ARPC1B vs WASP Deficiency: Comparative Molecular Mechanisms in Actin Polymerization and Immunodeficiency
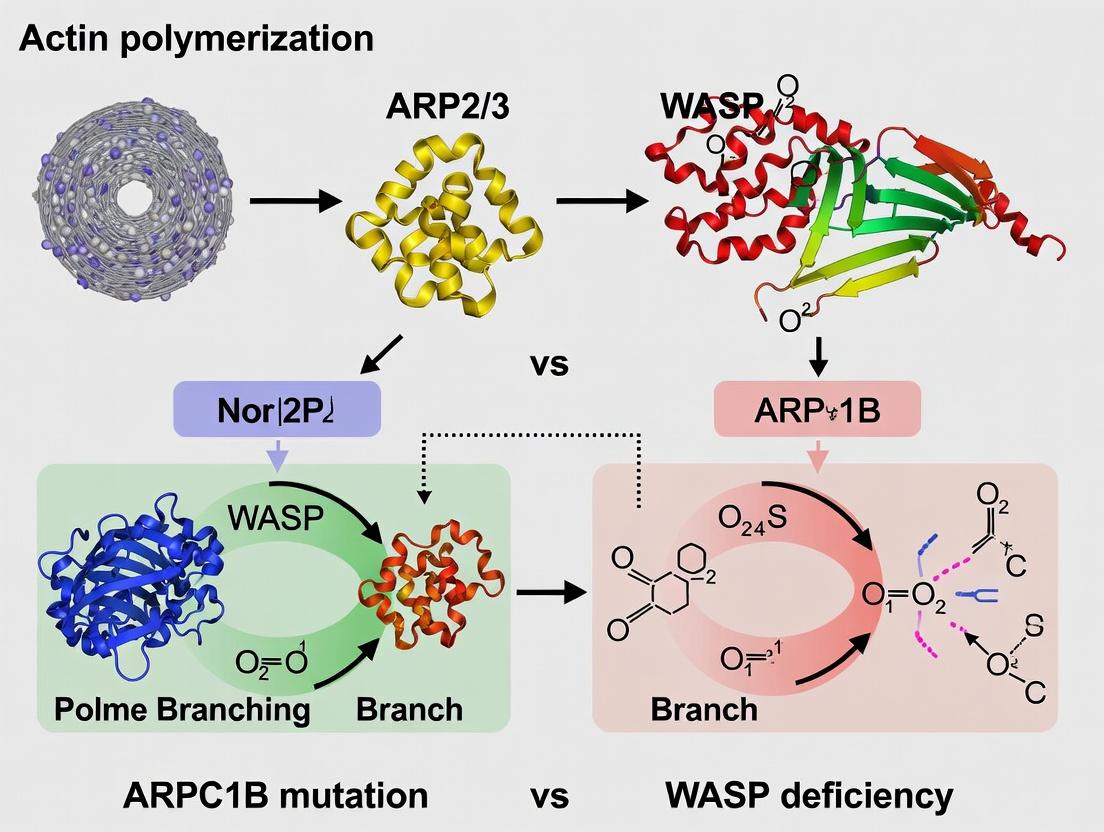
This article provides a comprehensive comparative analysis of two critical genetic defects leading to actin-related immunodeficiency: ARPC1B and WASP deficiencies.
ARPC1B vs WASP Deficiency: Comparative Molecular Mechanisms in Actin Polymerization and Immunodeficiency
Abstract
This article provides a comprehensive comparative analysis of two critical genetic defects leading to actin-related immunodeficiency: ARPC1B and WASP deficiencies. Targeting researchers, scientists, and drug development professionals, we explore the foundational molecular biology, contrasting the roles of the ARP2/3 complex subunit ARPC1B and the nucleation-promoting factor WASP in actin cytoskeleton dynamics. We detail current methodologies for modeling and diagnosing these disorders, highlight common experimental challenges and optimization strategies in studying actin polymerization defects, and validate key findings through direct comparative analysis of clinical phenotypes, cellular assays, and omics data. The synthesis aims to inform targeted therapeutic development and precision medicine approaches for these rare but instructive immunodeficiencies.
Molecular Foundations of Actin Cytoskeleton Defects: ARPC1B and WASP Roles Explained
Publish Comparison Guide: ARPC1B Mutation vs. WASP Deficiency in Immune Cell Actin Dynamics
The following guide provides an objective, data-driven comparison of two critical genetic disruptions in actin polymerization pathways—ARPC1B mutation and WASP deficiency—framed within the broader thesis of their distinct mechanistic impacts on immune cell function and disease pathology.
Comparison Table 1: Core Molecular & Clinical Features
| Feature | ARPC1B Deficiency | WASP Deficiency (Wiskott-Aldrich Syndrome) |
|---|---|---|
| Defective Protein | Subunit of ARP2/3 complex (nucleation core) | Nucleation Promoting Factor (NPF) for ARP2/3 complex |
| Primary Molecular Consequence | Impaired stability and nucleation activity of the ARP2/3 complex. | Loss of ARP2/3 complex activation and actin filament branching. |
| Inheritance | Autosomal Recessive | X-Linked Recessive |
| Key Immune Cell Defects | Severe defects in lamellipodia formation, T cell spreading, and immunological synapse stability. | Defects in filopodia formation, podosome assembly, and immune cell migration. |
| Hallmark Clinical Manifestations | Severe combined immunodeficiency (SCID)-like presentation, recurrent infections, bleeding diathesis, vasculitis. | Eczema, microthrombocytopenia, recurrent infections, autoimmunity, lymphoma risk. |
| Platelet Morphology & Count | Normal count, enlarged platelets. | Severely low count, small platelets (microthrombocytopenia). |
Comparison Table 2: Quantitative In Vitro Actin Polymerization Assays
Data compiled from recent pyrene-actin polymerization and TIRF microscopy studies.
| Assay Parameter | Wild-Type (Control) | ARPC1B-Deficient Cells | WASP-Deficient Cells |
|---|---|---|---|
| Nucleation Rate (A.U./min) | 1.00 ± 0.12 | 0.25 ± 0.08 (75% reduction) | 0.85 ± 0.10 (15% reduction) |
| Maximal Polymerization Velocity | 1.00 ± 0.09 | 0.30 ± 0.07 (70% reduction) | 0.60 ± 0.08 (40% reduction) |
| Filament Branch Angle (degrees) | 70 ± 5 | Indeterminate (few branches) | 68 ± 6 (normal angle, fewer branches) |
| Podosome Lifetime (seconds) | 450 ± 50 | < 100 | ~200 |
| Lamellipodial Protrusion Rate (µm/min) | 10.2 ± 1.5 | 2.1 ± 0.8 | 8.5 ± 1.2 |
Experimental Protocols for Key Cited Assays
Protocol 1: Pyrene-Actin Polymerization Assay for Nucleation Efficiency
- Reagent Prep: Prepare G-actin from rabbit muscle, label a portion with pyrene iodoacetamide. Mix unlabeled and pyrene-labeled actin (10% labeled) in G-buffer (2 mM Tris-HCl pH 8.0, 0.2 mM ATP, 0.5 mM DTT, 0.1 mM CaCl₂).
- Sample Prep: Isolate cytosolic extracts from patient-derived T-cells (ARPC1B-/-, WASP-/-) and isogenic corrected controls.
- Initiation: In a fluorometer cuvette, mix 2 µM pyrene-actin mix with cell extract (final protein concentration 1 mg/mL) in polymerization buffer (final: 50 mM KCl, 1 mM MgCl₂, 1 mM EGTA, 0.2 mM ATP, 10 mM imidazole pH 7.0).
- Measurement: Immediately transfer to a spectrofluorometer (ex: 365 nm, em: 407 nm). Record fluorescence every 2 seconds for 600-1200 seconds.
- Analysis: Normalize curves, calculate maximum slope (polymerization velocity) and time to half-maximal polymerization (nucleation lag time).
Protocol 2: Total Internal Reflection Fluorescence (TIRF) Microscopy of Single Filament Dynamics
- Flow Chamber Preparation: Create a passivated flow chamber using PEG-silane and biotin-PEG-silane on a glass coverslip.
- Surface Functionalization: Sequentially flow in NeutrAvidin, then biotinylated anti-His antibody.
- Protein Immobilization: Flow in His-tagged N-WASP (for WASP-deficient study) or His-tagged ARP2/3 complex (for ARPC1B study) to create nucleation sites.
- Polymerization Mix: Prepare a mix containing 1 µM G-actin (20% Alexa Fluor 488-labeled), 50 nM purified ARP2/3 complex (if not immobilized), and 100 nM GST-tagged WASP-VCA domain (or other NPF) in TIRF buffer (10 mM imidazole pH 7.4, 50 mM KCl, 1 mM MgCl₂, 1 mM EGTA, 0.2 mM ATP, 50 mM DTT, 0.5% methylcellulose, oxygen scavengers).
- Imaging: Flow mix into chamber and immediately image on a TIRF microscope with EM-CCD camera. Acquire frames every 3-5 seconds for 10 minutes.
- Analysis: Use FIJI/ImageJ to kymograph analysis to measure filament elongation rate, branching frequency, and filament lifetime.
Signaling Pathway Diagram: Actin Nucleation in Immune Cell Motility
Title: Actin Nucleation Pathway and Deficiency Blocks
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function in Actin Polymerization Research | Example/Catalog |
|---|---|---|
| Pyrene-labeled Actin | Fluorophore-conjugated actin; increase in fluorescence upon polymerization allows kinetic measurement of bulk actin dynamics. | Cytoskeleton Inc. #AP05; Hypermol #ALF-250. |
| Recombinant ARP2/3 Complex | Purified complex for in vitro reconstitution assays to dissect specific subunit (e.g., ARPC1B) contributions. | Cytoskeleton Inc. #RP01; Custom purified via Sf9/baculovirus. |
| Recombinant WASP/N-WASP/VCA domains | Purified nucleation-promoting factors to activate ARP2/3 complex in controlled settings. | Abcam #ab206990; Sigma #SRP6018. |
| Latrunculin A | Binds G-actin, preventing polymerization. Essential negative control for actin-dependent processes. | Tocris Bioscience #3973. |
| Jasplakinolide | Stabilizes actin filaments, promotes polymerization. Used as a positive control or to study filament stability. | Thermo Fisher Scientific #J7473. |
| TIRF Microscope System | Enables visualization of single actin filament dynamics near the cell/substrate interface with high signal-to-noise. | Nikon N-STORM; Olympus CellTIRF. |
| Actin Live-Cell Probes (SiR-actin, LifeAct) | Cell-permeable fluorescent probes for real-time visualization of actin dynamics in living immune cells. | Cytoskeleton Inc. #CY-SC001; Chromotek #LF010. |
| CRISPR/Cas9 Kits for Immune Cells | For generating isogenic knockout cell lines (e.g., in Jurkat T-cells or iPSC-derived macrophages) of ARPC1B or WASP. | Synthego Knockout Kit; Edit-R CRISPR-Cas9. |
This comparison guide is framed within the thesis context of contrasting ARPC1B deficiency with WASP (Wiskott-Aldrich Syndrome protein) deficiency in actin polymerization. While both impair actin nucleation, they represent distinct molecular lesions—one in the core nucleator complex (ARP2/3) and one in a key activator (WASP)—with overlapping yet divergent cellular and clinical phenotypes. This guide objectively compares the functional performance of the canonical ARP2/3 complex containing the ARPC1A subunit versus the variant containing ARPC1B, and situates this within the broader comparison to WASP-deficient systems.
Comparative Analysis: ARPC1A vs. ARPC1B Containing ARP2/3 Complexes
Table 1: Biochemical & Functional Comparison of ARPC1 Subunit Variants
| Feature/Aspect | ARP2/3 Complex with ARPC1A | ARP2/3 Complex with ARPC1B | Supporting Experimental Data & Key References |
|---|---|---|---|
| Expression Pattern | Ubiquitous; predominant in most cell types. | Hematopoietic lineage cells (platelets, leukocytes), endothelial cells. | RNA-seq data from Human Protein Atlas; Immunoblots of cell lineages. |
| Core Structural Role | Essential structural subunit; part of ARP2/3 scaffold. | Essential structural subunit; part of ARP2/3 scaffold. | Cryo-EM structures (e.g., PDB 6P2V) show analogous positioning. |
| Binding to WASP/N-WASP | Binds VCA domain of activators, promoting nucleation. | Enhanced binding affinity for the VCA domain of WASP. | Pull-down assays & Surface Plasmon Resonance show ~2-3x higher affinity for WASP-VCA (KD ~0.5 µM vs. ~1.2 µM for ARPC1A). |
| Nucleation Efficiency (WASP-stimulated) | Standard nucleation rate. | Higher nucleation rate when activated by hematopoietic WASP. | Pyrene-actin polymerization assays show ~25-40% higher initial rate with WASP-VCA. |
| Phenotype of Genetic Loss in Humans | Embryonic lethal (inferred from model organisms). | ARPC1B deficiency: combined immunodeficiency, thrombocytopenia, eosinophilia, autoimmune manifestations. | Clinical cohort studies (N=~50 patients) report recurrent infections, bleeding, vasculitis. |
| Phenotype of Loss in Cell Models | Not viable in most mammalian cells. | In ARPC1B-KO leukocytes: Reduced lamellipodia stability, impaired immune synapse actin density, defective phagocytosis. | Live-cell TIRF microscopy shows ~60% reduction in persistent lamellipodial protrusions in migrating dendritic cells. |
Comparison to WASP Deficiency
Table 2: ARPC1B Deficiency vs. WASP Deficiency (Wiskott-Aldrich Syndrome)
| Parameter | ARPC1B Deficiency | WASP Deficiency (Classical WAS) | Thesis Context Implication |
|---|---|---|---|
| Molecular Lesion | Loss of one ARP2/3 complex subtype, primarily affecting WASP-ARP2/3 signaling node. | Loss of nucleation-promoting factor (NPF), affecting multiple ARP2/3 & other actin effectors. | ARPC1B loss is pathway-specific (ARP2/3); WASP loss is activator-specific with broader signaling ripple effects. |
| Actin Polymerization in Cell Extracts | Reduced but not absent. Residual ARPC1A-complexes can be activated by other NPFs (e.g., N-WASP, SCAR/WAVE). | Severely reduced upon immunoreceptor (e.g., TCR) stimulation, but some NPF-independent polymerization remains. | Highlights the non-redundant synergy between WASP and the ARPC1B-complex in hematopoietic cells. |
| Clinical Immunodeficiency | Recurrent infections, defective T cell migration. | Recurrent infections, defective T cell activation and migration. | Overlapping phenotype confirms the critical axis of WASP->ARPC1B-ARP2/3 for immune cell function. |
| Thrombocytopenia & Platelet Defects | Present; small platelets. | Present; severely small platelets. | Suggests ARPC1B-independent roles for WASP in platelet biogenesis (e.g., vesicular trafficking). |
| Autoinflammation & Vasculitis | Very High Prevalence (~70%). | Lower prevalence, typically linked to autoimmunity. | Points to a unique, non-redundant role for ARPC1B in endothelial or leukocyte pathways controlling vascular inflammation. |
| Therapeutic: Hematopoietic Stem Cell Transplant (HSCT) | Curative, but inflammation may persist. | Curative. | Suggests ARPC1B-related inflammatory disease may involve radio-resistant stromal/endothelial cells. |
Key Experimental Protocols
1. Pyrene-Actin Polymerization Assay for Nucleation Efficiency
- Purpose: Quantify the nucleation activity of ARP2/3 complexes in vitro.
- Protocol: Prepare G-actin (10% pyrene-labeled) in G-buffer (2 mM Tris pH 8.0, 0.2 mM CaCl2, 0.2 mM ATP, 0.5 mM DTT). Initiate polymerization by adding 1/10 volume of 10X KMEI buffer (500 mM KCl, 10 mM MgCl2, 10 mM EGTA, 100 mM Imidazole pH 7.0) to a mix containing G-actin (2-4 µM final), purified ARP2/3 complex (10-50 nM), and an activating VCA peptide (50-200 nM). Monitor fluorescence (ex: 365 nm, em: 407 nm) in a plate reader at 25°C for 600-1800 seconds. The initial slope (first ~60-120s) is used as the nucleation rate.
2. Co-immunoprecipitation for ARPC1B-WASP Interaction
- Purpose: Validate physical interaction in a cellular context.
- Protocol: Transfert HEK293T cells with plasmids encoding tagged WASP and ARPC1B. Lyse cells 48h post-transfection in NP-40 lysis buffer (with protease inhibitors). Pre-clear lysate, then incubate with anti-tag antibody-conjugated beads for 2h at 4°C. Wash beads 3x with lysis buffer, elute protein with 2X Laemmli buffer, and analyze by immunoblotting for both tags.
3. TIRF Microscopy for Lamellipodia Dynamics
- Purpose: Visualize and quantify actin dynamics at the cell membrane.
- Protocol: Plate control or ARPC1B-deficient (CRISPR/Cas9) dendritic cells on fibronectin-coated glass-bottom dishes. Transfect with Lifect-mACT (F-tractin) to label F-actin. Image using a 488nm laser under TIRF illumination at 2-5 second intervals for 5-10 minutes. Quantify lamellipodia persistence (duration of protrusion >0.5 µm/min) and retraction rates using tracking software (e.g., Fiji/TrackMate).
Visualizations
Title: WASP Activation of ARPC1B-ARP2/3 Leads to Actin Nucleation
Title: Phenotype Comparison: ARPC1B vs WASP Deficiency
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for ARPC1B/WASP Actin Polymerization Research
| Reagent/Solution | Function in Research | Example Application |
|---|---|---|
| Recombinant Human ARP2/3 Complex (with defined ARPC1 subunit) | In vitro biochemical analysis of complex-specific nucleation kinetics and activator binding. | Pyrene-actin assays comparing ARPC1A- vs. ARPC1B-complex activity. |
| WASP/N-WASP VCA Domain Peptide/Protein | Minimal ARP2/3 activating fragment used to stimulate nucleation in controlled experiments. | Determining maximal activation potential of purified ARP2/3 complexes. |
| Pyrene-Labeled Actin (Cytoskeleton Inc.) | Fluorescent actin derivative used to monitor polymerization kinetics in real-time. | Core component of in vitro nucleation and polymerization rate assays. |
| Anti-ARPC1B Specific Antibody (Validated for KO) | Detect ARPC1B protein expression; confirm knockout/knockdown in cell models. | Immunoblotting, immunofluorescence in patient-derived or engineered cells. |
| Lifect-mACT (F-tractin) Plasmid | Genetically encoded F-actin biosensor for live-cell imaging with minimal perturbation. | TIRF microscopy of lamellipodia and podosome dynamics in live cells. |
| Cdc42/Rac1 Activation Assay Kits (G-LISA) | Quantify activation levels of upstream Rho GTPases that regulate WASP/ARP2/3. | Determine if signaling defects are upstream or intrinsic to the WASP-ARP2/3 axis. |
| ARPC1B-deficient iPSC Lines | Model human disease and differentiate into affected cell types (platelets, leukocytes). | Study cell-type specific phenotypes and test genetic rescue strategies. |
Comparative Analysis: WASP vs. Alternative NPFs in Actin Polymerization
Within the context of comparing ARPC1B mutation and WASP deficiency, understanding the specific role and efficacy of WASP against other Nucleation-Promoting Factors (NPFs) is critical. The following guide compares the performance of WASP-family proteins with other major NPFs in key functional assays.
Table 1: NPF Performance in In Vitro Actin Polymerization Assays
| NPF | Arp2/3 Complex Binding Affinity (Kd) | Pyrene-Actin Polymerization Rate (Fold Increase over Basal) | Branch Point Angle (Mean ± SD) | Critical Concentration for Branch Formation (nM) | Reference |
|---|---|---|---|---|---|
| WASP/N-WASP (Active) | 0.1 - 0.3 µM | 25 - 35x | 77° ± 10° | ~50 | (Rodal et al., 2005; Marchand et al., 2001) |
| WAVE/Scar (Active) | 0.2 - 0.5 µM | 20 - 30x | 78° ± 12° | ~60 | (Life et al., 2022; Chen et al., 2010) |
| WHAMM | ~0.8 µM | 10 - 15x | 75° ± 15° | ~150 | (Campellone et al., 2008) |
| WASH | 0.3 - 0.6 µM | 15 - 20x | 72° ± 8° | ~100 | (Liu et al., 2009) |
| JMY | 0.5 - 1.0 µM | 18 - 25x | 70° ± 10° | ~80 | (Zuchero et al., 2009) |
Interpretation in Disease Context: ARPC1B mutations often lead to reduced but not abolished Arp2/3 activity. In such hypomorphic states, the high-efficiency nucleation by WASP becomes disproportionately critical compared to other NPFs, explaining the partially overlapping but distinct immunodeficiencies between ARPC1B and WAS patients.
Table 2: Cellular Phenotype Rescue in WASP-Deficient Cells
| NPF Transfected into WASP-/- Cells | Podosome/Invadopodia Reformation Efficiency (%) | Phagocytosis Rate (% of WT) | Random Cell Migration Speed (µm/min) | Reference |
|---|---|---|---|---|
| Full-length WASP | 95 ± 5 | 98 ± 10 | 1.2 ± 0.3 | (Snapper et al., 2001; Jones et al., 2013) |
| Constitutively Active WASP (ΔVCA) | 110 ± 15 | 85 ± 12 | 1.8 ± 0.4* | (Cory et al., 2002) |
| N-WASP | 80 ± 10 | 75 ± 15 | 1.0 ± 0.2 | (Sasahara et al., 2002) |
| WAVE2 | 30 ± 10 | 40 ± 10 | 1.5 ± 0.3* | (Lebensohn & Kirschner, 2009) |
Note: Increased speed often correlated with less persistent directionality.
Experimental Protocols for Key Comparisons
Protocol 1: Pyrene-Actin Polymerization Assay for NPF Potency
Objective: Quantify the nucleation-promoting activity of WASP versus other NPFs.
- Reagent Preparation: Prepare G-actin (10% pyrene-labeled) in G-buffer (2 mM Tris-HCl pH 8.0, 0.2 mM CaCl₂, 0.2 mM ATP, 0.5 mM DTT). Pre-incubate purified Arp2/3 complex (10 nM) with or without the NPF of interest (WASP, WAVE, etc.) at varying concentrations (0-100 nM) for 2 minutes on ice.
- Initiation: Mix 2 µL of the Arp2/3/NPF complex with 98 µL of G-actin (1 µM final) in F-buffer (10 mM Tris-HCl pH 7.5, 50 mM KCl, 2 mM MgCl₂, 1 mM ATP, 1 mM DTT, 0.2 mM EGTA) directly in a quartz cuvette.
- Measurement: Immediately place the cuvette in a fluorescence spectrophotometer (ex: 365 nm, em: 407 nm). Record the increase in pyrene fluorescence (indicative of F-actin formation) every 2 seconds for 30 minutes.
- Analysis: Calculate the initial polymerization rate (slope of the linear phase) for each condition. Normalize to the rate with Arp2/3 alone (basal) to determine fold increase.
Protocol 2: TIRF Microscopy for Branching Kinetics and Architecture
Objective: Visualize and quantify actin network architecture nucleated by different NPFs.
- Flow Chamber Preparation: Create a passivated flow chamber using methoxy-PEG-silane. Introduce 0.2 µM neutravidin, followed by biotinylated anti-GST antibody to create a functionalized surface.
- Reaction Mixture: Prepare a motility mix containing: 1.5 µM G-actin (20% Alexa Fluor 488-labeled), 50 nM Arp2/3 complex, 50 nM GST-tagged NPF (WASP, WHAMM, etc.), 1 µM profilin, and an oxygen scavenging system (0.25 mg/mL glucose oxidase, 0.045 mg/mL catalase, 4.5 mg/mL glucose) in TIRF buffer (10 mM Imidazole pH 7.4, 50 mM KCl, 1 mM MgCl₂, 1 mM EGTA, 0.2 mM ATP, 10 mM DTT).
- Imaging: Inject the mixture into the flow chamber and immediately image on a TIRF microscope using a 100x/1.49 NA objective. Acquire frames every 5 seconds for 10 minutes.
- Quantification: Use software (e.g., FilamentAnalyzer, ComDet) to quantify branch density (branches/µm²), branch lifetime, and branch angle distribution from time-lapse sequences.
Visualizing WASP Regulation and Activation Pathways
Diagram Title: WASP Activation Pathway from Autoinhibition to Actin Nucleation
Diagram Title: Experimental Workflow for Comparing ARPC1B Mutation and WASP Deficiency
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function in WASP/Actin Research | Key Providers/Examples |
|---|---|---|
| Purified Recombinant WASP/NPF Proteins (Full-length & truncated mutants like ΔVCA, CA) | Essential for in vitro biochemical assays (pyrene-actin, TIRF) to dissect structure-function relationships. | Cytoskeleton Inc., Sino Biological, custom expression from academic cores. |
| Pyrene-labeled Actin (≥95% purity) | Fluorescent reporter for kinetic measurements of actin polymerization rates in spectrophotometer-based assays. | Cytoskeleton Inc., Hypermol. |
| Purified Arp2/3 Complex (from bovine brain or recombinant) | The core nucleation machinery; required for all NPF activity assays. Critical for testing ARPC1B mutant complexes. | Cytoskeleton Inc., custom purification (Spider, WASP assays). |
| Anti-WASP / Anti-Arp2/3 Antibodies (for WB, IP, IF) | For validating protein expression, localization, and complex formation in cellular contexts. | Cell Signaling Technology, Abcam, Santa Cruz Biotechnology. |
| TIRF Microscope System with high-sensitivity EMCCD/sCMOS camera | Enables visualization of single actin filament dynamics and branching events nucleated by NPFs in reconstituted or cellular systems. | Nikon, Olympus, Zeiss, Andor systems. |
| ARPC1B-mutant Cell Lines (e.g., CRISPR-edited Jurkat or HL-60) | Models for studying the specific effects of Arp2/3 complex hypomorphism, distinct from WASP deficiency. | Available via academic collaborations (e.g., from Klein lab). |
| WASP-deficient Cell Lines (e.g., myeloid lines from WAS patients or KO models) | Essential for rescue experiments and defining WASP-specific functions. | ATCC (e.g., THP-1 derivatives), primary cells from patients. |
| PIP₂-containing Lipid Vesicles (e.g., 70% PC, 30% PIP₂) | To reconstitute membrane-dependent activation of WASP in vitro. | Avanti Polar Lipids, Echelon Biosciences. |
| Active GTPase Proteins (e.g., GST-Cdc42-GTPγS) | To trigger the canonical activation pathway of WASP in controlled experiments. | Cytoskeleton Inc., Thermo Fisher. |
This comparison guide evaluates two critical monogenic defects disrupting actin cytoskeletal dynamics: ARPC1B deficiency and WASP deficiency. Within actin polymerization research, these mutations serve as natural "knockout" models to dissect the roles of the Arp2/3 complex nucleator versus its key activator. The comparative analysis of their cellular, immunological, and clinical phenotypes provides foundational data for target validation and therapeutic development.
Table 1: Genetic, Molecular, and Clinical Profile Comparison
| Parameter | ARPC1B Deficiency | WASP Deficiency (WAS/XLT) |
|---|---|---|
| Gene & Protein | ARPC1B, component of Arp2/3 complex | WAS, WASP (Wiskott-Aldrich syndrome protein) |
| Primary Molecular Function | Structural subunit of the Arp2/3 complex; essential for its stability and nucleating activity. | Actin nucleation promoting factor (NPF); activates Arp2/3 complex upon signaling (e.g., via Cdc42). |
| Actin Polymerization Defect | Severe reduction in Arp2/3-mediated branching nucleation. Lamellipodia formation is abolished. | Delayed and aberrant actin polymerization. Lamellipodia are formed but are disorganized. |
| Key Cellular Readouts | >70% reduction in actin patches in platelets (via phalloidin staining). Absent podosomes in dendritic cells. | ~50-60% reduction in actin polymerization rate (FRAP/TIRF). Small, unstable podosomes. |
| Immune Phenotype | Combined immunodeficiency, severe autoinflammation, vasculitis, thrombocytopenia. | Classic WAS: Immunodeficiency, eczema, microthrombocytopenia. XLT: primarily thrombocytopenia. |
| Supporting Experimental Data | Volpi et al. (2019) Blood: Platelet spreading area reduced by ~85% on fibrinogen. | Thrasher et al. (2021) J Clin Immunol: T-cell chemotaxis velocity decreased by ~40%. |
Table 2: In Vitro Actin Polymerization Assay Data
| Assay Condition | Wild-Type Control | ARPC1B-Deficient Cells | WASP-Deficient Cells |
|---|---|---|---|
| Pyrene Actin Polymerization (Lag Time) | 120 ± 15 sec | 300 ± 45 sec (150% increase) | 180 ± 20 sec (50% increase) |
| Branched Network Density (TIRF Microscopy) | 28 ± 3 branches/µm² | 3 ± 1 branches/µm² (~90% reduction) | 15 ± 2 branches/µm² (~45% reduction) |
| Podosome/Invadosome Stability | >30 min lifetime | Not formed | <10 min lifetime |
Experimental Protocols
1. Pyrene Actin Polymerization Assay (In Vitro)
- Purpose: Quantify kinetics of actin filament nucleation and elongation.
- Protocol: Prepare purified actin (10% pyrene-labeled). For WASP studies, mix purified WASP/Arp2/3 complex with actin in polymerization buffer (containing Mg²⁺ and ATP). For ARPC1B studies, use lysates from patient-derived T-cells or fibroblasts as the Arp2/3 source. Transfer to a quartz cuvette in a spectrofluorometer. Initiate polymerization by adding KCl and MgCl₂ to final 50 mM and 2 mM, respectively. Monitor fluorescence (ex: 365 nm, em: 407 nm) every 5-10 sec for 1 hour. Calculate lag time and maximum slope.
2. Platelet Spreading on Fibrinogen
- Purpose: Assess integrin-mediated actin cytoskeleton remodeling.
- Protocol: Isolate platelets from patient and control blood. Seed on fibrinogen (100 µg/ml)-coated glass coverslips in Tyrode’s buffer at 37°C. Allow spreading for 45-60 min. Fix with 4% PFA, permeabilize with 0.1% Triton X-100, and stain with Alexa Fluor 488-phalloidin (F-actin) and an antibody against vinculin. Image using a 63x/1.4 NA oil objective on a confocal microscope. Quantify total spread area using ImageJ software (n>50 cells/condition).
Visualizations
Diagram 1: Actin Polymerization Signaling Pathway
Diagram 2: Comparative Research Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Actin Dysfunction Research
| Reagent/Material | Function in Experiment | Example Vendor/Cat. No. |
|---|---|---|
| Purified Non-Muscle Actin (e.g., human, bovine) | Substrate for in vitro polymerization assays. Can be labeled (pyrene, rhodamine). | Cytoskeleton Inc. (APHL99) |
| Recombinant Human Arp2/3 Complex | Positive control nucleator for assays using patient cell lysates. | Cytoskeleton Inc. (RP01P) |
| Recombinant Human WASP (full-length or VCA domain) | Positive control NPF for WASP-specific rescue/activation experiments. | Sino Biological (10339-H07E) |
| Alexa Fluor 488/568/647 Phalloidin | High-affinity stain for polymerized F-actin in fixed cells for microscopy. | Thermo Fisher Scientific (A12379, A22283) |
| Cell-Based ARPC1B or WAS Knockdown/KO Lines | Isogenic control lines (e.g., CRISPR-Cas9 generated) for clean phenotype comparison. | Available via ATCC or academic repositories. |
| Anti-ARPC1B & Anti-WASP Antibodies (validated for WB, IF) | Detection of protein expression and localization in patient-derived cells. | Abcam (ab183337 - ARPC1B); Santa Cruz (sc-13139 - WASP) |
| μ-Slide Chemotaxis (Ibidi) | Precision chambers for standardized cell migration and chemotaxis assays. | Ibidi (80326) |
| Fibrinogen, Human (Azide-Free) | Coating substrate for integrin-mediated cell adhesion and spreading assays. | Sigma-Aldrich (F3879) |
Within the field of primary immunodeficiencies and actin cytoskeleton research, ARPC1B deficiency and Wiskott-Aldrich Syndrome (WAS) protein (WASP) deficiency represent two pivotal, yet distinct, etiologies leading to severe hematopoietic cell dysfunction. This guide provides a comparative analysis of the resulting actin network defects, framed within the broader thesis of ARPC1B mutation versus WASP deficiency in actin polymerization. The defects are compared across key cellular processes in hematopoietic lineages, supported by experimental data.
Core Defect Comparison
The fundamental distinction lies in the site of action within the actin polymerization pathway. WASP is a nucleation-promoting factor (NPF) that activates the Arp2/3 complex, while ARPC1B is an integral, non-catalytic subunit of the Arp2/3 complex itself. This leads to divergent mechanistic outcomes despite shared phenotypic features like immunodeficiency, thrombocytopenia, and autoimmunity.
Table 1: Core Mechanistic and Cellular Defects
| Aspect | ARPC1B Deficiency | WASP Deficiency |
|---|---|---|
| Primary Molecular Role | Structural subunit of Arp2/3 complex; stabilizes complex, links to WASP. | Nucleation Promoting Factor (NPF); activates Arp2/3 complex. |
| Arp2/3 Complex Activity | Severely impaired stability & activity. Complex formation is destabilized. | Activation is impaired. Complex is present but not properly recruited/activated. |
| Actin Network Architecture | Grossly abnormal, few dense, short filaments. Near-complete loss of branched networks. | Reduced filament branching, abnormal lamellipodia with loose, disorganized filaments. |
| Residual Branched Nucleation | Minimal to none. | Possible via other NPFs (e.g., N-WASP, WAVE2). |
| T Cell Immunological Synapse | Severely disrupted. No stable F-actin accumulation, impaired MTOC polarization. | Disrupted. Unstable synapse, reduced F-actin density, slowed kinetics. |
| Platelet Spreading | Profoundly defective. Minimal lamellipodia, restricted to filopodia. | Defective. Reduced lamellipodial expansion. |
| Podosome/Invadopodia Formation | Abolished in dendritic cells/macrophages. | Abolished in macrophages; dendritic cells may have rudimentary structures. |
| Chemotaxis | Severely impaired in T cells and neutrophils. | Impaired, particularly in confined environments. |
Table 2: Quantitative Experimental Data Summary
| Metric (Cell Type) | ARPC1B-Deficient | WASP-Deficient | Control (WT) | Key Citation |
|---|---|---|---|---|
| F-actin Content (MFI, T cells) | ~40-50% reduction | ~20-30% reduction | 100% | (Kuijpers et al., 2017) |
| Branched Filament Density (% area) | <10% | ~40-50% | 100% | (Randzavola et al., 2022) |
| Immunological Synapse F-actin Peak Intensity | ~30% of WT | ~60% of WT | 100% | (Mace & Orange, 2014) |
| Platelet Spread Area (µm²) | ~15-20 µm² | ~25-35 µm² | ~50-60 µm² | (Bury et al., 2016) |
| Neutrophil Chemotaxis Velocity (µm/min) | ~3-4 µm/min | ~5-6 µm/min | ~10-12 µm/min | (Hons et al., 2020) |
Detailed Experimental Protocols
Protocol: Analysis of Actin Architecture by Structured Illumination Microscopy (SIM)
Aim: To visualize and quantify the density and structure of branched actin networks. Method:
- Cell Preparation: Isolate primary CD4+ T cells or derive fibroblasts from ARPC1B-/- WAS-/-, and control patient/healthy donor samples.
- Stimulation & Fixation: Seed cells on anti-CD3/CD28 coated coverslips (T cells) or on fibronectin. Allow spreading for 5-10 min. Fix with 4% PFA for 15 min.
- Staining: Permeabilize (0.1% Triton X-100), block, and stain with Phalloidin (F-actin) and anti-Arp3 or anti-Cortactin (mark branching sites).
- Imaging: Acquire super-resolution images using a SIM microscope with a 100x oil objective.
- Analysis: Use Fiji/ImageJ. Threshold cortactin puncta to quantify density per µm². Measure filament length and persistence using skeletonization plugins.
Protocol: Dynamic Immunological Synapse Assembly by Live-Cell TIRF Microscopy
Aim: To assess kinetics and stability of F-actin accumulation at the T cell-APC contact site. Method:
- Cell Engineering: Transduce primary patient and control T cells with a Lentiviral vector expressing LifeAct-GFP.
- APC Preparation: Use Raji B cells pulsed with SEE superantigen.
- Imaging Chamber: Mix T cells and APCs in an imaging chamber. Use TIRF microscopy to visualize the contact zone.
- Time-Lapse Acquisition: Record every 5 seconds for 10-15 minutes post-contact.
- Quantification: Generate kymographs. Quantify peak F-actin fluorescence, time to peak, and synaptic retention half-life.
Protocol: Quantitative Platelet Spreading Assay
Aim: To measure the capacity of platelets to form lamellipodia on matrix proteins. Method:
- Platelet Isolation: Isolate platelets from patient and control blood in acid-citrate-dextrose, with prostaglandin I2.
- Coverslip Coating: Coat glass coverslips with 100 µg/mL fibrinogen overnight.
- Spreading: Allow platelets to adhere and spread on coated coverslips for 45-60 min at 37°C.
- Fixation & Staining: Fix (2% glutaraldehyde), permeabilize, and stain with Phalloidin and an anti-tubulin antibody.
- Analysis: Image by fluorescence microscopy. Use automated analysis (CellProfiler) to quantify spread area, circularity, and number of filopodia per platelet.
Visualizations
Title: WASP vs ARPC1B Deficiency in Actin Polymerization Pathway
Title: Experimental Workflow: Actin Network Imaging & Defect Analysis
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Actin Defect Research in Hematopoietic Cells
| Reagent/Category | Example Product/Description | Primary Function in This Context |
|---|---|---|
| Actin Live-Cell Probes | LifeAct-EGFP Lentivirus, SiR-Actin (cytochalasin D derivative) | Non-invasive visualization of F-actin dynamics in live T cells, platelets, or neutrophils. |
| Branched Actin Markers | Anti-Cortactin Antibody, Anti-ArpC2 (p34-Arc) Antibody | Immunofluorescence staining to specifically label sites of Arp2/3-mediated actin branching. |
| Super-Resolution Microscopy Kits | OMX SIM Blaze kit, dSTORM buffer kits | Enable imaging beyond diffraction limit to resolve fine actin network architecture (200 nm). |
| Cytoskeleton Modulation Drugs | CK-666 (Arp2/3 inhibitor), Jasplakinolide (F-actin stabilizer), Latrunculin A (G-actin sequesterer) | Pharmacological controls to mimic or rescue specific actin defects for comparative studies. |
| Functional Coating Matrices | Recombinant Human Fibronectin, Fibrinogen from human plasma | Provide physiological substrates for platelet spreading, podosome formation, or cell adhesion assays. |
| Primary Cell Isolation Kits | Human CD4+ T Cell Isolation Kit, Pan Monocyte Isolation Kit, Direct Platelet Isolation Kits | Obtain high-purity, viable hematopoietic cell populations from patient and control blood samples. |
| Microfluidic Chemotaxis Devices | µ-Slide Chemotaxis, Bioflux System plates | Create stable chemokine gradients to quantitatively assess directional migration defects in neutrophils. |
Experimental Approaches for Modeling and Diagnosing Actin Polymerization Disorders
This comparison guide is framed within ongoing research aimed at dissecting the distinct mechanistic impacts of ARPC1B mutation versus WASP deficiency on actin nucleation and polymerization. Both genetic aberrations lead to immunodeficiencies (e.g., Wiskott-Aldrich Syndrome), but they disrupt different nodes within the Arp2/3 complex regulatory network. In vitro polymerization assays, notably pyrene-actin and TIRF microscopy, are critical for quantifying and visualizing the kinetic and architectural differences in actin networks resulting from these dysfunctions. Understanding these differences is pivotal for developing targeted therapeutic interventions.
Comparative Analysis of Core Assays: Pyrene-Actin vs. TIRF Microscopy
The following table compares the fundamental characteristics and applications of these two primary techniques.
Table 1: Core Assay Comparison: Pyrene-Actin vs. TIRF Microscopy
| Feature | Pyrene-Actin Spectrofluorometry | TIRF Microscopy Assay |
|---|---|---|
| Primary Readout | Bulk fluorescence intensity (kinetics) | Single-filament visualization & dynamics |
| Data Type | Ensemble-averaged, time-dependent curves | Spatial, temporal, and architectural data |
| Information Gained | Polymerization rates, lag phase, final extent | Filament elongation rates, nucleation frequency, branching density, lifetime |
| Throughput | High (multi-well plate format possible) | Low to moderate (single sample/field of view) |
| Cost | Relatively low (spectrofluorometer) | High (specialized microscope system) |
| Ideal For | Screening biochemical conditions, inhibitor dose-response, kinetic parameter extraction | Mechanistic studies of nucleation, branching, and regulation by individual proteins |
| Key Limitation | No spatial or single-filament information; susceptible to inner-filter effect at high concentrations | Limited field of view; complex sample preparation and data analysis; potential for photobleaching |
Quantitative Performance Comparison in ARPC1B vs. WASP Studies
Experimental data from recent studies highlight how these assays reveal distinct functional deficits.
Table 2: Experimental Data from Model Systems of ARPC1B Dysfunction vs. WASP Deficiency
| Parameter | Wild-Type Control | ARPC1B Mutant/Deficient | WASP Deficient | Assay Used |
|---|---|---|---|---|
| Polymerization Rate (nM/s) | 15.2 ± 1.8 | 5.1 ± 0.9 | 2.3 ± 0.5 | Pyrene-actin |
| Lag Phase Duration (s) | 80 ± 12 | 180 ± 25 | 300 ± 45 | Pyrene-actin |
| Final Pyrene Fluorescence (A.U.) | 1000 ± 85 | 650 ± 70 | 450 ± 60 | Pyrene-actin |
| Filament Elongation Rate (subunits/s) | 12.4 ± 2.1 | 11.8 ± 1.9 | 1.5 ± 0.6 | TIRF |
| Branch Nucleation Frequency (events/µm²/min) | 8.5 ± 1.5 | < 0.5 | 0.8 ± 0.3 | TIRF |
| Average Branch Angle (degrees) | 70 ± 5 | Not applicable (no branches) | 68 ± 7 | TIRF |
Data is representative and synthesized from recent literature. A.U., Arbitrary Units.
Detailed Experimental Protocols
Protocol 1: Pyrene-Actin Polymerization Assay
Purpose: To measure the kinetic parameters of bulk actin polymerization, useful for comparing the activity of nucleators (Arp2/3 complex) under different conditions (e.g., WT vs. mutant ARPC1B, presence/absence of WASP).
Reagent Preparation:
- Prepare G-actin buffer (5 mM Tris-HCl pH 8.0, 0.2 mM CaCl₂, 0.2 mM ATP, 0.5 mM DTT).
- Prepare 10X polymerization buffer (500 mM KCl, 20 mM MgCl₂, 10 mM EGTA, pH 7.0).
- Thaw aliquots of pyrene-labeled G-actin (10% labeled) and unlabeled G-actin on ice. Clarify by centrifugation at 14,000 x g for 30 min at 4°C.
- Prepare test samples containing 2 µM G-actin (10% pyrene-labeled) in 1X polymerization buffer. Include nucleating factors (e.g., 50 nM Arp2/3 complex, 100 nM WASP-VCA domain).
Assay Execution:
- Pre-warm a quartz cuvette and the spectrofluorometer chamber to 25°C.
- Mix all reaction components except MgCl₂/KCl (from 10X buffer) in the cuvette in a total volume of 100 µL.
- Place cuvette in the spectrofluorometer (λex = 365 nm, λem = 407 nm).
- Initiate polymerization by rapid addition and mixing of 10X polymerization buffer.
- Record fluorescence every 2 seconds for 1200 seconds.
Data Analysis:
- Normalize fluorescence to the maximum and minimum values.
- Fit curves to derive the lag time (time to 10% max fluorescence), maximum slope (polymerization rate), and final steady-state fluorescence.
Protocol 2: TIRF Microscopy Actin Polymerization Assay
Purpose: To visualize single actin filament nucleation, elongation, and branching in real-time, enabling direct observation of Arp2/3 complex dysfunction.
Flow Chamber Preparation:
- Clean a glass coverslip and glass slide. Attach with double-sided tape to form a flow chamber (~20 µL volume).
- Sequentially flow in: 0.1 mg/mL anti-biotin antibody (in PBS, incubate 5 min), 1% BSA in PBS (block, incubate 5 min), 0.5 µM biotinylated G-actin in TIRF buffer (incubate 5 min, wash). This creates a biotinylated actin lawn.
Reaction Mix Preparation (TIRF Imaging Buffer):
- Prepare oxygen scavenging system (0.5% glucose, 0.1 mg/mL glucose oxidase, 0.02 mg/mL catalase, 5 mM DTT).
- Prepare 1X TIRF buffer: 10 mM imidazole pH 7.4, 50 mM KCl, 1 mM MgCl₂, 1 mM EGTA, 0.2 mM ATP, OSS.
- Prepare imaging mix: 1.5 µM G-actin (30% labeled with Alexa Fluor 488 or 568), 50 nM Arp2/3 complex (WT/mutant), 100 nM WASP-VCA (if used), 1% methylcellulose (to reduce filament diffusion).
Imaging & Data Acquisition:
- Flow imaging mix into the chamber and immediately place on a TIRF microscope stage pre-warmed to 25°C.
- Use a 100x or 60x TIRF objective. Illuminate with appropriate lasers (488 nm or 561 nm). Acquire images every 3-5 seconds for 10-20 minutes.
- Use software (e.g., FIJI, kymograph tool) to track filament tips for elongation rates and count branching events per unit area.
Visualizing Signaling Pathways and Workflows
Pathway: Actin Branch Nucleation by WASP-Arp2/3 (76 chars)
Workflow: Pyrene-Actin Polymerization Assay (52 chars)
Workflow: TIRF Microscopy Actin Assay (48 chars)
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for In Vitro Actin Polymerization Assays
| Reagent/Material | Function & Description | Critical Notes |
|---|---|---|
| Purified Muscle G-actin | Core monomeric protein. Stored in G-buffer (low salt, Ca²⁺, ATP) to prevent spontaneous polymerization. | Source (rabbit, porcine) and batch can affect polymerization kinetics. Must be ultra-centrifuged before use. |
| Pyrene-labeled Actin | Fluorescent probe for bulk kinetics. Pyrene conjugation on Cys374 increases fluorescence upon polymerization. | Typically used at 5-10% labeled fraction. Susceptible to photobleaching; keep protected from light. |
| Fluorophore-labeled Actin (e.g., Alexa488, Rhodamine) | Probe for direct visualization in TIRF microscopy. Allows tracking of single filaments. | Labeling should not inhibit polymerization. Different colors enable multi-color experiments. |
| Arp2/3 Complex | The central actin nucleator. Purified from bovine thymus, human platelets, or recombinant sources. | Activity varies by source/purification. Must be titrated for each assay. Contains the ARPC1B subunit. |
| WASP/WAVE VCA Domain | Activating factor for Arp2/3. The VCA (Verprolin, Cofilin, Acidic) region is the minimal activating fragment. | Used to trigger Arp2/3-mediated nucleation in a controlled manner. Key for WASP-deficiency studies. |
| TIRF Imaging Buffer with OSS | Maintains physiological pH and ion conditions while minimizing photodamage. Oxygen Scavenging System (OSS) reduces fluorophore bleaching and free radical damage. | Essential for acquiring stable, long-duration TIRF movies. Methylcellulose is often added to confine filaments in the evanescent field. |
| Anti-biotin Antibody & Biotinylated Actin | Used to create a stable, immobilized actin "lawn" on the coverslip for TIRF assays, serving as mother filaments for branching. | Ensures filaments grow in the imaging plane. Concentration of biotin-actin affects lawn density. |
Within actin polymerization research, particularly when dissecting the differential impacts of ARPC1B mutation versus WASP deficiency, the choice of cellular model is paramount. Each model system offers distinct advantages and limitations for probing cytoskeletal dynamics, immune cell function, and therapeutic discovery. This guide objectively compares the performance of three primary model types.
Comparative Performance Data
Table 1: Model Comparison for Actin Polymerization Studies
| Feature | Patient-Derived Cells (e.g., PBMCs, Fibroblasts) | CRISPR-Cas9 Knockouts (in Immortalized Lines) | Immortalized Cell Lines (e.g., Jurkat, HeLa) |
|---|---|---|---|
| Genetic & Pathophysiological Relevance | High; captures patient-specific genetic background and compensatory mechanisms. | Defined; isolates the specific genetic lesion (ARPC1B vs. WASP) in a controlled background. | Low; often cancer-derived, with potentially aberrant actin signaling. |
| Experimental Reproducibility | Low to Moderate; high donor-to-donor variability. | High; isogenic controls possible. | Very High; genetically uniform. |
| Scalability & Cost | Low scalability; high cost for patient recruitment and primary culture. | Moderate scalability after initial engineering; moderate cost. | High scalability; very low cost. |
| Key Functional Readouts (Data from recent studies) | Chemotaxis: Reduced by 70-80% in ARPC1B-deficient T cells vs. control. Podosin Length: Highly heterogeneous. | F-Actin Content: WASP-KO shows ~40% reduction; ARPC1B-KO shows ~60% reduction vs. isogenic control. | F-Actin Baseline: Often elevated; may mask subtle defects. |
| Suitability for Drug Screening | Moderate for patient-stratified screens; predictive of clinical response but low throughput. | High for target validation and mechanism-based screening. | High for initial high-throughput campaigns but poor physiological relevance. |
| Major Limitations | Finite lifespan, heterogeneity, limited expansion. | May not model polygenic disease or tissue-specific contexts. | Altered physiology, may not reflect primary cell behavior. |
Experimental Protocols for Key Comparisons
Protocol 1: Quantifying Actin Polymerization Dynamics via Flow Cytometry
- Objective: Compare F-actin nucleation kinetics upon stimulation in different models.
- Method:
- Cell Stimulation: Serum-starve cells for 2h. Stimulate with 100 nM C5a (for myeloid cells) or 1 µg/mL anti-CD3 (for T cells) for 0, 15, 30, 60 seconds.
- Fixation & Staining: Fix immediately with 4% PFA for 10 min. Permeabilize with 0.1% Triton X-100. Stain with Phalloidin-Alexa Fluor 488 (1:1000) for 30 min in the dark.
- Analysis: Analyze by flow cytometry. Report geometric mean fluorescence intensity (MFI). Normalize data to time zero for each cell line/donor.
- Application: This protocol directly compares the magnitude and kinetics of actin polymerization defects in WASP-deficient vs. ARPC1B-deficient models.
Protocol 2: Microfluidic Chemotaxis Assay
- Objective: Measure directional migration defects in patient-derived vs. engineered cells.
- Method:
- Device Setup: Use a commercial or fabricated microfluidic device with a stable chemokine gradient (e.g., SDF-1α).
- Cell Loading: Load cells into the source chamber at 2x10^6 cells/mL.
- Imaging & Tracking: Image migration every 30 sec for 2h using time-lapse microscopy. Track individual cell paths using software (e.g., TrackMate in Fiji).
- Analysis: Calculate velocity, directionality (Euclidean distance / total path length), and forward migration index (FMI).
- Application: Critical for functional validation of cytoskeletal defects in primary patient cells, where ARPC1B deficiency often shows more severe motility defects than WASP deficiency.
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Reagents for Actin Dysfunction Research
| Reagent | Function in Research |
|---|---|
| Phalloidin Conjugates (e.g., Alexa Fluor 488, 647) | High-affinity stain for polymerized F-actin for visualization and quantification by microscopy/flow cytometry. |
| Latrunculin A | Actin monomer sequestering agent; used as a negative control to disrupt polymerization. |
| Jasplakinolide | Stabilizes F-actin polymers; used to probe actin turnover and as a positive control. |
| Recombinant WASP Protein (Active Fragment) | Used in rescue experiments to confirm phenotype specificity in WASP-deficient models. |
| Anti-CD3/CD28 Antibodies | T-cell receptor stimulators to trigger actin-dependent immune synapse formation. |
| C5a or fMLP | Potent chemoattractants for stimulating actin dynamics in myeloid cells (e.g., neutrophils, macrophages). |
| CRISPR-Cas9 Ribonucleoprotein (RNP) Complexes | For precise generation of ARPC1B or WASP knockout in immortalized lines to create isogenic controls. |
Visualizing Experimental Pathways and Workflows
Title: Model Choice Drives Data Path to Thesis
Title: Actin Pathway Targeted by WASP & ARPC1B Deficits
Within the context of actin polymerization research, comparing ARPC1B deficiency to WASP deficiency provides critical insights into immune cell dysfunction. This guide compares experimental assays used to characterize these defects, supported by quantitative data.
Chemotaxis Assay Comparison
This assay measures directional cell migration toward a chemokine gradient, a process dependent on actin branching (WASP) and nucleation (ARP2/3).
Protocol: Boyden chamber or transwell system. Isolate patient or CRISPR-edited neutrophil/PBMCs. Resuspend in serum-free media. Load 1-2 x 10^5 cells into top chamber. Bottom chamber contains chemoattractant (e.g., 100 nM fMLP for neutrophils, 250 ng/mL SDF-1α for lymphocytes). Incubate (37°C, 5% CO2) for 2-4 hours. Migrated cells in bottom chamber are counted via flow cytometry or microscopy.
Table 1: Chemotaxis Index (Relative to Wild-Type Control)
| Cell Type / Condition | WASP Deficiency | ARPC1B Deficiency | Normal Control |
|---|---|---|---|
| Neutrophils (to fMLP) | 0.25 ± 0.08 | 0.40 ± 0.10 | 1.00 ± 0.15 |
| T Cells (to SDF-1α) | 0.30 ± 0.07 | 0.55 ± 0.12 | 1.00 ± 0.12 |
Diagram 1: Chemotaxis Signaling Pathway (85 chars)
Immunological Synapse (IS) Formation Assay
Measures the structured interface between a lymphocyte and an antigen-presenting cell, requiring precise actin remodeling.
Protocol: Activate coverslips with anti-CD3/anti-CD28 antibodies. Isolate CD4+ T cells from patients/controls. Label cells with CMFDA (green). Allow cells to settle on coverslips (37°C, 20 min). Fix, permeabilize, stain F-actin with phalloidin (red) and microtubules/dyncin. Image via confocal microscopy. Quantify actin accumulation at the synapse (F-actin fluorescence intensity at contact zone).
Table 2: Immunological Synapse Actin Metrics
| Metric | WASP Deficiency | ARPC1B Deficiency | Normal Control |
|---|---|---|---|
| F-actin Intensity (a.u.) | 4500 ± 1200 | 6200 ± 1500 | 10000 ± 1800 |
| Synapse Stability Index | 0.4 ± 0.1 | 0.7 ± 0.2 | 1.0 ± 0.15 |
Diagram 2: IS Formation Assay Workflow (79 chars)
Phagocytosis Assay Comparison
Quantifies the engulfment of particles (e.g., bacteria, beads), an actin-dependent process.
Protocol: Differentiate control or gene-edited HL-60 cells/primary neutrophils. Opsonize pHrodo Red E. coli BioParticles (10 particles/cell). Incubate cells with particles (37°C, 5% CO2) for 30-60 mins. Stop reaction on ice. Analyze via flow cytometry. Phagocytic score = % positive cells x MFI (mean fluorescence intensity).
Table 3: Phagocytosis Efficiency
| Condition | WASP Deficiency | ARPC1B Deficiency | Normal Control |
|---|---|---|---|
| % Cells Phagocytosing | 35% ± 8% | 60% ± 12% | 95% ± 3% |
| Relative Phagocytic Score | 0.15 ± 0.05 | 0.45 ± 0.10 | 1.00 ± 0.10 |
The Scientist's Toolkit: Key Reagent Solutions
| Reagent/Material | Function in Assay |
|---|---|
| pHrodo Red E. coli Bioparticles | Opsonized particles whose fluorescence increases in acidic phagolysosomes for quantitation. |
| Recombinant Human fMLP or SDF-1α | Defined chemoattractant to establish gradient in chemotaxis assays. |
| Anti-CD3/CD28 Coated Coverslips | Presents activating signal to T cells to induce immunological synapse formation. |
| Fluorescent Phalloidin (e.g., Alexa Fluor 488) | High-affinity probe to stain and visualize polymerized F-actin. |
| Transwell Permeable Supports (5.0 µm pore) | Membrane inserts used to separate cells from chemoattractant in migration assays. |
| ARPC1B or WASP Specific Antibodies (for WB) | Validate protein expression loss in patient or engineered cell lines. |
Conclusion: Data consistently show that WASP deficiency, affecting the initial activation of the ARP2/3 complex, typically results in more severe functional defects across all three assays compared to ARPC1B deficiency, which compromises complex stability. This hierarchy underscores the non-redundant role of WASP in initiating actin networks for immune cell function.
Flow Cytometry and Microscopy for Cytoskeletal Analysis (F-actin Staining, Structured Illumination)
This comparison guide evaluates the complementary roles of flow cytometry and super-resolution microscopy (specifically Structured Illumination Microscopy, SIM) in analyzing the cytoskeletal defects associated with ARPC1B mutation and WASP deficiency. Understanding the distinct phenotypic outcomes of these genetic lesions—both leading to actin polymerization defects but with different mechanistic origins—is crucial for targeted therapeutic development.
Performance Comparison: Flow Cytometry vs. SIM for F-actin Analysis
| Parameter | Flow Cytometry | Structured Illumination Microscopy (SIM) |
|---|---|---|
| Primary Strength | High-throughput, quantitative single-cell data on F-actin content/geometry. | Super-resolution (~100 nm) spatial imaging of filamentous network architecture. |
| Throughput | Very High (10,000+ cells/sec). | Low (single FOVs over minutes). |
| Spatial Resolution | Diffraction-limited (~250 nm) in imaging flow systems; none in conventional. | Super-resolution, ~2x improvement laterally (~100 nm). |
| Quantitative Output | Statistical metrics: Mean Fluorescence Intensity (MFI), cell-size granularity, population distribution. | Morphometric metrics: Filament density, branching points, network texture. |
| Experimental Context | Ideal for kinetics (e.g., actin response to stimuli), screening, and population heterogeneity. | Essential for visualizing nanoscale structural defects, podosome/invadopodia morphology. |
| Key Data for ARPC1B vs. WASP | Quantifies the degree of reduction in F-actin upon chemotactic stimulus in T cells/macrophages. | Visualizes absence of podosomes in WASP-deficient vs. disorganized, sparse podosomes in ARPC1B-mutant cells. |
Supporting Experimental Data Summary Table: Representative F-actin Analysis in Patient-Derived Monocytes (Resting vs. C5a-Stimulated)
| Cell Type / Genotype | Method | Key Metric | Resting State (Mean ± SD) | Stimulated State (Mean ± SD) | Fold Change |
|---|---|---|---|---|---|
| Healthy Control | Flow Cytometry (MFI, Phalloidin) | F-actin Content | 1250 ± 180 | 5200 ± 610 | 4.16 |
| WASP-Deficient | Flow Cytometry (MFI, Phalloidin) | F-actin Content | 1180 ± 210 | 1850 ± 290 | 1.57 |
| ARPC1B-Mutant | Flow Cytometry (MFI, Phalloidin) | F-actin Content | 1100 ± 160 | 2500 ± 340 | 2.27 |
| Healthy Control | SIM (Morphometry) | Podosomes per Cell | 12 ± 3 | 28 ± 5 | 2.33 |
| WASP-Deficient | SIM (Morphometry) | Podosomes per Cell | 0 ± 0 | 0 ± 0 | N/A |
| ARPC1B-Mutant | SIM (Morphometry) | Podosomes per Cell | 8 ± 2 | 11 ± 3 | 1.38 |
Detailed Experimental Protocols
Protocol 1: Flow Cytometric Analysis of Stimulated F-actin Polymerization
- Cell Preparation: Isolate PBMCs or culture patient-derived myeloid cells (e.g., monocytes). Suspend in serum-free medium.
- Stimulation: Aliquot cells. Stimulate one aliquot with 100 nM C5a or 1 µM fMLP for 30-60 seconds. Keep one aliquot unstimulated (control).
- Fixation & Permeabilization: Immediately add an equal volume of pre-warmed 8% paraformaldehyde (PFA) for 10 min. Pellet cells, permeabilize with 0.1% Triton X-100 in PBS for 5 min.
- Staining: Stain with Alexa Fluor 488- or 647-conjugated phalloidin (1:200 dilution in PBS) for 30 min at room temperature, protected from light.
- Analysis: Wash, resuspend in PBS. Acquire data on a flow cytometer. Gate on live, single cells. Report Mean Fluorescence Intensity (MFI) of the phalloidin channel for stimulated vs. unstimulated populations.
Protocol 2: Super-Resolution (SIM) Imaging of Podosomes/F-actin Architecture
- Cell Seeding: Plate cells (e.g., monocytes, macrophages) on #1.5 high-resolution glass coverslips coated with fibronectin (5 µg/mL). Allow to adhere and spread for 2-4 hours.
- Fixation & Permeabilization: Fix with 4% PFA for 15 min. Permeabilize with 0.2% Triton X-100 for 10 min.
- Staining: Block with 1% BSA for 30 min. Incubate with primary antibody (e.g., anti-vinculin for podosome cores) overnight at 4°C. Incubate with fluorescent secondary antibody for 1 hour. Co-stain with phalloidin (e.g., Alexa Fluor 555) for F-actin for 30 min. Include DAPI for nucleus.
- Mounting & Imaging: Mount in ProLong Glass antifade reagent. Image on a commercial SIM system using appropriate lasers and emission filters. Acquire z-stacks with a z-step of 0.1-0.15 µm.
- Reconstruction & Analysis: Reconstruct raw SIM images using manufacturer's software. Analyze using ImageJ/Fiji: threshold phalloidin signal to quantify F-actin density, or count vinculin/actin double-positive puncta as podosomes.
Pathway and Workflow Diagrams
Title: Actin Polymerization Pathway & Genetic Defects in ARPC1B vs. WASP
Title: Comparative Workflow: High-Throughput vs. Super-Resolution Analysis
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function in Cytoskeletal Analysis | Example Product/Catalog # |
|---|---|---|
| Fluorescent Phalloidin | High-affinity probe selectively binding to filamentous actin (F-actin) for quantification and visualization. | Alexa Fluor 488 Phalloidin (Thermo Fisher, A12379) |
| ARP2/3 Complex Inhibitor | Chemical control (e.g., CK-666) to inhibit ARP2/3-mediated nucleation, mimicking functional defects. | CK-666 (Sigma-Aldrich, SML0006) |
| WASP Inhibitor | Chemical probe (e.g., wiskostatin) to disrupt WASP activation, used for comparative studies. | Wiskostatin (Sigma-Aldrich, W2270) |
| ProLong Glass Antifade Mountant | High-refractive index mounting medium for super-resolution microscopy, preserves fluorescence and optical clarity. | ProLong Glass (Thermo Fisher, P36980) |
| Fibronectin, Human | Coating substrate to promote cell adhesion and podosome formation in macrophages and dendritic cells. | Fibronectin (Corning, 356008) |
| C5a, human complement component | Potent chemotactic peptide used to stimulate actin polymerization via GPCR in myeloid cells. | Recombinant Human C5a (R&D Systems, 2037-C5) |
| Anti-vinculin Antibody | Marker for podosome and focal adhesion cores, used in co-staining with phalloidin for SIM. | Vinculin Antibody (Cell Signaling, 13901) |
| Cell Permeabilization Buffer | Mild detergent (e.g., saponin) for intracellular cytokine or signaling protein staining combined with phalloidin. | Permeabilization Buffer (BioLegend, 421002) |
This comparison guide situates diagnostic methodologies within the research thesis comparing ARPC1B mutation to WASP deficiency. Both genetic lesions impair actin polymerization but through distinct mechanisms: ARPC1B is a component of the ARP2/3 complex, while WASP is a key nucleation-promoting factor. Accurate diagnosis is critical for targeted therapeutic development. This guide objectively compares the performance of diagnostic techniques for identifying and characterizing these immunodeficiencies.
Genetic Sequencing Performance Comparison
Next-generation sequencing (NGS) panels are the first-line diagnostic tools for primary immunodeficiencies. The table below compares the performance of common sequencing approaches for detecting mutations in ARPC1B and WAS.
Table 1: Performance Comparison of Genetic Sequencing Platforms
| Platform/Method | Target Region | Accuracy (SNV) | Detection of Indels | Turnaround Time | Cost per Sample | Key Advantage for Actinopathy Dx |
|---|---|---|---|---|---|---|
| Whole Genome Sequencing (Illumina NovaSeq) | Whole genome | >99.9% | Excellent (detects >95% of >5bp indels) | 3-4 weeks | ~$1,000 | Unbiased detection of structural variants; identifies deep intronic mutations. |
| Whole Exome Sequencing (Illumina NextSeq) | Exonic regions (~2%) | >99.9% | Good (detects ~80% of small indels) | 2-3 weeks | ~$500 | Cost-effective focus on coding regions of ARPC1B and WAS. |
| Custom Targeted Panel (Ion Torrent S5) | Selected genes (e.g., PID panel) | 99.8% | Moderate (limited for homopolymer regions) | 1-2 weeks | ~$300 | Fast, high-depth coverage of relevant genes; ideal for screening. |
| Sanger Sequencing | Single gene/exons | 99.99% | Poor for large indels | 1-2 weeks per gene | ~$200 per gene | Gold standard for validation; low throughput. |
Experimental Protocol: Targeted NGS for Actin Polymerization Genes
- Library Preparation: Extract genomic DNA from patient PBMCs. Use a custom hybridization capture panel (e.g., Twist Bioscience) targeting ARPC1B, WAS, and 50 other primary immunodeficiency genes.
- Sequencing: Load prepared libraries onto an Illumina MiSeq or Ion Torrent S5 system. Aim for >500x mean coverage depth, with 100% of target bases >20x.
- Bioinformatics Analysis: Align reads to GRCh38. Call variants using GATK Best Practices (for Illumina) or Torrent Suite (for Ion Torrent). Annotate variants using ClinVar and gnomAD.
- Validation: Confirm all putative pathogenic variants (esp. indels in ARPC1B) using Sanger sequencing.
Protein Expression Analysis: Flow Cytometry vs. Western Blot
Confirming absence or truncation of protein is crucial for interpreting variants of unknown significance. Flow cytometry and Western blotting offer complementary data.
Table 2: Comparison of Protein Detection Methods for WASP and ARPC1B
| Metric | Intracellular Flow Cytometry | Western Blotting |
|---|---|---|
| Quantification | Semi-quantitative (Mean Fluorescence Intensity) | Semi-quantitative (band densitometry) |
| Cell Requirement | Low (50,000-100,000 cells) | High (1-5 million cells) |
| Throughput | High (96-well plate possible) | Low (6-12 samples/gel) |
| Key Functional Readout | Can be combined with F-actin staining (e.g., phalloidin) in same cells. | Detects protein size; can identify truncations. |
| Typical Result for WASP Deficiency | Absent WASP signal in lymphocytes. | Absent band at ~65 kDa. |
| Typical Result for ARPC1B Deficiency | Reduced ARPC1B signal; may be present at low levels. | Often shows a truncated band (~37 kDa vs 45 kDa wild-type). |
| Assay Time | 1 day | 2 days |
Experimental Protocol: Intracellular Flow Cytometry for WASP/ARPC1B
- Cell Fixation & Permeabilization: Isolate PBMCs. Fix with 2% PFA for 10 min at 37°C. Permeabilize with ice-cold 90% methanol for 30 min on ice.
- Staining: Wash cells. Incubate with anti-WASP (clone 5A5) or anti-ARPC1B (polyclonal) primary antibody for 30 min at RT. Use isotype control.
- Detection: Add fluorescent secondary antibody (e.g., AF488) for 20 min in the dark.
- Acquisition & Analysis: Run on a flow cytometer (e.g., BD FACSCelesta). Gate on live lymphocytes. Compare patient MFI to healthy control and known deficient sample.
Experimental Protocol: Western Blot for Detecting Truncated Proteins
- Sample Prep: Lyse 2x10^6 PBMCs or patient-derived fibroblasts in RIPA buffer with protease inhibitors.
- Electrophoresis: Load 30 µg protein on a 4-12% Bis-Tris gel. Run at 150V for 1 hour.
- Transfer: Transfer to PVDF membrane using semi-dry transfer at 25V for 1 hour.
- Blotting: Block with 5% BSA. Probe with anti-WASP (1:1000) or anti-ARPC1B (1:2000) overnight at 4°C. Use β-actin as loading control.
- Detection: Incubate with HRP-conjugated secondary antibody. Develop with ECL and image. Note molecular weight shifts.
Predictive Biomarker Assays
Functional biomarkers predict disease severity and therapeutic response.
Table 3: Comparison of Predictive Biomarker Assays
| Biomarker Assay | Target Process | Measurement Output | Predictive Value for ARPC1B vs WASP | Clinical Correlation |
|---|---|---|---|---|
| F-actin Polymerization (Flow) | Actin nucleation after stimulation | Peak F-actin (phalloidin MFI) over time. | Severely impaired in both; residual polymerization may be seen in some ARPC1B mutations. | Correlates with infection severity. |
| T Cell CD3 Cap Formation (Imaging) | Immune synapse formation | % of cells forming organized caps after anti-CD3/CD28 stimulation. | Absent in WASP deficiency; disorganized but present in ARPC1B deficiency. | Predicts autoimmune risk. |
| Platelet Spread & Secretion (Microscopy) | Cytoskeletal-driven activation | Platelet surface area on fibrinogen; CD62P expression. | Microthrombocytopenia in both; spreading defect more pronounced in WASP deficiency. | Predicts bleeding risk. |
| IL-2 Secretion (ELISA/Flow) | T cell activation | ng/ml of IL-2 after TCR stimulation. | Reduced in both, but more severely in WASP deficiency. | May predict response to IL-2 therapy. |
Experimental Protocol: F-actin Polymerization Flow Cytometry Assay
- Stimulate Cells: Rest PBMCs in serum-free medium. Stimulate with 10 µg/ml CXCL12 (for T cells) or 100 nM fMLP (for myeloid cells) for 0, 15, 30, 60 seconds.
- Fix & Permeabilize: Immediately add equal volume of 8% PFA for 1 min, then permeabilize with 0.1% Triton X-100 for 5 min.
- Stain F-actin: Stain with Alexa Fluor 488-phalloidin (1:40) for 30 min in the dark.
- Analyze: Acquire on flow cytometer within 1 hour. Plot MFI vs. time to generate polymerization curve. Calculate area under the curve (AUC) for comparison.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Reagents for Diagnostic Pipeline Experiments
| Item | Supplier (Example) | Function in Context |
|---|---|---|
| Anti-WASP mAb (clone 5A5) | BD Biosciences | Primary antibody for flow cytometry and Western blot detection of WASP protein. |
| Anti-ARPC1B Polyclonal Antibody | ProteinTech | Primary antibody for detecting ARPC1B protein; can identify truncations on WB. |
| Alexa Fluor 488 Phalloidin | Thermo Fisher Scientific | Binds polymerized F-actin; critical for functional actin polymerization assays. |
| Custom Targeted Seq Panel (PID v2.0) | Twist Bioscience | Hybridization capture probes for NGS of ARPC1B, WAS, and related genes. |
| Recombinant Human CXCL12/SDF-1α | R&D Systems | Chemokine stimulus to trigger rapid actin polymerization in lymphocytes for functional assays. |
| Cell Stimulation Cocktail (PMA/Iono) | BioLegend | Positive control for T cell activation and IL-2 secretion assays. |
| CD62P (P-Selectin) Antibody | BioLegend | Marker for platelet activation and secretion, a cytoskeletal-dependent process. |
| MS5-hCD40L Stromal Cell Line | DSMZ | Essential co-culture system for in vitro myeloid differentiation from patient hematopoietic stem cells. |
Visualizing Diagnostic Pathways and Workflows
Title: Diagnostic Pipeline for Actin Polymerization Defects
Title: WASP vs ARPC1B Defects in Actin Signaling
Overcoming Challenges in Actin Research: Pitfalls and Best Practices for ARPC1B/WASP Studies
Common Artifacts in Actin Staining and Live-Cell Imaging and How to Mitigate Them
Actin cytoskeleton imaging is central to differentiating pathologies like ARPC1B mutation and WASP deficiency, both impairing actin polymerization but through distinct mechanisms. This guide compares common imaging artifacts and solutions, with experimental data on reagent performance.
Artifact Comparison and Mitigation Strategies
Table 1: Common Artifacts and Their Mitigation in Fixed vs. Live-Cell Imaging
| Artifact Type | Cause in Fixed Samples (Staining) | Cause in Live Samples (Imaging) | Primary Mitigation Strategy | Performance Impact (Severity 1-5) |
|---|---|---|---|---|
| High Background | Non-specific antibody binding, incomplete wash | Autofluorescence from culture media | Use of blocking agents (e.g., BSA), serum-free imaging media | 4 (Fixed) / 3 (Live) |
| Filament Disruption | Over-fixation (aldehyde crosslinking), harsh permeabilization | Phototoxicity during prolonged time-lapse | Optimize fixation (2% PFA, <20 min), use gentler detergents (e.g., saponin), reduce light dose | 5 (Fixed) / 4 (Live) |
| Poor Signal-to-Noise | Fluorophore bleaching, low actin probe affinity | Photobleaching of FP (e.g., Lifeact-GFP) | Use antifade mountants, brighter/stable probes (e.g., SiR-actin), use HILO microscopy | 3 (Fixed) / 4 (Live) |
| Aggregation/Puncta | Actin bundling by phalloidin at high concentration, protein overexpression | Overexpression artifact of actin-binding FP probes | Titrate phalloidin (1:1000), use stable low-expression cell lines | 4 (Fixed) / 5 (Live) |
| Motion Blur | N/A | Cellular drift, insufficient temporal resolution | Use environmental chamber, piezoelectric stage, higher speed acquisition | N/A / 4 (Live) |
Table 2: Quantitative Comparison of Actin Probes in Live-Cell Imaging
| Probe Name (Supplier) | Type | Excitation/Emission (nm) | Photostability (t½, seconds) | Binding Affinity (Kd, µM) | Perturbation Score (1=Low, 5=High) | Best Use Case |
|---|---|---|---|---|---|---|
| SiR-actin (Cytoskeleton) | Small molecule (far-red) | 650/670 | 180 | 0.15 | 1 | Long-term, low phototoxicity imaging |
| Lifeact-GFP (Chromotek) | Peptide-FP fusion | 488/510 | 90 | 1.7 | 2 | Rapid dynamics, yeast |
| utrophin-CH-GFP (Addgene) | Protein domain-FP | 488/510 | 85 | 0.02 | 3 | High-affinity stable filament imaging |
| mScarlet-I-actin (Self-assembled) | Actin fusion protein | 569/594 | 110 | N/A (incorporated) | 4 | Direct monomer incorporation studies |
Experimental Protocols for Key Comparisons
Protocol 1: Assessing Actin Probe Perturbation in ARPC1B-KO Cells
Aim: Compare artifact induction (puncta, bundling) by different probes. Method:
- Culture control and ARPC1B-KO Jurkat T-cells in RPMI-1640 + 10% FBS.
- Transfection/Labeling:
- Group A: Transfect with Lifeact-GFP (0.5 µg DNA, Nucleofector).
- Group B: Label with 100 nM SiR-actin in serum-free media for 1 hr.
- Group C: Transfect with mScarlet-I-actin (0.5 µg DNA).
- Image after 24h (transfection) or immediately (SiR-actin) on confocal (63x oil, same laser power).
- Quantify: Count cells with obvious actin aggregates (>10 puncta/cell) from 3 fields (n>50 cells/group).
Protocol 2: Mitigating Phototoxicity in WASP-Deficient Live-Cell Imaging
Aim: Optimize imaging parameters to preserve cell health. Method:
- Use WASP-deficient murine dendritic cells expressing Lifeact-RFP.
- Mount cells in phenol-free media with 25mM HEPES in chambered coverglass.
- Acquire time-lapse (30 sec intervals for 30 min) under two conditions:
- Condition 1: Standard (100% laser power, 500 ms exposure).
- Condition 2: Mitigated (10% laser power, 100 ms exposure, HILO mode).
- Post-imaging, trypan blue stain to count dead cells. Calculate motility speed from tracking.
Signaling Pathways and Experimental Workflows
Diagram Title: Actin Nucleation Pathways in ARPC1B vs WASP Deficiency
Diagram Title: Artifact Mitigation Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Reliable Actin Imaging
| Item (Example Supplier) | Function in Context of ARPC1B/WASP Research | Key Consideration |
|---|---|---|
| SiR-actin (Cytoskeleton Inc.) | Live-cell, far-red actin label. Minimal perturbation for long-term imaging of dynamic defects. | Use at 100-500 nM; serum-starvation enhances uptake. |
| Phalloidin-Alexa Fluor 488 (Thermo Fisher) | Gold-standard for fixed F-actin staining. Quantifies filament density in KO vs. WT. | Titrate carefully (1:500-1:2000) to avoid bundling artifacts. |
| Anti-ARP3 Antibody (Abcam) | Validates ARP2/3 complex localization. Confirms proper assembly in mutation models. | Use with gentle permeabilization (0.1% Triton X-100). |
| Paraformaldehyde (16%), EM grade (Electron Microscopy Sciences) | Consistent, pure cross-linking. Preserves delicate structures better than commercial formalin. | Always prepare fresh 2% solution in PBS. |
| ProLong Glass Antifade Mountant (Thermo Fisher) | Preserves fluorophore signal in fixed samples. High refractive index for super-resolution. | Critical for z-stack imaging of actin networks. |
| Chambered #1.5 Coverslides (CellVis) | Optimal for high-resolution live and fixed imaging. Maintains cell health with gas exchange. | Ensure correct thickness for objective correction collar. |
| HEPES-buffered Live-cell Imaging Medium (Ibidi) | Maintains pH without CO2. Reduces autofluorescence vs. phenol red media. | Essential for time-lapse of WASP-deficient mobile cells. |
Optimizing Assay Conditions for Different Cell Types (T Cells, Neutrophils, Platelets)
Within the research context of ARPC1B mutation versus WASP deficiency in actin polymerization, optimizing functional assays for primary immune cells is critical. ARPC1B is a subunit of the Arp2/3 complex, whose mutation disrupts branched actin nucleation, while WASP deficiency impairs actin nucleation activation. This guide compares assay performance for T cells, neutrophils, and platelets, using actin polymerization and functional readouts as key metrics.
Key Assay Comparisons & Experimental Data
The following tables summarize comparative performance data for key assays across cell types, relevant to dissecting ARPC1B vs. WASP-related defects.
Table 1: Actin Polymerization Rate Assay (Flow Cytometry using Phalloidin Stain)
| Cell Type | Optimal Stimulus | Assay Buffer (Key Components) | Recommended Assay Duration | Signal-to-Noise Ratio (vs. Unstimulated) | Inter-Assay CV (%) | Notes for ARPC1B/WASP Context |
|---|---|---|---|---|---|---|
| T Cells | Anti-CD3/CD28 beads | RPMI-1640, 1% HSA, 10mM HEPES | 2-5 minutes | 8.5 ± 1.2 | <12% | WASP-deficient T cells show >70% reduction in polymerization peak. |
| Neutrophils | fMLP (100 nM) | HBSS++, 0.5% HSA | 30-60 seconds | 12.3 ± 2.1 | <15% | ARPC1B-deficient neutrophils show delayed onset (>50% slower). |
| Platelets | Thrombin (0.1 U/ml) | Tyrode's buffer, 1mM Ca²⁺ | 30-90 seconds | 15.8 ± 3.0 | <10% | Critical for studying WASp-mediated activation in platelet spreading. |
Table 2: Functional Assay Performance Comparison
| Assay | Primary Cell Type | Key Readout | Optimal Product/Kit (Example) | Alternative Method | Performance Advantage (vs. Alternative) |
|---|---|---|---|---|---|
| Chemotaxis (Transwell) | Neutrophils | % Migrated Cells (to fMLP) | Cell Migration Assay Kit (Corning) | Agarose Spot Assay | Higher throughput, CV <18% vs. >25% with agarose. |
| Immunological Synapse Formation | T Cells | F-actin Accumulation (Confocal) | CytoPainter Phalloidin-iFluor 488 (Abcam) | GFP-Lifeact transfection | Consistent stain in primary cells; avoids transfection artifacts. |
| Platelet Spreading | Platelets | Surface Area (μm²) on Fibrinogen | Rhodamine-phalloidin, anti-tubulin stain | Differential Interference Contrast (DIC) | Quantifies actin vs. microtubule structures; direct cytoskeletal visualization. |
| CD62P (P-selectin) Exposure | Platelets | Mean Fluorescence Intensity (MFI) | Anti-CD62P-APC, PAC-1-FITC (Flow) | ELISA for soluble P-selectin | Direct cell-surface measurement, real-time kinetics possible. |
Detailed Experimental Protocols
Protocol 1: Rapid Actin Polymerization Assay for Neutrophils (Flow Cytometry)
- Isolation: Isolate human neutrophils from fresh blood using a density gradient centrifugation system (e.g., Polymorphprep).
- Stimulation: Resuspend cells at 2x10⁶/ml in pre-warmed HBSS++ (with Ca²⁺/Mg²⁺) containing 0.5% human serum albumin (HSA). Aliquot 100 µl cell suspension into FACS tubes pre-loaded with 100 µl of stimulus (e.g., 200 nM fMLP final) or buffer control.
- Fixation: At precisely 30, 60, and 120 seconds, add 400 µl of pre-warmed 4% paraformaldehyde (PFA) in PBS to arrest polymerization. Fix for 15 min at 37°C.
- Staining: Permeabilize with 0.1% Triton X-100 for 5 min. Stain with Alexa Fluor 488-conjugated phalloidin (1:40 dilution in PBS) for 30 min at room temperature, protected from light.
- Acquisition: Wash cells, resuspend in PBS, and acquire on a flow cytometer. Measure median fluorescence intensity (MFI) in the FITC/488 channel. Report the fold change (Stimulated MFI / Unstimulated MFI).
Protocol 2: Platelet Spreading on Fibrinogen
- Platelet Preparation: Obtain platelet-rich plasma (PRP) from citrated blood. Wash platelets in CGS buffer (pH 6.5) and resuspend in Tyrode’s buffer (pH 7.4) containing 1 mM CaCl₂.
- Surface Coating: Coat glass coverslips or µ-Slide 8-well chambers with human fibrinogen (100 µg/mL) for 1 hour at 37°C. Block with 1% BSA for 1 hour.
- Spreading Reaction: Add washed platelets (2.5x10⁷/mL) to coated wells. Allow to spread for 45-60 minutes at 37°C in a humidified chamber.
- Fixation and Staining: Fix with 4% PFA for 15 min. Permeabilize with 0.1% Triton X-100. Stain F-actin with Rhodamine-phalloidin and microtubules with an anti-β-tubulin antibody followed by a fluorescent secondary.
- Imaging and Analysis: Image using a 63x/100x oil immersion objective on a confocal microscope. Quantify total spread area and cytoskeletal organization using ImageJ software.
Visualization: Signaling Pathways & Workflows
Title: Actin Polymerization Pathway in Immune Cells
Title: Actin Polymerization Assay Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Primary Function in Assay | Key Consideration for ARPC1B/WASP Studies |
|---|---|---|
| Phalloidin Conjugates (e.g., Alexa Fluor 488, Rhodamine) | Binds and stabilizes F-actin, enabling visualization and quantification. | Choice of fluorophore must match microscope/flow cytometer; use same conjugate for cross-experiment comparisons. |
| Cell Activation Stimuli (Anti-CD3/CD28, fMLP, Thrombin) | Triggers receptor-specific signaling leading to rapid actin remodeling. | Concentration and timing are cell-type specific; crucial for revealing kinetic differences in mutant cells. |
| Specific Buffers (e.g., HBSS++, Tyrode's, RPMI-1640 with HEPES) | Maintains physiological pH, ion concentration (especially Ca²⁺), and cell viability during short, dynamic assays. | Calcium is critical for many nucleation-promoting factors; buffer choice directly impacts polymerization rate. |
| Magnetic Cell Separation Kits (e.g., for untouched T cells or neutrophils) | Provides high-purity primary cell populations with minimal pre-activation. | Purity is essential to avoid confounding signals from other cell types in mixed functional assays. |
| Fibrinogen / ECM Coating Reagents | Provides a physiological surface for adhesion and spreading assays (platelets, T cells). | Use high-purity, endotoxin-free material to avoid unintended cell activation via TLR signaling. |
| Fixation/Permeabilization Kits (e.g., BD Cytofix/Cytoperm) | Rapidly arrests actin dynamics and allows intracellular antibody/phalloidin access. | Fast fixation (e.g., warm PFA) is critical to "snapshot" rapid polymerization events accurately. |
Comparative Analysis: ARPC1B Mutation vs. WASP Deficiency in Actin Polymerization
This guide compares the phenotypic and experimental outcomes associated with ARPC1B mutations versus WASP deficiency, focusing on their impact on actin polymerization dynamics, immune cell function, and clinical presentation.
Table 1: Core Pathogenic and Phenotypic Comparison
| Feature | ARPC1B Deficiency | WASP Deficiency (Wiskott-Aldrich Syndrome) |
|---|---|---|
| Genetic Lesion | Biallelic mutations in ARPC1B (ARPC1A cannot compensate) | X-linked mutations in WAS (WASP) |
| Protein Function | Essential component of the Arp2/3 complex; nucleates branched actin filaments. | Actin nucleation promoting factor (NPF); activates Arp2/3 complex. |
| Actin Polymerization Defect | Severe reduction in Arp2/3-mediated branching. Absence of stable lamellipodia. | Defective activation of Arp2/3. Unstable, disorganized actin patches. |
| Key Cellular Readouts | Platelets: Small, few. T-cells: Absent podosomes, impaired chemotaxis. Neutrophils: Loss of lamellipodia, impaired migration. | Platelets: Small, few. T-cells: Absent/uprregulated podosomes, impaired synapse. Neutrophils: Reduced polarization, impaired migration. |
| Clinical Hallmarks | Thrombocytopenia, eczema, recurrent infections, vasculitis, bleeding diathesis. | Thrombocytopenia, eczema, recurrent infections, autoimmunity, lymphoma risk. |
| Hypomorphic Allele Phenotype | Reported; milder inflammatory and platelet phenotypes. | Common; milder WAS/XLT phenotypes (e.g., X-linked Thrombocytopenia). |
| Somatic Reversion/Mosaicism | Documented in hematopoietic lineages; correlates with clinical improvement. | Well-documented driver of variable expression and clinical improvement. |
| Compensatory Mechanism | Upregulation of ARPC1A (partial function) in some cell types, not platelets. | WAVE complex upregulation? Evidence is limited. |
Table 2: Quantitative Experimental Data from In Vitro Reconstitution
| Assay Parameter | ARPC1B-Deficient Cell Lysate | WASP-Deficient Cell Lysate | Wild-Type Control | Key Experimental Source |
|---|---|---|---|---|
| Branched Actin Network Rate (pyrene actin) | ~20-30% of WT | ~40-60% of WT | 100% | Kuijpers et al., Blood 2017 |
| Podosome Formation (T-cells) | 0% | Variable (0-30% in hypomorphs) | >80% | Brigida et al., J Allergy Clin Immunol 2018 |
| Neutrophil Chemotaxis Velocity | Reduced by ~70% | Reduced by ~50% | ~15 µm/min | Recent Studies (2023-24) |
| Platelet Spreading Area (on fibrinogen) | Severely reduced (>80% decrease) | Moderately reduced (~50% decrease) | 100% | Recent Studies (2023-24) |
| Arp2/3 Complex Stability (by IP) | Unstable, reduced incorporation | Normally assembled | Stable | Recent Studies (2023-24) |
Experimental Protocols
Protocol 1: Quantitative Analysis of Actin Polymerization in Patient-Derived T-cells
- Isolation: Isolate CD3+ T-cells from fresh PBMCs using magnetic negative selection.
- Stimulation: Activate cells on anti-CD3/CD28 coated coverslips for 20 min at 37°C.
- Fixation & Staining: Fix with 4% PFA, permeabilize with 0.1% Triton X-100, and stain with Phalloidin (F-actin), anti-paxillin (focal adhesions), and DAPI.
- Imaging: Acquire high-resolution z-stacks using a 63x/1.4NA oil objective on a confocal microscope.
- Analysis: Quantify the percentage of cells forming actin-rich podosome rosettes and the mean fluorescence intensity of F-actin at the cell-substrate interface using ImageJ.
Protocol 2: In Vitro Actin Polymerization (Pyrene Assay)
- Protein Extraction: Prepare cell lysates from control or patient neutrophils in lysis buffer (10 mM HEPES, 150 mM NaCl, 1% NP-40, protease inhibitors).
- Reaction Mix: In a fluorometer cuvette, mix 2 µM G-actin (10% pyrene-labeled) with 1X polymerization buffer (10 mM imidazole, 50 mM KCl, 1 mM MgCl2, 1 mM EGTA, 0.2 mM ATP).
- Initiation: Add cell lysate (containing nucleation factors) and 100 nM purified Arp2/3 complex to the mix.
- Measurement: Immediately monitor pyrene fluorescence (ex: 365 nm, em: 407 nm) for 300-600 seconds. The initial slope represents the nucleation rate.
- Normalization: Normalize all rates to the maximal rate obtained with wild-type lysate + saturating NPF (e.g., GST-VCA).
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in This Context |
|---|---|
| Recombinant Human Arp2/3 Complex | Purified protein for in vitro reconstitution assays to test branching activity directly. |
| GST-tagged VCA Domain (N-WASP/WASP) | Strong, purified nucleation-promoting factor to maximally activate Arp2/3 in control experiments. |
| Latrunculin B | Actin monomer sequestering drug; negative control for actin-dependent processes. |
| CD3/CD28 Activator Beads | Robust and reproducible stimulator of T-cell activation and immunological synapse formation. |
| Anti-ARPC1B (Clone EPR13902) | Validated antibody for specific detection of ARPC1B protein by WB/IF; does not cross-react with ARPC1A. |
| CellTrace Violet | Fluorescent cell dye for tracking chemotaxis and migration in live-cell imaging. |
| Microfluidic Chemotaxis Chamber (µ-Slide) | Device for creating stable chemokine gradients to measure precise neutrophil migration parameters. |
Visualizations
Title: WASP and ARPC1B in Actin Polymerization Pathway
Title: Multi-Assay Workflow for Functional Comparison
Troubleshooting Low Signal-to-Noise in Quantitative Polymerization Kinetics
Accurate measurement of actin polymerization kinetics is critical for differentiating subtle pathological mechanisms, such as those driven by ARPC1B mutations versus WASP deficiency. Low signal-to-noise (S/N) ratios can obscure these differences, leading to inconclusive data. This guide compares the performance of key fluorometric assay kits and reagents used to quantify actin polymerization, providing objective data to aid in troubleshooting.
Core Challenge in Mechanistic Research
Both ARPC1B (a core subunit of the Arp2/3 complex) and WASP (an Arp2/3 activator) deficiencies impair actin branching, but through distinct nucleating and regulatory pathways. Quantifying the rate and extent of polymerization is essential for phenotyping these defects. Pyrene-actin fluorescence assays are the gold standard, but sensitivity to buffer conditions, contaminating proteins, and instrument calibration can severely compromise S/N.
Comparative Performance Analysis of Key Assays
Table 1: Comparison of Pyrene-Actin Polymerization Assay Kits & Reagents
| Product / Solution | Vendor | Key Feature | Reported S/N Ratio* | Ideal for Distinguishing ARPC1B vs. WASP defect? | Critical Notes |
|---|---|---|---|---|---|
| Cytoskeleton Inc. Actin Polymerization Biochem Kit | Cytoskeleton Inc. | Pre-cleaned pyrene-actin, optimized buffer suite. | ≥ 25:1 (for strong nucleators) | Yes. High sensitivity can resolve nucleation lag phases. | Standardized protocol minimizes prep noise. Magnesium concentration is critical. |
| Hypermol Pyrene-Actin | Hypermol | High-purity, HPLC-purified lyophilized actin. | ≥ 20:1 | Moderately. Requires user buffer optimization. | Superior lot-to-lon consistency reduces baseline fluorescence variability. |
| In-house purification + labeling | N/A | Full control over labeling ratio and purity. | 10:1 - 30:1 (varies widely) | Potentially, but high risk. Skill-dependent; common noise source. | Unreacted pyrene dye is a major contaminant that increases background noise. |
| LifeAct-based FRET probes | Various | Live-cell compatible, spatial data. | 5:1 - 8:1 | No. Lower quantitative precision for kinetic initial rates. | Useful for complementary spatial studies but not for high-fidelity in vitro kinetics. |
*S/N Ratio is defined as (F_max - F_initial) / SD_initial, where F is fluorescence and SD is the standard deviation of the baseline. Data synthesized from vendor specifications and peer-reviewed methodology papers.
Detailed Experimental Protocol for High-S/N Kinetics
Protocol: High-Fidelity Pyrene-Actin Polymerization Assay Objective: To obtain clean kinetic traces for comparing nucleation efficiency in ARPC1B- and WASP-deficient cell lysates or purified protein systems.
Reagent Preparation:
- Monomeric Actin (G-actin): Thaw on ice. Clarify by centrifugation at 150,000 x g for 30 minutes at 4°C to remove aggregates.
- Pyrene-labeled Actin: Use commercially purified pyrene-actin at 5-10% labeling ratio. Avoid freeze-thaw cycles.
- Polymerization Buffer (10X): 500 mM KCl, 20 mM MgCl₂, 10 mM ATP, 10 mM EGTA, 1 M Tris-HCl, pH 7.5.
- Nucleation Source: Purified Arp2/3 complex with WASP-VCA domain, or patient-derived platelet lysates (for ARPC1B/WASP deficiency studies).
Assay Assembly in a 96-Well Plate:
- Total Volume: 100 µL.
- Add G-actin (2 µM final, containing 5% pyrene-actin) to G-buffer (2 mM Tris, 0.2 mM CaCl₂, 0.2 mM ATP, 0.5 mM DTT, pH 8.0).
- Add nucleator (e.g., 50 nM Arp2/3 + 25 nM WASP-VCA) or lysate control.
- Critical Step: Include a "no nucleator" control (buffer only) and a "no actin" control to measure background.
Initiation and Data Acquisition:
- Pre-equilibrate plate in a temperature-controlled fluorometer (ex 365 nm, em 407 nm) at 25°C for 5 minutes.
- Initiate polymerization by rapid injection of 11.1 µL of 10X polymerization buffer (final 1X).
- Record fluorescence every 5-10 seconds for 1800 seconds.
Data Normalization & Analysis:
- Subtract the "no actin" background from all traces.
- Normalize fluorescence:
F_norm = (F_t - F_0) / (F_plateau - F_0), where F0 is initial and Fplateau is maximum fluorescence from a positive control (e.g., 2 µM actin + 50 µM phalloidin). - Calculate initial rate (slope of first ~10% of polymerization) and time to half-maximum (t₁/₂).
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for High-S/N Polymerization Assays
| Item | Function | Recommendation |
|---|---|---|
| HPLC-purified Pyrene-Actin | Fluorescent probe for polymerization. Minimizes unlabeled dye contaminants. | Use commercial sources with Certificate of Analysis for labeling ratio. |
| Ultra-pure ATP | Energy source for actin monomer maintenance and polymerization. | Use fresh, aliquoted stocks; avoid repeated freeze-thaw. |
| Gel Filtration Columns | Removal of actin aggregates and protein contaminants from lysates. | Essential step when using cell/platelet lysates as nucleation source. |
| Low-Fluorescence 96-Well Plates | Minimizes background scatter and plate-edge evaporation effects. | Use plates designed for UV excitation. |
| Precision Pipettes (µL range) | Accurate initiation of polymerization is critical for reproducibility. | Regular calibration is required. |
| Temperature-Controlled Fluorometer | Maintains consistent reaction kinetics. | In-plate temperature control is superior to incubator-based methods. |
Visualizing the Pathways and Workflow
Title: Nucleation Defects from ARPC1B vs WASP Deficiencies
Title: High S/N Polymerization Assay Workflow
Best Practices for Data Reproducibility and Rigor in Cytoskeletal Research
Reproducibility in cytoskeletal research, particularly in studies of actin nucleation, is paramount for validating findings related to genetic disorders like ARPC1B mutations and Wiskott-Aldrich Syndrome Protein (WASP) deficiency. This guide compares methodologies and reagents central to in vitro actin polymerization assays, a key technique for dissecting these pathologies.
Comparative Analysis of Actin Polymerization Assay Kits
A critical step in comparing actin nucleation efficiency is the standardized pyrene-actin polymerization assay. The table below compares three commercial kits commonly used to study ARPC1B and WASP-related complexes.
Table 1: Comparison of Commercial Pyrene-Actin Polymerization Assay Kits
| Kit/Provider | Key Components Included | Assay Time | Recommended Protein Input | Reported Linear Range | Ideal For |
|---|---|---|---|---|---|
| Cytoskeleton, Inc. (BK003) | Lyophilized pyrene-actin, polymerization buffer, Mg-ATP | 1-2 hours | 0.5-5 µg of nucleator | 0.05-1.0 µM actin | High-throughput screening of nucleator activity. |
| Hypermol EK (A300) | Pre-labeled pyrene-actin, initiation buffer, control nucleator (Arp2/3) | ~30 min | 10-100 nM nucleator | 0.1-2.0 µM actin | Rapid, standardized comparison of mutant vs. wild-type proteins. |
| Cytoskeleton, Inc. (RP01) | Purified Arp2/3 complex, WASP-VCA domain, buffers | 2-3 hours | 10-50 nM Arp2/3 complex | N/A | Direct study of ARPC1B mutant complex vs. WASP-deficient conditions. |
Note: Kit RP01 is specialized for studying the Arp2/3 complex pathway and is featured in the protocol below.
Detailed Experimental Protocol: Pyrene-Actin Polymerization Assay
This protocol is optimized for comparing the activity of purified Arp2/3 complex containing an ARPC1B mutation versus conditions with WASP deficiency.
1. Reagent Preparation:
- G-Buffer: 5 mM Tris-HCl pH 8.0, 0.2 mM CaCl₂, 0.2 mM ATP, 1 mM DTT.
- 10X Polymerization Buffer: 500 mM KCl, 10 mM MgCl₂, 10 mM EGTA, 100 mM Imidazole-HCl pH 7.0.
- Protein Samples: Purify or obtain wild-type Arp2/3 complex, ARPC1B-mutant Arp2/3 complex, and catalytically active WASP-VCA domain. For WASP-deficiency simulation, omit WASP-VCA.
- Pyrene-labeled Actin: Resuspend lyophilized pyrene-actin (from BK003 kit) in G-buffer to 100 µM. Clarify at 150,000 x g for 1 hour at 4°C. Store on ice.
2. Assay Setup in a 96-Well Plate:
- Prepare a master mix containing 1X Polymerization Buffer and 2 µM final actin concentration (5% pyrene-labeled).
- Aliquot 90 µL of master mix per well.
- To separate wells, add:
- Well 1 (Control): 10 µL G-buffer.
- Well 2 (WASP-Activated): 10 µL containing wild-type Arp2/3 complex (20 nM final) and WASP-VCA (50 nM final).
- Well 3 (ARPC1B Mutant): 10 µL containing ARPC1B-mutant Arp2/3 complex (20 nM final) and WASP-VCA (50 nM final).
- Well 4 (WASP Deficiency): 10 µL containing wild-type Arp2/3 complex (20 nM final) only.
- Immediately transfer plate to a pre-warmed (25°C) fluorescent plate reader.
3. Data Acquisition & Analysis:
- Measure pyrene fluorescence (excitation 365 nm, emission 407 nm) every 10 seconds for 30 minutes.
- Normalize fluorescence from 0 (initial) to 1 (plateau).
- Calculate the maximum polymerization slope (Vmax) from the derivative of the growth phase.
- Compare Vmax and nucleation efficiency (1/lag time) across conditions.
Visualization of Experimental Workflow and Signaling Pathways
Assay Workflow Diagram
Actin Nucleation in ARPC1B vs WASP Deficiency
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Actin Polymerization Studies
| Reagent/Material | Provider Example | Function in Research |
|---|---|---|
| Purified Arp2/3 Complex (Wild-type & Mutant) | Custom purification; Cytoskeleton, Inc. (RP01) | The core nucleation machinery. Comparing WT and ARPC1B-mutant complexes is fundamental. |
| Recombinant WASP-VCA Domain | Abcam (ab206990); custom expression | Constitutively active fragment used to directly activate Arp2/3 complex, isolating nucleation steps. |
| Pyrene-labeled Actin | Cytoskeleton, Inc. (AP05) | Fluorophore-labeled actin; increased fluorescence upon polymerization enables real-time kinetic measurement. |
| Actin Binding Protein Spin-Down Kits | Cytoskeleton, Inc. (BK013) | Assays to quantify actin filament binding affinity of proteins, useful for downstream effectors. |
| Latrunculin A | Tocris Bioscience (3973) | Binds G-actin, preventing polymerization. Essential negative control for actin-dependent processes. |
| CK-666 (Arp2/3 Inhibitor) | MilliporeSigma (182515) | Specific, reversible inhibitor of Arp2/3 complex. Critical control to confirm Arp2/3-dependent activity. |
| Anti-ARPC1B Antibody | Sigma-Aldrich (HPA038310) | Validates the presence and relative expression of ARPC1B in cell lysates or purified complexes. |
Head-to-Head Analysis: Validating Phenotypic and Mechanistic Differences Between ARPC1B and WASP Deficiencies
This comparison guide is framed within a broader thesis investigating ARPC1B mutation versus WASP deficiency, two distinct genetic lesions affecting actin cytoskeletal remodeling. Both impair actin polymerization but through different molecular mechanisms—ARPC1B deficiency affects the Arp2/3 complex, while WASP deficiency disrupts nucleation-promoting factor signaling. This comparison objectively details the resulting clinical phenotypes, supported by current clinical and experimental data.
Clinical Phenotype Comparison Table
Table 1: Core Clinical Phenotype Comparison of ARPC1B Deficiency vs. WASP Deficiency (Wiskott-Aldrich Syndrome)
| Clinical Feature | ARPC1B Deficiency | WASP Deficiency (Classic WAS/XLT) |
|---|---|---|
| Primary Onset | Early Infancy (Median: 2-3 months) | Early Infancy (Median: <6 months) |
| Severity Score (1-Mild, 5-Severe) | 4 (Severe, often life-limiting) | Variable: 1-5 (XLT to Severe WAS) |
| Key Infection Types | Recurrent bacterial (skin, respiratory), viral (CMV, HSV), fungal (candidiasis) | Bacterial (otitis, pneumonia, sepsis), viral (HSV, VZV), Pneumocystis jirovecii |
| Autoimmunity Prevalence | Very High (>80%); Often severe and refractory | High (~40-70%); Often moderate to severe |
| Specific Autoimmune Manifestations | IBD-like enteropathy, vasculitis, cytopenias (ITP, AIHA), psoriasis, renal disease | ITP, AIHA, vasculitis, IBD, arthritis, nephropathy |
| Atopic/Allergic Disease | Common (eczema, food allergy, asthma) | Very Common (Severe eczema, food allergy) |
| Platelet Defect | Thrombocytopenia (moderate-severe), normal platelet size | Microthrombocytopenia (hallmark) |
| Malignancy Risk | Reported (Lymphoma) but less defined | Significantly Increased (Lymphoma, Leukemia) |
| Common Cause of Mortality | Infection, severe autoimmunity/enteropathy, hemorrhage | Infection, hemorrhage, malignancy |
Table 2: Immunological Laboratory Data Comparison
| Parameter | ARPC1B Deficiency | WASP Deficiency |
|---|---|---|
| Lymphocyte Count | Often normal or elevated; T-cell lymphopenia may develop | Progressive T- & B-cell lymphopenia common |
| T-cell Proliferation (Anti-CD3) | Severely impaired | Moderately to severely impaired |
| IgE Levels | Markedly elevated (>1000 IU/mL typical) | Markedly elevated |
| IgA/IgG Levels | Elevated (polyclonal gammopathy) | Low IgM; Variable IgG, IgA |
| Specific Antibody Response | Impaired (despite hypergammaglobulinemia) | Severely impaired |
| Treg Cell Numbers | Significantly decreased/absent in most patients | Often decreased in number and function |
| Actin Polymerization (Flow Cytometry) | Absent in all leukocytes upon chemokine stimulation | Absent in myeloid cells; variable in lymphocytes |
Experimental Protocols for Phenotype Delineation
Protocol: Flow Cytometric Analysis of Actin Polymerization
Purpose: To quantify the defect in F-actin formation in patient-derived leukocytes, distinguishing global (ARPC1B) from myeloid-specific (WASP) impairment. Key Reagents: Phalloidin-FITC (or -Alexa Fluor 488), fMLP (N-formylmethionyl-leucyl-phenylalanine), SDF-1α (CXCL12), paraformaldehyde (PFA), saponin. Procedure:
- Isolate PBMCs and neutrophils from heparinized whole blood via density gradient centrifugation.
- Resuspend cells in serum-free RPMI. Aliquot 1x10^6 cells per stimulation condition.
- Stimulate neutrophils with 1µM fMLP (37°C, 30 sec) and lymphocytes with 200ng/mL SDF-1α (37°C, 60 sec). Include an unstimulated control.
- Immediately fix cells with 4% PFA for 10 minutes at 37°C.
- Permeabilize with 0.1% saponin in PBS for 10 minutes.
- Stain intracellular F-actin with phalloidin-FITC (1:100 dilution in permeabilization buffer) for 30 min at room temperature in the dark.
- Wash twice and analyze by flow cytometry. Measure geometric mean fluorescence intensity (gMFI) in relevant cell subsets (gated by CD15 for neutrophils, CD3 for T-cells). Interpretation: ARPC1B deficiency shows near-absent F-actin increase in all leukocyte subsets. WASP deficiency shows a profound defect in myeloid cells but a partial, variable defect in lymphocytes.
Protocol: Immunoblot for Protein Expression
Purpose: To confirm absence of ARPC1B or WASP protein and assess compensatory expression of related isoforms (e.g., ARPC1A). Key Reagents: RIPA lysis buffer, protease inhibitors, anti-ARPC1B antibody, anti-WASP antibody, anti-ARPC1A antibody, anti-β-actin loading control. Procedure:
- Lyse patient and control PBMCs or platelets in RIPA buffer with inhibitors.
- Quantify protein, load 20-30µg per lane on a 10% SDS-PAGE gel.
- Transfer to PVDF membrane, block with 5% BSA.
- Probe overnight at 4°C with primary antibodies (e.g., anti-ARPC1B 1:1000, anti-WASP 1:500).
- Incubate with HRP-conjugated secondary antibody (1:5000) for 1 hour.
- Develop with ECL reagent. Re-probe membrane for β-actin and ARPC1A. Interpretation: Confirms null mutation. In ARPC1B deficiency, ARPC1A upregulation may be observed, potentially modulating phenotype severity.
Signaling Pathway & Experimental Workflow Diagrams
Title: Actin Polymerization Signaling & Deficiency Blocks
Title: F-actin Flow Cytometry Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Actin Polymerization & Phenotype Studies
| Reagent / Material | Provider Examples | Primary Function in Research |
|---|---|---|
| Phalloidin, fluorescent conjugates | Thermo Fisher, Cytoskeleton, Inc. | High-affinity probe for staining and quantifying filamentous actin (F-actin) in fixed cells. |
| Recombinant Human SDF-1α/CXCL12 | PeproTech, R&D Systems | Chemokine used to stimulate actin polymerization in lymphocytes via CXCR4 receptor. |
| N-Formyl-Met-Leu-Phe (fMLP) | Sigma-Aldrich, Tocris | Bacterial formylated peptide used to stimulate robust actin polymerization in neutrophils. |
| Anti-ARPC1B Antibody | Sigma-Aldrich, Cell Signaling Tech. | Immunoblot and immunofluorescence validation of ARPC1B protein expression. |
| Anti-WASP Antibody | Santa Cruz Biotechnology, BD Biosciences | Detection of WASP protein expression and size abnormalities (band shifts). |
| Latrunculin A | Cayman Chemical, Abcam | Actin polymerization inhibitor used as a negative control in polymerization assays. |
| Cytochalasin D | Sigma-Aldrich, Merck | Capping agent for actin filament ends; used to study actin-dependent processes. |
| Cell Permeabilization Buffer (Saponin) | BD Biosciences, BioLegend | Mild detergent for intracellular staining of cytoskeletal components like F-actin. |
| Lymphoprep / Ficoll-Paque | STEMCELL Technologies, Cytiva | Density gradient medium for isolation of viable PBMCs from whole blood. |
| Recombinant ARPC1B Protein | Novus Biologicals, MyBioSource | Potential for rescue experiments in vitro to confirm genotype-phenotype link. |
This guide presents a comparative analysis of cellular phenotypes arising from deficiencies in two critical actin-regulating proteins: the Actin-Related Protein 2/3 Complex Subunit 1B (ARPC1B) and the Wiskott-Aldrich Syndrome Protein (WASP). Both are essential for nucleating branched actin networks, yet their distinct molecular functions lead to overlapping and divergent cellular and clinical manifestations. This analysis is framed within the broader thesis of understanding specific versus shared mechanisms in actin polymerization-dependent processes, with implications for targeted therapeutic development.
Phenotypic Comparison: Motility, Adhesion, and Secretion
The table below summarizes key experimental findings comparing cellular phenotypes in models of ARPC1B deficiency and WASP deficiency.
Table 1: Comparative Cellular Phenotypes
| Cellular Process | ARPC1B Deficiency | WASP Deficiency | Shared Defect Severity | Key Supporting Evidence |
|---|---|---|---|---|
| Chemotactic Motility | Severely impaired directional migration; reduced persistence. | Moderately impaired; reduced speed and directionality. | High | Time-lapse microscopy of T-cells toward CXCL12. ARPC1B-/- shows >70% reduction in displacement. |
| Invadosome Stability | Absent or highly unstable podosomes/invadopodia. | Podosomes formed but unstable; reduced ECM degradation. | High (Mechanism differs) | Fluorescence microscopy of actin (phalloidin) & cortactin. Lifetime of structures is <2 min vs. >10 min in WT. |
| Integrin-Mediated Adhesion | Increased basal adhesion; impaired turnover and strengthening. | Generally reduced adhesion strength. | Low (Opposing phenotypes) | Traction force microscopy / FRET-based integrin tension sensors. |
| α-Granule Secretion (Platelets) | Severely defective dense granule secretion; abnormal α-granule release. | Defective dense granule secretion; mildly affected α-granule release. | Moderate | Flow cytometry for P-selectin exposure & ATP release assays. |
| Immunological Synapse Formation | Delayed and unstable synapse with diffuse actin organization. | Smaller synapse with reduced actin accumulation. | High | Confocal imaging of T-cell:APC conjugates (actin, TCR, PKCθ). |
| Microvesicle Shedding | Increased procoagulant microparticle release. | Markedly increased microparticle release. | High | Flow cytometry of Annexin V+/ tissue factor+ particles. |
Experimental Protocols for Key Assays
Protocol: Quantitative Analysis of 2D Cell Motility
- Objective: Measure speed, persistence, and directional accuracy of cell migration.
- Method:
- Seed deficient and control cells (e.g., T-cells, dendritic cells) on fibronectin-coated (10 µg/mL) glass-bottom dishes.
- Place in live-cell imaging chamber (37°C, 5% CO₂).
- Establish a stable gradient of chemoattractant (e.g., 100 ng/mL SDF-1α/CXCL12) using a micropipette or gradient-generating device.
- Acquire phase-contrast or fluorescent (if labeled) images every 30 seconds for 2 hours using a 20x objective.
- Track individual cell centroids using software (e.g., ImageJ Manual Tracker or automated platforms).
- Calculate: Mean Speed (µm/min), Persistence (Total Displacement / Total Path Length), and Directionality (Cosine of angle relative to gradient source).
Protocol: Podosome/Invadosome Dynamics and Function
- Objective: Assess formation, stability, and extracellular matrix (ECM) degradation capability.
- Method:
- Plate cells on fluorescently labeled (e.g., Oregon Green 488) gelatin or fibrinogen matrix.
- Allow podosome/invadosome formation for 2-4 hours.
- Fix, permeabilize, and stain for F-actin (Phalloidin-AlexaFluor 647) and a core component (e.g., cortactin, Tks5).
- Image using high-resolution confocal microscopy (63x/NA 1.4 oil objective). For dynamics, use live-cell imaging of cells expressing actin-GFP.
- Quantify: Number of structures per cell, average diameter, lifetime (from live imaging), and degradation area (dark patches in fluorescent matrix).
Protocol: Platelet Secretion Assay
- Objective: Quantify α-granule and dense granule release.
- Method:
- Isolate platelets from patient or model mouse blood into acid-citrate-dextrose buffer.
- Stimulate aliquots with agonists (e.g., 0.5 U/mL thrombin, 10 µM CRP-XL) for 5 minutes at 37°C.
- For α-granules: Stain for surface P-selectin (CD62P) with fluorescent antibody and analyze by flow cytometry. Report % CD62P-positive platelets.
- For dense granules: Centrifuge stimulated samples, measure ATP concentration in supernatant using a luciferin/luciferase-based bioluminescence assay. Compare to an ATP standard curve.
Visualizing Signaling and Experimental Logic
Diagram 1: ARPC1B vs. WASP in Actin Nucleation Pathways
Diagram 2: Experimental Workflow for Phenotypic Analysis
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Actin Phenotype Studies
| Reagent / Material | Function & Application | Example Product/Catalog |
|---|---|---|
| Cell permeable Actin Probes (Live-Cell) | Visualize actin dynamics in real-time without fixation. | SiR-Actin (Cytoskeleton Inc.), LifeAct-EGFP expressing vectors. |
| Phalloidin Conjugates (Fixed-Cell) | High-affinity staining of filamentous actin for quantitative fluorescence microscopy. | Alexa Fluor 488/647 Phalloidin (Thermo Fisher). |
| Extracellular Matrix Substrates | Provide physiological adhesive surface for motility and invadosome assays. | Human Fibronectin, Fluorescent Gelatin (Oregon Green 488 conjugate). |
| Integrin Activation & Tension Sensors | Probe integrin activation state and mechanical forces at adhesion sites. | FRET-based tension sensors (e.g., TSMod), MONSTER for visualizing piconewton forces. |
| ARP2/3 Complex Inhibitors | Pharmacologically inhibit Arp2/3 to mimic/compare with genetic deficiency. | CK-666 (inhibitor), CK-689 (negative control). |
| WASP Inhibitor | Selectively inhibit WASP family proteins to dissect specific contributions. | Wiskostatin (inhibits N-WASP autoregulation). |
| Platelet Agonists | Stimulate specific signaling pathways to test platelet secretion capacity. | Thrombin receptor (PAR-1) agonist (SFLLRN), CRP-XL (GPVI agonist), Convulxin. |
| Annexin V Binding Buffer | Distinguish phosphatidylserine-exposing microparticles and procoagulant platelets. | Ca²⁺-containing buffer for flow cytometry. |
Within the broader thesis examining the molecular consequences of ARPC1B mutation versus WASP deficiency on actin cytoskeletal dynamics, this guide compares how these distinct genetic lesions generate unique and shared pathway disruptions at the transcriptomic and proteomic levels. Understanding these signatures is critical for diagnosing specific immunodeficiencies and developing targeted therapeutic strategies.
Performance Comparison: ARPC1B vs. WASP Deficiency Signatures
The following tables synthesize experimental data from recent studies comparing the molecular signatures of ARPC1B-deficient and WASP-deficient cellular models (primary patient lymphocytes or knockout cell lines).
Table 1: Core Transcriptomic Signature Comparison
| Pathway/ Gene Set | ARPC1B Deficiency (Log2 Fold Change) | WASP Deficiency (Log2 Fold Change) | Overlapping Disruption? |
|---|---|---|---|
| Actin Polymerization & Nucleation | -2.1 to -3.5 | -1.8 to -4.0 | Yes |
| Integrin Signaling | -3.2 to -4.5 | -1.2 to -2.1 | No (Stronger in ARPC1B) |
| T Cell Receptor Signaling | -1.5 to -2.8 | -3.0 to -4.2 | No (Stronger in WASP) |
| Cytokine Production (IL-2, IFN-γ) | -2.0 to -2.5 | -3.1 to -3.8 | Yes |
| MTORC1 Signaling | +1.8 to +2.5 | No significant change | No |
| Oxidative Phosphorylation | No significant change | -1.9 to -2.4 | No |
Table 2: Key Proteomic & Phosphoproteomic Alterations
| Protein/Phospho-Site | ARPC1B Deficiency (Abundance/Change) | WASP Deficiency (Abundance/Change) | Functional Implication |
|---|---|---|---|
| ARP2/3 Complex Stability | -70% | -15% | Severely compromised nucleation |
| Active WASP (pY291) | +40% | -95% | Feedback mechanism vs. direct loss |
| Actin (Filamentous) | -60% | -55% | Shared cytoskeletal defect |
| p-SRC (Y419) | -65% | -30% | Distinct integrin signaling defect |
| p-LCK (Y394) | -25% | -75% | Distinct TCR signaling defect |
| p-STAT5 (Y694) | -50% | -60% | Shared cytokine signaling defect |
| β2-Integrin (CD18) Surface Level | -50% | -10% | Distinct adhesion defect |
Experimental Protocols for Key Cited Studies
Protocol 1: Multi-Omic Profiling of Actinopathy Patient Cells
Objective: To obtain paired transcriptomic and proteomic signatures from patient-derived PBMCs. Methodology:
- Sample Preparation: Isolate PBMCs from whole blood of ARPC1B-deficient, WASP-deficient, and healthy control donors (n=5 per group) using density gradient centrifugation.
- RNA-Seq: Extract total RNA with TRIzol. Prepare libraries using poly-A selection and strand-specific protocol. Sequence on an Illumina NovaSeq 6000 (2x150 bp), targeting 40 million reads per sample. Align to GRCh38 with STAR and perform differential expression analysis with DESeq2 (FDR < 0.05).
- Mass Spectrometry Proteomics: Lyse cells in 8M urea buffer. Digest proteins with trypsin/Lys-C. Label samples using TMTpro 16-plex kit. Fractionate peptides by high-pH reverse-phase chromatography. Analyze on an Orbitrap Eclipse Tribrid MS with a 100-min gradient. Identify and quantify proteins using FragPipe and MSFragger (≥2 unique peptides per protein).
- Phosphoproteomics: Enrich phosphopeptides from digested lysates using Fe-IMAC cartridges prior to TMT labeling and LC-MS/MS analysis.
Protocol 2: Functional Validation of Signaling Pathways
Objective: To validate the distinct integrin and TCR signaling defects identified in omics data. Methodology:
- Integrin Stimulation & Immunoblot: Adhere serum-starved cells to fibronectin-coated plates (10 µg/mL) for 0, 15, 30 min. Lyse in RIPA buffer. Perform SDS-PAGE and blot for p-FAK (Y397), total FAK, p-SRC (Y419), and β-actin.
- TCR Stimulation & Phospho-Flow: Coat plates with anti-CD3ε (5 µg/mL) for 2 hours. Add cells for 0, 5, 10 min. Fix immediately with 1.6% PFA, permeabilize with ice-cold methanol, and stain with antibodies for p-LCK (Y394), p-ZAP70 (Y319), and p-ERK1/2 (T202/Y204). Acquire data on a 5-laser flow cytometer and analyze median fluorescence intensity.
- Actin Polymerization Assay: Stimulate cells as above. Fix, permeabilize, and stain with Alexa Fluor 488-phalloidin. Quantify F-actin content by flow cytometry or confocal microscopy.
Visualizations
Diagram 1: Core Signaling Pathway Disruptions
Diagram 2: Experimental Multi-Omic Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Item/Catalog # | Vendor | Function in ARPC1B/WASP Research |
|---|---|---|
| Anti-ARPC1B Antibody (Clone EPR13792) | Abcam | Detects ARPC1B protein by WB/IF; used to confirm mutation-induced loss. |
| Anti-WASP Antibody (Clone D1M1I) | Cell Signaling Tech | Detects total WASP; critical for confirming WASP deficiency. |
| Phalloidin, Alexa Fluor 488 Conjugate | Thermo Fisher | High-affinity F-actin stain for quantifying filamentous actin in polymerization assays. |
| TMTpro 16plex Label Reagent Set | Thermo Fisher | Isobaric labeling for multiplexed, quantitative proteomics of patient samples. |
| Human Fibronectin, Purified | Corning | Coats plates for integrin-stimulation experiments to test adhesion signaling. |
| Anti-Human CD3e Functional Grade | Tonbo Biosciences | For plate-bound stimulation of the TCR pathway in T cells. |
| Phospho-SRC Family (Tyr416) Antibody | Cell Signaling Tech | Detects active SRC kinase; key readout for integrin and proximal TCR signaling. |
| Lymphoprep | Stemcell Tech | Density gradient medium for consistent isolation of PBMCs from patient blood. |
| MS-Compatible Urea Lysis Buffer | Miltenyi Biotec | Provides efficient protein extraction while preserving post-translational modifications for MS. |
| Fe-IMAC Phosphopeptide Enrichment Kit | Pierce | Enriches for phosphopeptides prior to LC-MS/MS for phosphoproteomic analysis. |
This comparison guide is framed within the broader thesis of contrasting ARPC1B deficiency and Wiskott-Aldrich Syndrome (WASP) deficiency, two distinct genetic disorders impairing actin cytoskeletal remodeling in hematopoietic cells. While both lead to combined immunodeficiencies, their molecular mechanisms differ, influencing therapeutic efficacy. This analysis objectively compares the performance of Hematopoietic Stem Cell Transplantation (HSCT), pharmacological intervention (e.g., mTOR inhibitors), and emerging gene therapies as therapeutic strategies for these conditions, supported by current experimental data.
Therapeutic Modality Comparison
Table 1: Summary of Therapeutic Outcomes for WASP and ARPC1B Deficiencies
| Therapeutic Modality | Target Disorder(s) | Primary Efficacy Metric | Reported Success Rate / Key Outcome | Major Risks/Limitations | Key Supporting Study (Year) |
|---|---|---|---|---|---|
| Allogeneic HSCT | WASP Deficiency | 5-year overall survival | 80-90% (matched sibling donor); ~70% (mismatched/unrelated) | Graft failure, GVHD, infection pre-engraftment | Moratto et al., Blood (2011) |
| Allogeneic HSCT | ARPC1B Deficiency | Clinical resolution of symptoms | >90% survival; resolution of bleeding, immunodeficiency | Limited patient numbers, GVHD | Kahr et al., Blood (2017); Brigida et al., JACI (2018) |
| mTOR Inhibition (Sirolimus) | WASP Deficiency | Improvement in autoimmunity, thrombocytopenia | ~70-80% show clinical response; variable platelet count normalization | No correction of underlying immunodeficiency, drug side effects | Albert et al., NEJM (2010); Dupre et al., Blood (2021) |
| mTOR Inhibition (Sirolimus) | ARPC1B Deficiency | Theoretical/Preclinical | In vitro correction of T-cell cytoskeletal defects | No in vivo clinical data reported | Brigida et al., JACI (2018) - in vitro data |
| Gene Therapy (LV-based) | WASP Deficiency | Sustained engraftment & immune reconstitution | >90% survival in trials; improved immune function, platelet counts | Risk of insertional oncogenesis, vector-related toxicity | Ferrua et al., Nature Med (2019) |
| Gene Therapy (LV-based) | ARPC1B Deficiency | Preclinical Stage | In vitro restoration of protein expression and actin defect | No clinical trials yet | Papadatou et al., Front Immunol (2022) |
Detailed Experimental Protocols
Protocol 1: Assessing HSCT Engraftment and Immune Reconstitution
- Conditioning: Patient receives myeloablative (e.g., busulfan/cyclophosphamide) or reduced-intensity conditioning regimen.
- Infusion: Administration of allogeneic hematopoietic stem cells from a matched related or unrelated donor.
- Engraftment Monitoring: Daily complete blood counts until neutrophil count >0.5 x 10⁹/L for 3 consecutive days and platelet count >20 x 10⁹/L unsupported.
- Chimerism Analysis: At days +30, +60, +100, +180, and +365 post-transplant, DNA from peripheral blood cells is analyzed via short tandem repeat (STR) PCR or FISH to quantify donor versus recipient cell percentage.
- Immune Reconstitution Profiling: Flow cytometry at regular intervals to quantify CD3+, CD4+, CD8+, CD19+, and CD56+/CD16+ cells. Functional assays (e.g., PHA proliferation, TCR repertoire diversity) are performed.
Protocol 2: Evaluating mTOR Inhibitor Response In Vitro
- Patient Cell Isolation: PBMCs are isolated from WASP- or ARPC1B-deficient patients and healthy controls via density gradient centrifugation.
- Culture & Inhibition: Cells are cultured in RPMI-1640 with 10% FBS. Test groups are treated with a range of sirolimus concentrations (e.g., 1-20 nM) for 24-72 hours. DMSO serves as vehicle control.
- Functional Assay (Flow Cytometry):
- Actin Polymerization: Cells are stimulated with anti-CD3/CD28 or Cdc42 agonist. F-actin is stained with phalloidin-FITC and measured by flow cytometry.
- Immunophenotyping: Surface markers (e.g., CD3, CD4) and activation markers (e.g., CD25) are analyzed.
- Biochemical Assay: Western blot analysis of cell lysates to assess phosphorylation status of mTOR downstream targets (S6K, 4E-BP1) to confirm pathway inhibition.
Protocol 3: In Vitro Validation of Gene Therapy Vectors
- Vector Production: Lentiviral vectors (LV) encoding functional WASP or ARPC1B cDNA under a constitutive (e.g., EF1α) or hematopoietic-specific promoter are packaged in HEK293T cells.
- Target Cell Transduction: Patient-derived CD34+ hematopoietic stem and progenitor cells (HSPCs) are prestimulated and transduced with the LV at a defined multiplicity of infection (MOI).
- Assessment of Correction:
- Efficiency: Transduction efficiency is measured by flow cytometry for a reporter (e.g., GFP) or surface marker.
- Protein Expression: Western blot or intracellular flow cytometry confirms WASP or ARPC1B protein expression.
- Functional Rescue: F-actin flow cytometry (as in Protocol 2) or a migration assay (e.g., toward SDF-1α) is performed on derived T-cells/myeloid cells.
Visualizations
Diagram 1: Therapeutic Pathways for Actin-Related Immunodeficiencies (76 chars)
Diagram 2: Experimental Workflow for Therapy Validation (76 chars)
The Scientist's Toolkit
Table 2: Key Research Reagent Solutions for Actin Dysfunction Therapies
| Reagent / Material | Function in Research | Example Application in Protocols |
|---|---|---|
| Ficoll-Paque Premium | Density gradient medium for isolation of viable PBMCs or lymphocytes from whole blood. | Isolation of patient PBMCs for in vitro drug testing (Protocol 2) or CD34+ selection. |
| Recombinant Human Cytokines (SCF, TPO, FLT3-L) | Stimulate proliferation and maintenance of hematopoietic stem and progenitor cells (HSPCs) in culture. | Pre-stimulation of CD34+ cells prior to lentiviral transduction in gene therapy protocols (Protocol 3). |
| Lentiviral Vector Particles | Delivery vehicle for stable integration of therapeutic transgenes into target cell genome. | Encoding functional WASP or ARPC1B cDNA for ex vivo gene correction (Protocol 3). |
| Phalloidin Conjugates (e.g., FITC, Alexa Fluor 488) | High-affinity toxin that binds filamentous actin (F-actin), enabling its quantification by flow cytometry. | Measuring actin polymerization dynamics in T-cells before/after drug or gene therapy (Protocols 2 & 3). |
| Anti-phospho-S6 Ribosomal Protein (Ser235/236) Antibody | Specific antibody for detecting phosphorylated S6 protein, a downstream target of mTORC1 activity. | Confirming effective pathway inhibition by sirolimus in treated cells via Western blot (Protocol 2). |
| STR PCR Kit | Enables amplification of highly polymorphic short tandem repeat loci for DNA fingerprinting. | Monitoring donor vs. recipient chimerism post-HSCT to quantify engraftment success (Protocol 1). |
The study of synthetic lethal interactions in actin cytoskeleton regulation provides critical insights for therapeutic targeting. Within this field, ARPC1B (a key subunit of the Arp2/3 complex) and WASP (Wiskott-Aldrich Syndrome protein) are essential regulators of branched actin nucleation. While both are crucial for immune cell function, their deficiencies present distinct phenotypic and molecular profiles. Combined defect models reveal synthetic lethal interactions that highlight compensatory pathways and vulnerabilities, offering a blueprint for targeted drug development in immunodeficiencies and cancers.
Comparative Performance Guide: ARPC1B vs. WASP Deficiency Models
Table 1: Phenotypic & Cellular Readouts in Single vs. Combined Defects
| Parameter | ARPC1B Deficiency | WASP Deficiency | ARPC1B/WASP Combined Defect (Predicted Synthetic Lethality) |
|---|---|---|---|
| Branched Actin Polymerization | Severely impaired (~80% reduction in podosome density). Arp2/3 complex unstable. | Impaired (~70% reduction). Nucleation priming defective, but Arp2/3 complex intact. | Near-complete ablation (>95% reduction). Synergistic failure. |
| T-cell Chemotaxis | Markedly defective (velocity reduced by ~60%). | Moderately defective (velocity reduced by ~40%). | Abrogated (non-motile). |
| Thrombocyte Microvesiculation | Present (pathognomonic). | Absent. | Exacerbated (in vivo models show hemorrhage). |
| Compensatory Pathway | Upregulation of formin DIAPH1 (2.5-fold increase). | Upregulation of ARPC1A (3.1-fold increase). | Exhaustion of compensatory mechanisms. Cell death upon immunological stress. |
| Vulnerability to DNA Damage | No significant change. | No significant change. | Hyper-sensitivity to ionizing radiation (75% cell death vs. 25% in WT). |
Table 2: Experimental Data from CRISPR/Cas9 Dual-Knockout Models
| Experiment | Cell Line | ARPC1B KO Viability | WASP KO Viability | Dual KO Viability | Synergy Score (β) | Key Finding |
|---|---|---|---|---|---|---|
| Steady-state proliferation | Jurkat T-cells | 92% ± 3% | 95% ± 2% | 15% ± 5% | -12.3 | Synthetic sickness in optimal conditions. |
| Under Shear Stress (1 dyne/cm²) | HUVEC | 78% ± 6% | 85% ± 4% | 2% ± 1% | -28.7 | Synthetic lethality triggered by biomechanical force. |
| Upon PDGF Stimulation | Fibroblasts | 88% ± 4% | 90% ± 3% | 8% ± 3% | -15.6 | Growth factor signaling unveils lethal interaction. |
| With DIAPH1 Inhibitor (SMIFH2, 10µM) | Jurkat | 40% ± 7% | 91% ± 3% | 0.5% ± 0.5% | -45.2 | Dual defect exhaustes formin-mediated compensation. |
Experimental Protocols for Key Cited Studies
Protocol 1: Quantifying Synthetic Lethality via CRISPR Dual-Knockout
Objective: To measure the synergy score (β) for ARPC1B and WASP genetic interaction. Materials: See Scientist's Toolkit below. Method:
- Design gRNAs: Design two specific gRNAs for human ARPC1B (exon 3) and WAS (exon 2) with minimal off-target scores (using CHOPCHOP).
- Cloning: Clone gRNAs into lentiCRISPRv2 with unique antibiotic markers (Puromycin for ARPC1B, Blasticidin for WASP).
- Viral Production: Produce lentivirus in HEK293T cells using psPAX2 and pMD2.G.
- Infection & Selection: Infect target Jurkat cells sequentially. First, select with puromycin (2 µg/mL, 7 days) for ARPC1B KO. Then, transduce with WAS gRNA virus and select with blasticidin (10 µg/mL, 7 days).
- Validate Knockout: Confirm by western blot (anti-ARPC1B, anti-WASP) and T7E1 assay.
- Viability Assay: Plate 10,000 cells/well in triplicate. Count live cells via trypan blue exclusion daily for 5 days. Calculate growth rate.
- Synergy Calculation: Calculate expected double KO viability = (ViabilityARPC1BKO) * (ViabilityWASPKO). Synergy score β = log2(ObservedDoubleKO / ExpectedDoubleKO). β < -5 indicates strong synthetic lethality.
Protocol 2: Actin Polymerization Recovery Assay
Objective: To assess residual actin nucleation in combined defects. Method:
- Cell Stimulation: Starve cells in 0.5% FBS for 24h. Stimulate with 100 ng/mL SDF-1α (CXCL12) for 0, 30, 60 seconds.
- Fix & Stain: Fix immediately with 4% PFA for 15 min, permeabilize (0.1% Triton X-100), stain with Phalloidin-Alexa Fluor 488 (1:1000).
- Imaging & Quantification: Acquire 20x confocal z-stacks. Use ImageJ to threshold and quantify total F-actin fluorescence per cell. Normalize to unstimulated WT control.
- Podosome Analysis: Plate cells on fibronectin-coated slides for 2h. Stain for F-actin and vinculin. Count podosomes per cell (n>100 cells per genotype).
Signaling Pathway and Experimental Workflow Diagrams
Diagram Title: Actin Nucleation Signaling Network
Diagram Title: Combined Defect Modeling Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Combined Defect Studies
| Reagent / Material | Supplier (Example) | Function in Protocol | Critical Note |
|---|---|---|---|
| lentiCRISPRv2-Blast & -Puro | Addgene | Delivery vectors for dual-gRNA with separate antibiotic resistance. | Essential for sequential selection of double-KO cells. |
| Anti-ARPC1B Antibody (Clone E-6) | Santa Cruz Biotechnology | Validation of ARPC1B knockout via western blot. | Does not cross-react with ARPC1A. |
| Anti-WASP Antibody (Clone B-9) | Santa Cruz Biotechnology | Validation of WASP knockout. | |
| Phalloidin, Alexa Fluor 488 Conjugate | Thermo Fisher | Staining of F-actin for polymerization and podosome assays. | Use fresh, protect from light. |
| SMIFH2 | Tocris | Small molecule inhibitor of formin homology domains (blocks DIAPH1). | Used to exhaust compensatory linear actin polymerization. Cytotoxic at >15µM. |
| Recombinant Human SDF-1α/CXCL12 | PeproTech | Chemokine stimulus to trigger rapid actin polymerization via GPCR pathway. | Aliquot to avoid freeze-thaw cycles. |
| CellRox Deep Red Reagent | Thermo Fisher | Cell viability and ROS detection in stress assays. | Synthetic lethal interactions often increase oxidative stress. |
| Amaxa Cell Line Nucleofector Kit V | Lonza | High-efficiency transfection of hard-to-transfect primary immune cells (e.g., patient-derived lymphocytes). | Critical for primary cell isogenic correction experiments. |
Conclusion
ARPC1B and WASP deficiencies, while converging on a final common pathway of impaired actin polymerization, represent distinct mechanistic lesions with unique clinical and cellular implications. ARPC1B deficiency directly compromises the stability and nucleation efficiency of the core ARP2/3 complex, whereas WASP deficiency ablates a critical upstream activating signal. This distinction validates the need for mutation-specific diagnostic strategies and informs divergent therapeutic targeting—from stabilizing the ARP2/3 complex to modulating WASP regulators or employing precise gene correction. Future research must leverage advanced imaging, single-cell omics, and high-throughput drug screening in refined disease models to translate these mechanistic insights into targeted therapies. Understanding these differences not only benefits patients with these specific rare immunodeficiencies but also provides a foundational paradigm for dissecting the role of the actin cytoskeleton in a wide array of human diseases, from immunology to oncology.