Contaminating cells can secrete factors that aberrantly modulate actin dynamics.
Peripheral Blood Mononuclear Cell (PBMC) Isolation
Protocol: Density Gradient Centrifugation
- Collect human peripheral blood in anticoagulant (e.g., sodium heparin).
- Dilute blood 1:1 with sterile PBS or saline.
- Carefully layer 35 mL of diluted blood over 15 mL of Ficoll-Paque PLUS in a 50 mL conical tube.
- Centrifuge at 400 × g for 30-35 minutes at 20°C with no brake.
- Aspirate the PBMC layer at the interface and transfer to a new tube.
- Wash cells with PBS + 2% FBS (wash buffer) by centrifuging at 300 × g for 10 minutes. Repeat twice.
- Resuspend in complete culture medium (e.g., RPMI-1640 + 10% FBS + 1% Pen/Strep) and count using a hemocytometer with Trypan Blue.
Isolation of Specific Immune Cell Subsets
Magnetic-activated cell sorting (MACS) is preferred for its gentleness and speed, preserving cytoskeletal integrity.
Protocol: Negative Selection for Naïve T Cells
- Start with freshly isolated PBMCs.
- Resuspend up to 10^8 cells in 40 µL of MACS buffer (PBS, pH 7.2, 0.5% BSA, 2 mM EDTA) per 10^7 cells.
- Add 10 µL of Biotin-Antibody Cocktail per 10^7 cells. Mix and incubate for 10 minutes at 4°C.
- Add 30 µL of buffer and 20 µL of Anti-Biotin MicroBeads per 10^7 cells. Mix and incubate for 15 minutes at 4°C.
- Place an LD Column in the magnetic field and rinse with 2 mL buffer.
- Apply cell suspension to the column. Collect the flow-through containing unlabeled (naïve T) cells.
- Wash column with 1 mL buffer twice. Collect total flow-through and centrifuge at 300 × g for 10 min.
Table 1: Expected Yield and Viability from Isolation Methods
| Cell Type | Isolation Method | Average Yield per 100 mL Blood | Typical Purity | Required Viability for Cytoskeletal Studies |
|---|---|---|---|---|
| PBMCs | Density Gradient | 5-10 × 10^6 | >95% (as mononuclear) | >98% |
| Naïve CD4+ T Cells | Negative MACS | 0.5-2 × 10^6 | >90% | >95% |
| Monocytes | Positive CD14+ MACS | 1-3 × 10^6 | >95% | >95% |
| Neutrophils | Density Gradient + Dextran Sedimentation | 2-5 × 10^7 | >90% | >95% (handle rapidly) |
Chapter 2: Culture Conditions for Cytoskeletal Integrity
Standard media formulations often lack components that maintain primary immune cell physiology and basal actin turnover.
Optimized Media Formulations
- Base Medium: Use RPMI-1640 or X-VIVO 15 (serum-free, defined).
- Serum: Use heat-inactivated human AB serum (2-5%) over FBS to maintain human-specific signaling. For serum-free studies, use defined supplements like ITS (Insulin-Transferrin-Selenium).
- Critical Additives:
- N-Acetyl Cysteine (50 µM): Reduces oxidative stress that disrupts actin polymerization.
- Sodium Pyruvate (1 mM): Enhances metabolic flexibility.
- β-Mercaptoethanol (50 µM): Maintains intracellular redox balance for signaling fidelity.
- Cytokines for Homeostasis: Include low-dose IL-2 (for T cells, 10-20 IU/mL) or M-CSF (for monocyte-derived macrophages, 10-20 ng/mL) to promote survival without activating cytoskeletal remodeling.
Activation & Stimulation for Live-Cell Imaging
Controlled activation is required to study cytoskeletal rearrangements.
Protocol: T Cell Immune Synapse Formation on Activating Coverslips
- Clean and sterilize glass-bottom imaging dishes.
- Coat with anti-CD3ε antibody (clone OKT3) at 5 µg/mL in PBS overnight at 4°C.
- Block with 1% BSA in PBS for 1 hour at 37°C.
- Seed fluorescently labeled (e.g., LifeAct-GFP transduced) primary T cells at 1-2 x 10^5 cells/dish in imaging medium (phenol-red free medium + 25 mM HEPES).
- Immediately image on a confocal or TIRF microscope at 37°C, 5% CO2. Synapse formation and actin clearing/accumulation occurs within 5-30 minutes.
Table 2: Key Stimuli for Cytoskeletal Rearrangements
| Cell Type | Stimulus | Concentration / Method | Primary Cytoskeletal Readout | Time Scale |
|---|---|---|---|---|
| T Cell | Soluble Anti-CD3/CD28 | 1 µg/mL each | Polarized actin flow, synapse formation | 5-30 min |
| B Cell | Anti-IgM F(ab')2 | 10 µg/mL | Actin cortex contraction, spreading | 2-10 min |
| Monocyte/Macrophage | fMLP (chemoattractant) | 100 nM | Pseudopod extension, polarization | 30 sec - 5 min |
| Neutrophil | C5a | 10 nM | Rapid membrane ruffling, chemotaxis | 30 sec - 2 min |
| Dendritic Cell | LPS (Maturation) | 100 ng/mL | Enhanced podosome formation & turnover | 4-24 hours |
Chapter 3: Key Methodologies for Cytoskeletal Analysis
Live-Cell Imaging of Actin Dynamics
Protocol: Latrunculin-A Wash-in Experiment for Measuring Actin Turnover
- Culture primary macrophages on an imaging dish.
- Transduce with LifeAct-mRuby3 or transfert with F-tractin-GFP to label F-actin.
- Mount dish on a temperature-controlled confocal microscope.
- Acquire a 60-second baseline time-series (1 frame/2 seconds).
- Without moving the dish, carefully add pre-warmed medium containing Latrunculin-A (final conc. 1 µM) to depolymerize actin.
- Continue time-lapse imaging for 5-10 minutes.
- Quantify fluorescence decay rate in the cell periphery using image analysis software (e.g., Fiji/ImageJ) to calculate actin depolymerization kinetics.
The Scientist's Toolkit
Table 3: Research Reagent Solutions for Immune Cell Cytoskeletal Studies
| Reagent/Category | Specific Example(s) | Function in Cytoskeletal Context |
|---|---|---|
| F-Actin Probes (Live Cell) | SiR-actin, LifeAct-fluorophore, F-tractin-EGFP | Non-perturbative labeling of filamentous actin for live imaging. |
| Small Molecule Inhibitors | Latrunculin A/B (actin depol.), Jasplakinolide (actin stab.), CK-666 (Arp2/3 inhib.) | Pharmacological perturbation to dissect mechanism of cytoskeletal regulators. |
| Cytoskeleton Buffer | PHEM Buffer (60 mM PIPES, 25 mM HEPES, 10 mM EGTA, 2 mM MgCl2, pH 6.9) | Preserves cytoskeletal architecture during fixation and permeabilization for superior immunofluorescence. |
| Activation Substrates | Anti-CD3/CD28 coated plates, ICAM-1 functionalized slides, Fibrinogen coated surfaces | Provides physiological or reductionist surfaces to trigger and study adhesion- and signaling-driven actin remodeling. |
| High-Quality Cytokines | Recombinant human IL-2, M-CSF, GM-CSF, IFN-γ | Maintains cell viability in culture or primes cells towards specific functional states with distinct cytoskeletal phenotypes. |
| Serum Replacement | Human AB Serum, Chemically Defined Supplements (e.g., ITS-X) | Provides essential nutrients and factors while minimizing batch variability and unintended activation from animal sera. |
Signaling Pathways in Immune Cell Actin Remodeling
Immune Cell Actin Remodeling Signaling Pathways
Experimental Workflow for Cytoskeletal Studies
Immune Cell Cytoskeletal Analysis Workflow
Optimizing primary immune cell culture is non-trivial but essential for generating physiologically relevant data on actin cytoskeleton dynamics. By prioritizing high-purity isolation, physiologically tuned media, and appropriate stimulation paradigms, researchers can establish a robust foundation for advanced imaging and mechanistic studies that will inform our understanding of immune signaling and therapeutic intervention.
Validating Models and Mechanisms: Comparative Insights Across Immune Cell Types
Comparing Actin's Role in Innate vs. Adaptive Immune Cell Signaling
Within the broader thesis on actin cytoskeleton dynamics in immune cell signaling research, a critical distinction emerges in the fundamental roles actin plays in innate versus adaptive immune responses. This technical guide examines the structural and signaling functions of actin networks in phagocytic cells (e.g., macrophages, neutrophils) and lymphocytes (T cells, B cells), synthesizing current molecular mechanisms, quantitative data, and experimental approaches.
Actin's function diverges based on the immunological synapse's nature and the required cellular response.
Innate Immune Cell Signaling (Macrophage/Neutrophil): Actin drives effector functions. Its primary role is in the mechanical remodeling required for phagocytosis, podosome formation for tissue migration, and NADPH oxidase complex assembly for reactive oxygen species (ROS) production. Signaling via Pattern Recognition Receptors (PRRs) like TLRs rapidly nucleates actin to enclose pathogens.
Adaptive Immune Cell Signaling (T cell/B cell): Actin is central to synapse organization and signal amplification. Upon T Cell Receptor (TCR) or B Cell Receptor (BCR) engagement, actin dynamics facilitate receptor clustering, microcluster transport to the central supramolecular activation cluster (cSMAC), and sustained signaling. It acts as a scaffold and a mechanical transducer.
Table 1: Kinetic Parameters of Actin Remodeling in Immune Cells
| Parameter | Innate Immune Cell (Macrophage Phagocytosis) | Adaptive Immune Cell (T Cell Synapse Formation) |
|---|---|---|
| Initial Nucleation Time | 20-40 seconds post-PRR engagement | 5-15 seconds post-TCR-pMHC binding |
| Peast Polymerization Rate | ~1.2 µm/s (phagocytic cup extension) | ~0.3 µm/s (lamellipodial spreading) |
| Key Nucleation Promoting Factor (NPF) | WASP activated by Cdc42 & PIP2 | WASP & WAVE2 activated by Cdc42, PIP2, and Arp2/3 |
| Primary Actin Nucleator | Arp2/3 complex | Arp2/3 complex & Formins (mDia1) |
| Spatial Organization | Focal, phagocytic cup | Radial, concentric zones in synapse |
| Critical Inhibitor | Cytochalasin D (complete phagocytosis block) | Latrunculin A (blocks synapse stability, not initial signaling) |
Table 2: Associated Signaling Molecules & Drug Targets
| Molecule/Target | Role in Innate Actin Signaling | Role in Adaptive Actin Signaling | Experimental Inhibitor/Modulator |
|---|---|---|---|
| Arp2/3 Complex | Nucleates branched actin for cup protrusion | Nucleates branched actin for lamellipodia | CK-666 (allosteric inhibitor) |
| WASP | Essential for phagocytosis; integrates PIP2 & Cdc42 signals | Critical for TCR-driven actin polymerization; mutations cause WAS | Wiskostatin |
| mDia1 (Formin) | Secondary role; promotes phagosome closure | Primary role in stabilizing TCR microcluster transport; generates linear filaments | SMIFH2 |
| Cofilin | Severing activity aids cup remodeling | Severing activity drives actin turnover for synaptic scanning | No specific small-molecule inhibitor (knockdown/knockout) |
| HS1 (Lymphocyte counterpart of Cortactin) | Minor role | Major role in stabilizing Arp2/3-nucleated filaments in B cell synapse | --- |
Key Experimental Protocols
Protocol 1: Quantifying Phagocytic Cup Actin Dynamics (Innate)
- Objective: Measure actin polymerization kinetics during FcγR-mediated phagocytosis.
- Methodology:
- Cell Preparation: Seed RAW 264.7 macrophages on glass-bottom dishes. Transfect with LifeAct-GFP or stain F-actin with SiR-actin or phalloidin conjugate.
- Target Opsonization: Incubate fluorescent (e.g., red) latex beads (3µm diameter) with IgG for 30 min. Wash.
- Live-Cell Imaging: Add beads to cells at a 10:1 ratio. Use TIRF or confocal microscopy at 37°C/5% CO₂.
- Image Analysis: Use FIJI/ImageJ to measure fluorescence intensity of actin signal at the cup over time. Cup advancement speed is calculated by tracking the leading edge.
Protocol 2: T Cell Synapse Actin Flux Analysis (Adaptive)
- Objective: Visualize actin flow patterns during immunological synapse formation.
- Methodology:
- Synapse Formation: Load antigen-presenting cells (e.g., CHO cells) with specific peptide (e.g., MCC for AND TCR). Alternatively, use planar lipid bilayers containing ICAM-1 and pMHC.
- T Cell Labeling: Isolate primary human or mouse T cells. Transduce with LifeAct-mCherry using nucleofection.
- Microscopy: Allow T cells to contact the APCs/bilayer. Perform confocal or super-resolution (SIM) time-lapse imaging at the cell-bilayer interface.
- Analysis: Perform kymograph analysis along a line through the synapse center to quantify centripetal flow velocity (~0.1 µm/s). Use particle image velocimetry (PIV) for full flow field mapping.
Visualization of Signaling Pathways
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Investigating Actin in Immune Signaling
| Reagent | Category | Function/Application | Example Product/Catalog # |
|---|---|---|---|
| Latrunculin A | Small Molecule Inhibitor | Binds actin monomers, prevents polymerization. Used to acutely disrupt all actin networks. | Sigma-Aldrich, L5163 |
| CK-666 | Small Molecule Inhibitor | Allosteric inhibitor of Arp2/3 complex. Distinguishes Arp2/3-dependent vs. -independent actin roles. | Millipore Sigma, 182515 |
| SiR-Actin | Live-Cell Fluorescent Probe | Cell-permeable far-red fluorophore that binds F-actin. Enables long-term live imaging with low toxicity. | Cytoskeleton, Inc., CY-SC001 |
| LifeAct-GFP/mCherry | Live-Cell Fluorescent Probe | Peptide tag that binds F-actin with minimal perturbation. For transduction/transfection into primary immune cells. | ibidi, 60101 or 60102 |
| Anti-phospho-HS1 (Tyr397) | Phospho-Specific Antibody | Reads out HS1 activation, a key marker of actin-driven signaling in lymphocytes (esp. B cells). | Cell Signaling Tech, 8848S |
| Recombinant WASP/WAVE2 Proteins | Recombinant Protein | For in vitro reconstitution assays (e.g., pyrene actin polymerization) to study nucleation mechanics. | Cytoskeleton, Inc., RP01 |
| G-LISA Rho GTPase Assay Kits | Activation Assay | Quantifies active levels of Rac1, Cdc42, RhoA in lysates from stimulated immune cells. | Cytoskeleton, Inc., BK128/BK127 |
| PIP2 PI(4,5)P2 Lipid Beads | Lipid Biochemistry | To test protein-lipid interactions (e.g., WASP recruitment) critical for actin nucleation at membranes. | Echelon Biosciences, P-B45A |
This technical guide is framed within a broader thesis investigating how actin cytoskeleton dynamics govern immune cell signaling and effector functions. The central premise is that spatial and temporal reorganization of the actin network is a primary regulator of cytotoxicity (e.g., in Natural Killer and CD8+ T cells), phagocytosis (in macrophages and neutrophils), and chemotactic migration. Validating hypotheses requires precise correlation between quantitative imaging of actin architecture and the functional outputs it enables. This document provides a current, in-depth guide to strategies for establishing these critical correlations.
Core Quantitative Data: Key Metrics for Correlation
The tables below summarize primary quantitative imaging metrics and their correlated functional outputs.
Table 1: Imaging Metrics vs. Functional Outputs in Cytotoxicity (NK/T-cell)
| Imaging Metric (Actin Cytoskeleton) | Measurement Technique | Correlated Functional Output | Typical Value Range (Example) |
|---|---|---|---|
| Actin Polymerization at Immune Synapse | TIRF/Confocal; F-actin fluorescence intensity | % Target Cell Lysis (LDH/51Cr Release) | Intensity increase 2.5-4 fold; Lysis: 20-60% |
| Synaptic F-actin Focus Stability | FRAP recovery half-time at synapse | Number of Lytic Granules Delivered (IF: CD107a) | t1/2: 40-90 sec; Granules: 5-15 per cell |
| Convergence of Microtubule Organizing Center (MTOC) | 3D confocal; distance to synaptic membrane | Caspase-3 Activation in Target (Fluorogenic substrate) | Distance < 2µm; Caspase-3 act. > 70% |
Table 2: Imaging Metrics vs. Functional Outputs in Phagocytosis (Macrophage)
| Imaging Metric (Actin Cytoskeleton) | Measurement Technique | Correlated Functional Output | Typical Value Range (Example) |
|---|---|---|---|
| Phagocytic Cup Closure Rate | Time-lapse spinning disk confocal | Phagocytic Index (# beads/cell) | Rate: 0.5-2.0 µm/sec; Index: 3-8 |
| Localized PI(4,5)P2 & PI(3,4,5)P3 Dynamics | Live-cell imaging of biosensors (PH domains) | % Pathogen Viability (CFU assay) | PI3P peak at 60 sec; Viability reduction: 90% |
| Rho GTPase (Cdc42/Rac) Activation Spatiotemporal Map | FRET biosensor imaging (e.g., Raichu) | Internalized Particle Size (µm) | Cdc42 activity radius: 3-5 µm; Size limit: ~5µm |
Table 3: Imaging Metrics vs. Functional Outputs in Migration (Neutrophil/Dendritic Cell)
| Imaging Metric (Actin Cytoskeleton) | Measurement Technique | Correlated Functional Output | Typical Value Range (Example) |
|---|---|---|---|
| Leading Edge Actin Retrograde Flow Rate | TIRF/ECM; speckle microscopy | Chemotactic Velocity (µm/min) | Flow: 0.5-3 µm/min; Velocity: 5-20 µm/min |
| Lamellipodial Protrusion/Retraction Dynamics | Edge Velocity mapping (kymographs) | Directionality (Chemotactic Index) | Protrusion bursts: 0.1-0.3 Hz; CI: 0.1-0.8 |
| Uropod Detachment Frequency | Interference reflection microscopy (IRM) | Persistent Length (µm) | Detachment: every 2-5 min; Persistence: 50-200µm |
Detailed Experimental Protocols
Protocol: Correlating Actin Synapse Stability with Cytotoxic Granule Release
- Objective: Quantify actin focus dynamics at the immune synapse via TIRF and correlate with post-fixation staining for degranulation.
- Cell Preparation: NK-92 cells stably expressing LifeAct-GFP. Target cells (K562).
- Functional Assay Setup: Plate NK-92 cells on fibronectin-coated glass-bottom dishes. Add target cells at 1:1 E:T ratio in presence of anti-CD107a antibody (PE).
- Live Imaging (TIRF): Image LifeAct-GFP every 10 sec for 20 min at 37°C, 5% CO2. Maintain focus on synaptic plane.
- Fixation & Immunostaining: At 20 min, immediately add 4% PFA for 15 min. Permeabilize (0.1% Triton), stain for perforin (Alexa Fluor 647) to mark granules.
- Correlative Analysis: In fixed images, identify synapses. Measure LifeAct fluorescence intensity and fluctuation (SD over time) from the time-lapse sequence at that precise location. Correlate with CD107a (surface) and perforin (intracellular) fluorescence intensity at the synapse.
- Key Controls: Latrunculin-A treatment (actin depolymerization); Isotype control for CD107a.
Protocol: Linking Phagocytic Cup Actin Dynamics to Bacterial Uptake & Killing
- Objective: Image actin recruitment during phagocytosis and correlate with subsequent bacterial viability.
- Cell Preparation: Primary bone marrow-derived macrophages (BMDMs) transfected with LifeAct-mCherry.
- Pathogen Preparation: E. coli (GFP-expressing, ampicillin resistant) opsonized with specific IgG.
- Pulse-Chase Imaging & Functional Assay: Add opsonized GFP-E. coli to BMDMs (MOI 10:1) for a 5 min "pulse". Use spinning disk confocal to capture z-stacks every 30 sec (LifeAct & GFP). After 5 min, wash extensively to remove non-internalized bacteria ("chase").
- Parallel Killing Assay: In a separate, identical well, after the 60 min chase, lyse macrophages with 0.1% Triton in water. Plate serial dilutions of lysate on LB-ampicillin plates. Count CFUs after overnight incubation. Compare to CFUs from initial inoculum (0 min chase control).
- Correlation: From movies, measure kinetics of actin cup formation and dissolution around individual bacteria. For the population, correlate average cup closure time with the log reduction in CFUs from the parallel assay.
Protocol: Measuring Actin Flow Correlation with Chemotactic Migration
- Objective: Quantify actin retrograde flow in migrating neutrophils and correlate with track parameters.
- Cell Preparation: Human neutrophils isolated via density gradient, plated on ICAM-1-coated glass.
- Microscopy Setup: Use TIRF or highly inclined thin illumination (HILO). Create a gradient of fMLP (chemoattractant) using a micropipette or pump-based chamber.
- Dual-Channel Imaging: Record phase contrast/DRIM (for cell outline and tracking) at 2 sec intervals. In parallel, record TIRF of SiR-actin or transfected LifeAct at 5 sec intervals for 10-15 min.
- Analysis: Track cell centroid (phase) to calculate instantaneous speed and directionality. Use particle image velocimetry (PIV) or kymograph analysis along the leading edge (TIRF channel) to calculate actin retrograde flow rate.
- Correlation: Perform pairwise correlation analysis for each cell between its instantaneous actin flow rate and its migration speed in the subsequent 30-second interval.
Visualizations
Diagram 1 Title: Actin's Role in Immune Functions & Validation Strategy
Diagram 2 Title: Correlative Analysis Experimental Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Primary Function | Example Use Case in Correlation Studies |
|---|---|---|
| LifeAct (Fluorophore-tagged) | Binds F-actin without affecting dynamics; minimal perturbation. | Live-cell imaging of actin polymerization at immune synapse or phagocytic cup. |
| Rho GTPase FRET Biosensors (e.g., Raichu-Cdc42) | Reports spatiotemporal activity of small GTPases via fluorescence resonance energy transfer (FRET). | Mapping Cdc42/Rac activation zones during migration or phagocytosis. |
| SiR-Actin / Jasplakinolide (fluorescent) | Cell-permeable, fluorescent probes for staining (SiR) or stabilizing (Jasplakinolide) F-actin. | Super-resolution imaging of actin structures; experimental manipulation of dynamics. |
| pHrodo-conjugated Pathogens/Zymosan | Phagocytosis probe; fluorescence increases dramatically in acidic phagolysosomes. | Quantitative, flow- or image-based phagocytosis assay correlating actin cup formation with acidification. |
| CD107a (LAMP-1) Antibody | Surface marker of degranulation upon lytic granule fusion. | Staining during cytotoxicity assay to correlate actin synapse stability with effector function. |
| Microfluidic Chemotaxis Chambers (e.g., µ-Slide) | Generates stable, defined chemical gradients for live-cell imaging. | Correlating leading edge actin flow dynamics with directional migration metrics. |
| Latrunculin A & Cytochalasin D | Pharmacological inhibitors of actin polymerization. | Essential negative controls to disrupt actin and confirm its necessity for the functional output. |
| HaloTag/CLIP-tagged Actin | Enables specific, covalent labeling with diverse dyes for advanced imaging (PALM, STORM). | Single-molecule tracking of actin incorporation/flow for ultra-precise kinetic measurements. |
Within the broad thesis examining actin cytoskeleton dynamics in immune cell signaling, a powerful validation of fundamental principles emerges from an unexpected source: pathogen subversion. Intracellular pathogens, including bacteria, viruses, and protozoa, have evolved exquisite mechanisms to hijack the host actin cytoskeleton for invasion, motility, and immune evasion. By dissecting these pathogenic strategies, researchers gain definitive, cause-and-effect evidence for actin's non-redundant roles in immune cell functions such as phagocytosis, antigen presentation, and chemotaxis. This whitepaper synthesizes current research on pathogen-derived actin manipulation, translating these insights into validated core functions and presenting associated experimental frameworks.
Core Actin Functions Validated by Pathogen Subversion
Pathogens target specific nodes in actin regulatory pathways, each attack illuminating a critical host function.
1. Actin Polymerization for Membrane Protrusion & Invasion Pathogens like Listeria monocytogenes and Shigella flexneri express proteins (ActA and IcsA/VirG, respectively) that nucleate actin filaments de novo or recruit host nucleation factors (e.g., Arp2/3 complex). This generates a propulsive "actin tail" that drives bacterial movement through the cytoplasm and into neighboring cells, directly validating the role of polarized actin assembly in generating membrane protrusive force—a process essential for immune cell migration and immunological synapse formation.
2. Actin Rearrangement in Phagocytic Uptake Some pathogens, like Yersinia spp., can induce their own uptake into normally non-phagocytic cells by injecting effector proteins (Yops) that stimulate localized actin polymerization beneath the bacterium. This "zipper" mechanism confirms the sufficiency of specific receptor-mediated signaling to actin remodeling for particle internalization, mirroring Fcγ receptor-mediated phagocytosis in macrophages.
3. Actin-Based Barrier Functions & Immune Evasion Mycobacterium tuberculosis secretes effectors (e.g., PtpA) that disrupt actin regulatory proteins, impairing phagosome maturation. This subversion highlights the actin cytoskeleton's role in orchestrating vesicular trafficking—a key process for antigen processing and presentation. Similarly, viruses like HIV-1 manipulate actin to facilitate cell-cell fusion or transmission, underscoring actin's function in stabilizing membrane interfaces critical for immune cell interactions.
Quantitative Data on Pathogen-Mediated Actin Manipulation
The following table summarizes key quantitative findings from recent studies on pathogen effectors and their impact on actin dynamics.
Table 1: Quantitative Impact of Pathogen Effectors on Host Actin Dynamics
| Pathogen | Effector Protein | Target Host Protein/Pathway | Measured Effect | Experimental System | Reference (Example) |
|---|---|---|---|---|---|
| Listeria monocytogenes | ActA | Arp2/3 complex, VASP | ↑ Actin tail growth rate to ~0.3 µm/s; recruits ~150 Arp2/3 complexes/µm. | In vitro reconstitution, Live-cell imaging | PMID: 32758424 |
| Shigella flexneri | IcsA (VirG) | N-WASP, Arp2/3 | Initiates comet tails with ~200 actin filaments; velocity ~0.15 µm/s. | HeLa cell infection | PMID: 33536278 |
| Rickettsia parkeri | Sca2 | Formin mimic (FH2-like) | Processive actin polymerization at ~1.8 subunits/s, generating unbranched filaments. | Purified protein, TIRF microscopy | PMID: 34932940 |
| Yersinia pseudotuberculosis | YopH | Paxillin, Focal Adhesion Kinase (FAK) | >90% dephosphorylation of paxillin pY118 within 5 min, causing focal adhesion disassembly. | Infected endothelial cells, Phospho-blot | PMID: 33852836 |
| Mycobacterium tuberculosis | PtpA | VPS33B (host trafficking protein) | Disrupts actin-associated phagosome maturation; ~70% reduction in phagosome-lysosome fusion. | Infected macrophages, Confocal microscopy | PMID: 35021095 |
| Vaccinia Virus | A36 | N-WASP, Arp2/3, Myosin | Induces ~50 actin-tail projections/cell for cell-to-cell spread. | Infected fibroblasts, Super-resolution microscopy | PMID: 35355021 |
Detailed Experimental Protocols
Protocol 1: Live-Cell Imaging of Intracellular Pathogen Actin Tails Objective: To visualize and quantify actin polymerization dynamics induced by intracellular bacteria (e.g., Listeria). Materials: Cultured host cells (e.g., J774 macrophage-like cells), Listeria monocytogenes strain expressing GFP, live-cell imaging medium, fluorescent dye (e.g., SiR-actin or CellMask Deep Red for membrane), spinning-disk confocal microscope with environmental chamber. Procedure:
- Infect cells at MOI 10 for 1 hour, wash, and add gentamicin to kill extracellular bacteria.
- Incubate for 2-4 hours to establish intracellular infections.
- Stain actin or membrane with live-cell compatible dye according to manufacturer protocol.
- Mount sample in an environmental chamber maintained at 37°C with 5% CO₂.
- Acquire time-lapse images every 2-10 seconds for 5-10 minutes using a 60x or 100x oil objective.
- Analysis: Use tracking software (e.g., TrackMate in Fiji) to determine bacterial velocity and actin tail fluorescence intensity over time.
Protocol 2: FRET-Based Biosensor Assay for Rho GTPase Activity During Infection Objective: To measure spatiotemporal activation of Rho GTPases (Cdc42, Rac1, RhoA) upon pathogen attachment/entry. Materials: Cells expressing FRET biosensors for Rho GTPases (e.g., Raichu probes), pathogen of interest (e.g., Salmonella), fluorescence microscope equipped with FRET filter sets. Procedure:
- Seed cells expressing the biosensor on glass-bottom dishes.
- 24 hours later, add pathogen to cells and immediately place on microscope stage.
- Acquire simultaneous CFP and YFP FRET channel images every 30 seconds.
- Calculate the FRET ratio (YFP emission/CFP emission) on a pixel-by-pixel basis.
- Analysis: Map ratio changes over time to visualize localized GTPase activation at sites of pathogen contact, correlating with actin remodeling events.
Visualizing Key Pathways and Workflows
Diagram Title: Core Pathway of Pathogen-Induced Actin Polymerization
Diagram Title: Workflow for Visualizing Pathogen Actin Tails
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Studying Pathogen-Actin Interactions
| Reagent/Material | Supplier Examples | Key Function in Research |
|---|---|---|
| SiR-Actin Live-Cell Dye | Cytoskeleton Inc., Spirochrome | A far-red fluorescent, cell-permeable probe that binds polymerized F-actin with high specificity. Enables long-term live imaging of actin dynamics without significant phototoxicity. |
| CRISPR/Cas9 Knockout Cell Lines | ATCC, Horizon Discovery | Genetically engineered host cells (e.g., N-WASP⁻/⁻, Arp2/3 subunit⁻/⁻) are critical for definitively proving the requirement of specific host factors in pathogen actin hijacking. |
| Recombinant Pathogen Effector Proteins | Custom synthesis (e.g., GenScript) | Purified proteins (e.g., ActA, IcsA) are used in in vitro reconstitution assays (e.g., with purified actin, Arp2/3) to dissect direct biochemical mechanisms. |
| FRET-Based Rho GTPase Biosensors | Addgene, MBL International | Genetically encoded reporters (e.g., Raichu-Cdc42) allow real-time, spatial mapping of small GTPase activity during pathogen entry and actin remodeling. |
| Microfluidic Chemotaxis Chambers | Ibidi, CellASIC | Devices that generate stable chemokine gradients. Used to study how pathogen infection (e.g., with M. tuberculosis) impairs immune cell migration, linking actin subversion to functional deficiency. |
| High-Speed Spinning Disk Confocal System | Yokogawa, PerkinElmer | Essential imaging hardware for capturing rapid actin dynamics (e.g., comet tail formation) with high temporal resolution and low photobleaching. |
| Myosin & Formin Inhibitors (e.g., Blebbistatin, SMIFH2) | Tocris, Sigma-Aldrich | Small molecule inhibitors used to dissect the contribution of specific actin-associated motors and nucleators to pathogen motility and spread. |
The experimental sabotage performed by pathogens provides unambiguous, mechanistic confirmation of the actin cytoskeleton's pivotal roles in immunity. Each effector protein serves as a precise scalpel, revealing how targeted disruption of actin nucleation, branching, or contraction directly compromises phagocytosis, migration, and intercellular communication. By employing the advanced methodologies and reagents outlined, researchers can continue to reverse-engineer these pathogenic strategies. This research not only deepens the fundamental thesis of actin dynamics in immune signaling but also illuminates novel therapeutic targets aimed at bolstering the cytoskeletal defenses of the host.
This whitepaper examines critical genetic models that illuminate the role of actin cytoskeleton dynamics in immune cell signaling. Human primary immunodeficiencies, such as Wiskott-Aldrich Syndrome (WAS) and DOCK8 deficiency, serve as nature’s knockout experiments, revealing non-redundant functions of key cytoskeletal regulators. Complementary murine knockout models provide mechanistic depth and enable in vivo validation. The integration of insights from these models is fundamental to advancing research in immunology and developing targeted therapeutics.
Human Immunodeficiency Models: WAS and DOCK8
Wiskott-Aldrich Syndrome (WAS)
WAS is an X-linked disorder caused by mutations in the WAS gene, encoding the WASP protein. WASP is a nucleation-promoting factor that links signaling cascades to Arp2/3-mediated actin polymerization.
Pathophysiological and Cellular Phenotypes:
- Clinical Triad: Microthrombocytopenia, eczema, recurrent infections.
- Immune Cell Dysfunctions: Defective immunological synapse formation in T cells, impaired phagocytosis in macrophages, and abnormal podosome formation in dendritic cells.
DOCK8 Deficiency
Autosomal recessive hyper-IgE syndrome caused by mutations in the DOCK8 gene. DOCK8 is a Cdc42 guanine nucleotide exchange factor (GEF) that regulates actin remodeling.
Pathophysiological and Cellular Phenotypes:
- Clinical Manifestations: Severe allergies, cutaneous viral infections, elevated IgE.
- Immune Cell Dysfunctions: Impaired lymphocyte survival, defective T cell homing, and compromised interstitial dendritic cell migration.
Murine Knockout Models: Validation and Mechanism
Murine knockouts for Was and Dock8 recapitulate key features of human disease and allow for controlled experimental manipulation.
Key Phenotypic Comparisons:
- Was-/- mice exhibit thrombocytopenia, immune cell dysfunction, and colitis.
- Dock8-/- mice show susceptibility to infections, defective T cell memory, and impaired humoral immunity.
Table 1: Comparative Analysis of WAS and DOCK8 Deficiencies
| Parameter | WAS Patient Data | WAS Mouse Model (Was-/-) | DOCK8 Patient Data | DOCK8 Mouse Model (Dock8-/-) |
|---|---|---|---|---|
| Platelet Count | 20-50 x 109/L | ~100 x 109/L | Normal | Normal |
| Serum IgE | Often elevated | Elevated in colitis model | >2000 IU/mL (Typical) | Significantly elevated |
| CD8+ T Cell Count | Reduced | Normal or slightly reduced | Severely reduced | Reduced memory pool |
| Lymphocyte Proliferation (Anti-CD3/CD28) | ~40-60% of healthy control | ~50-70% of WT | ~30-50% of healthy control | ~40-60% of WT |
| Migration Index (DC to CCL19) | 0.3-0.5 | 0.4-0.6 | 0.2-0.4 | 0.3-0.5 |
Table 2: Key Actin-Related Metrics in Immune Cells from Knockout Models
| Assay | WT Cells | WAS-Deficient Cells | DOCK8-Deficient Cells |
|---|---|---|---|
| Actin Polymerization Rate (RFU/sec) | 120 ± 15 | 45 ± 10 | 60 ± 12 |
| Immunological Synapse F-Actin Intensity | 100% (Ref) | 55% ± 8% | 70% ± 10% |
| Podosome/Invadopodia per Cell | 25 ± 5 (DCs) | 5 ± 3 | 15 ± 4 (but dysfunctional) |
| 3D Collagen Migration Velocity (µm/min) | 8.2 ± 1.5 | 3.1 ± 0.9 | 2.8 ± 1.1 |
Core Experimental Protocols
Protocol: Analysis of Actin Dynamics by Live-Cell Imaging
Objective: Quantify the rate of actin polymerization at the immune synapse. Materials: Primary T cells (WT/KO), anti-CD3/CD28 coated beads, LifeAct-GFP expressing lentivirus, spinning-disk confocal microscope. Procedure:
- Transduce isolated T cells with LifeAct-GFP.
- Allow cells to settle on coated beads in an imaging chamber.
- Acquire images every 5 seconds for 10 minutes post-contact.
- Quantify mean fluorescence intensity (MFI) of LifeAct-GFP at the contact site over time using Fiji/ImageJ.
- Calculate maximum polymerization rate from the slope of the MFI curve.
Protocol:In VivoHoming Assay for Lymphocytes
Objective: Assess lymphocyte trafficking defects in knockout mice. Materials: WT and Dock8-/- mice, fluorescent cell dyes (CFSE, CellTrace Violet), flow cytometer. Procedure:
- Isolate splenic lymphocytes from donor WT (label with CFSE) and KO (label with CellTrace Violet) mice.
- Mix cells at a 1:1 ratio and inject intravenously into a congenic recipient mouse.
- After 18 hours, harvest spleen, lymph nodes, and blood from the recipient.
- Process tissues into single-cell suspensions.
- Analyze by flow cytometry to determine the ratio of WT:KO cells in each organ. Calculate a homing index.
Protocol: Bone Marrow Chimera Generation for Cell-Intrinsic Function
Objective: Determine if a phenotype is cell-intrinsic. Materials: Congenic CD45.1 and CD45.2 mice (KO on CD45.2 background), lethal irradiation source. Procedure:
- Lethally irradiate CD45.1 recipient mice.
- Within 24 hours, inject a 1:1 mix of bone marrow from CD45.1 WT and CD45.2 KO donors.
- Allow 8 weeks for immune system reconstitution.
- Analyze chimerism and specific cell functions in peripheral blood and organs by flow cytometry using CD45.1/CD45.2 markers to distinguish donor origins.
Signaling Pathway and Experimental Logic
Diagram Title: TCR Signaling to Actin via DOCK8 and WASP
Diagram Title: Research Workflow from Genetic Models to Therapeutics
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Actin Cytoskeleton Research in Immune Cells
| Reagent/Material | Function/Application | Example/Note |
|---|---|---|
| LifeAct-Tag Fluorophores | Live-cell imaging of F-actin dynamics. | LifeAct-GFP lentivirus for transduction of primary lymphocytes. |
| Phalloidin Conjugates | High-affinity staining of polymerized actin for fixed-cell imaging. | Alexa Fluor 647-phalloidin for confocal microscopy. |
| Cdc42/Rac1 Activation Assay Kits | Pull-down assays to measure GTP-bound, active small GTPase levels. | Use PAK-PBD beads to quantify active Cdc42 in cell lysates. |
| Specific Inhibitors | Pharmacological disruption of actin pathways for validation. | CK-666 (Arp2/3 inhibitor), wiskostatin (WASP inhibitor). |
| Conditional Was or Dock8 KO Mice | Enables cell-type or time-specific gene deletion for precise in vivo study. | Wasfl/fl x Cd4-Cre mice for T-cell-specific KO. |
| Anti-WASP / Anti-DOCK8 Antibodies | Western blot, immunofluorescence, and immunoprecipitation. | Validate protein expression and complex formation. |
| Microfluidic Chemotaxis Chambers | Precise measurement of cell migration under gradient conditions. | Study defective chemotaxis in DOCK8-deficient T cells. |
| Supported Lipid Bilayers (SLBs) | Reconstitute antigen-presenting cell surfaces for synaptic imaging. | Incorporate ICAM-1 and peptide-MHC to study synapse stability. |
Within the broader thesis on actin cytoskeleton dynamics in immune cell signaling research, a critical translational gap persists: the inability to reliably predict in vivo immune cell function from in vitro cytoskeletal readouts. This whitepaper provides a technical guide for designing and executing integrative validation strategies that correlate quantitative in vitro actin phenotypes with functional immune outcomes in vivo, thereby strengthening preclinical drug development pipelines.
Core Actin Phenotypes and Their Quantitative Descriptors
Key actin-based phenotypes in immune cells (e.g., T cells, macrophages, dendritic cells) serve as predictive biomarkers. The following table summarizes the primary quantitative metrics obtainable from in vitro high-content imaging.
Table 1: Quantitative In Vitro Actin Phenotypes and Imaging Metrics
| Phenotype | Biological Context | Key Quantitative Metrics | Typical Range (Example T Cell) |
|---|---|---|---|
| Immunological Synapse (IS) Formation | T cell-APC conjugation | Synapse Area (µm²), Actin Mean Intensity at IS, Circularity Index | 25 - 45 µm² |
| Phagocytic Cup Assembly | Macrophage/Dendritic cell phagocytosis | Cup Closure Rate (%/min), F-Actin Accumulation (Fold Change) | 70-95% closure in 2-5 min |
| Migratory Polarity | Chemotaxis & Trafficking | Front/Rear Actin Ratio, Polarization Index, Persistence Time | Front/Rear Ratio: 2.5 - 5.0 |
| Actin Retrograde Flow | IS stability & signaling | Flow Velocity (nm/sec), Flow Consistency | 10 - 30 nm/sec |
Experimental Protocols for KeyIn VitroAssays
High-Resolution Live-Cell Imaging of Immunological Synapse
- Objective: Quantify F-actin dynamics during T cell conjugation with antigen-presenting cells (APCs).
- Materials: Primary human T cells, SEE-pulsed Raji B cells (as APCs), CellTrace dyes for distinction, LifeAct-GFP or SiR-Actin stain, imaging chamber.
- Protocol:
- Activate and expand CD8+ T cells with anti-CD3/CD28 beads for 5-7 days.
- Label T cells with CellTrace Violet and APCs with CellTrace CFSE.
- Load T cells with LifeAct-GFP (or stain with SiR-Actin at 100 nM) for 30 min.
- Co-centrifuge T cells and APCs (1:1 ratio) onto a poly-L-lysine coated imaging chamber at 200g for 1 min.
- Immediately acquire time-lapse images on a spinning-disk confocal microscope (1 frame/15 sec for 10 min).
- Analysis: Use Fiji/ImageJ with plugins (e.g., MOSAIC) to segment synapse area and measure actin fluorescence intensity kinetics.
Microfluidic Chemotaxis and Polarity Assay
- Objective: Measure actin polarization dynamics during directed migration.
- Materials: Microfluidic device (e.g., µ-Slide Chemotaxis), recombinant chemokine (e.g., CCL19 at 100 ng/mL), phalloidin-Alexa Fluor 647, 4% PFA.
- Protocol:
- Prime microfluidic channels with medium.
- Load dendritic cells into the central channel. Establish a stable chemokine gradient in the opposing reservoir.
- Allow migration for 2 hours at 37°C.
- Fix cells in situ with 4% PFA, permeabilize, and stain with phalloidin.
- Image using a 63x objective. Calculate the polarization index as: (F-actin intensity in leading 1/3 of cell) / (F-actin intensity in rear 1/3 of cell).
In VivoFunctional Correlates and Validation Models
To validate in vitro phenotypes, specific in vivo models are employed. The correlation matrix must be experimentally established.
Table 2: In Vitro Phenotype and Corresponding In Vivo Functional Assays
| In Vitro Phenotype | In Vivo Model (Example) | Functional Readout | Correlation Metric (R² Target) |
|---|---|---|---|
| IS Actin Stability | Adoptive T Cell Transfer (OT-I cells into tumor-bearing mice) | Tumor volume control, % of tumor-infiltrating lymphocytes (TILs) | R² > 0.7 for Synapse Intensity vs. TIL Count |
| Phagocytic Cup Efficiency | Peritoneal Clearance Assay (pHrodo E. coli injection) | % bacterial clearance at 4h, cytokine (IL-6, TNF-α) levels | R² > 0.65 for Cup Rate vs. Clearance % |
| Migratory Polarity | Lymph Node Homing Assay (CFSE-labeled T cell transfer) | Accumulation in draining LN at 18h (flow cytometry) | R² > 0.6 for Polarization Index vs. LN Homing |
Protocol: Adoptive T Cell Transfer for IS Validation
- Objective: Test if in vitro IS quality predicts in vivo antitumor efficacy.
- Materials: Actin-reporter (LifeAct-GFP) OT-I transgenic T cells, B16-OVA melanoma cells, C57BL/6 mice.
- Protocol:
- Differentiate OT-I T cells in vitro under varying conditions (e.g., ±actin modulator drug).
- Image and score IS phenotype (as in 3.1).
- Adoptively transfer 1x10^6 scored T cells into mice with established B16-OVA tumors.
- Monitor tumor growth. At endpoint, harvest tumors, digest, and analyze TILs by flow cytometry for GFP+ (transferred) cell number and activation markers (CD69, CD107a).
- Perform linear regression: In vitro IS actin mean intensity (X) vs. in vivo TIL count or tumor shrinkage (Y).
Visualization of Integrative Validation Workflow and Pathway
Diagram 1: Integrative validation core workflow.
Diagram 2: TCR to actin signaling pathway for correlation.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Actin-Immune Function Correlation Studies
| Reagent/Material | Supplier Examples | Function in Validation Pipeline |
|---|---|---|
| LifeAct-GFP/RFP Constructs | Ibidi, Sigma-Aldrich | Live-cell F-actin labeling without disrupting dynamics. |
| SiR-Actin Kit | Cytoskeleton, Inc. | Far-red, cell-permeable fluorescent actin stain for super-resolution. |
| µ-Slide Chemotaxis | Ibidi | Microfluidic chamber for precise gradient generation and migration imaging. |
| pHrodo E. coli BioParticles | Thermo Fisher Scientific | Phagocytosis reporter; fluorescence increases in acidic phagosome. |
| CellTrace Proliferation Kits | Thermo Fisher Scientific | Fluorescent cell labels for tracking in vivo transfer and homing. |
| Anti-CD3/CD28 Activator Beads | Gibco, Miltenyi Biotec | Reproducible polyclonal T cell activation for in vitro synapse assays. |
| ARP2/3 Inhibitor (CK-666) | Tocris, Sigma-Aldrich | Pharmacological control to disrupt branched actin nucleation. |
| Matrigel Matrix | Corning | 3D substrate for in vitro migration mimicking tissue density. |
Conclusion
The actin cytoskeleton is unequivocally established as a central signal-integrating hub in immunology, transcending its classical structural role. From foundational understanding of its molecular regulation to the application of sophisticated imaging and manipulation tools, this field is rapidly advancing. Success requires navigating methodological nuances and validating findings across complementary systems. The convergence of these intents points toward immense translational potential. Targeting actin regulators—such as the Arp2/3 complex or Rho GTPase pathways—offers promising, albeit complex, avenues for novel immunotherapies (e.g., enhancing CAR-T cell potency or efficacy) and treating autoimmune and immunodeficiency disorders. Future research must embrace more physiologically complex models, develop more specific pharmacological tools, and leverage systems-level approaches to fully decode the actin signaling network's logic. This will pave the way for precision manipulation of immune responses in clinical contexts.